Coordination Polymerization Explained: The Secret Behind HDPE and PP
JUL 3, 2025 |
Coordination polymerization is a fascinating and essential process in the world of polymer chemistry, playing a crucial role in the production of high-density polyethylene (HDPE) and polypropylene (PP). These polymers are ubiquitous in our daily lives, found in everything from plastic bottles to car parts, from packaging materials to textiles. Understanding the science behind their production not only highlights the innovation in chemical engineering but also emphasizes the importance of polymer chemistry in modern manufacturing.
The Basics of Coordination Polymerization
At its core, coordination polymerization is a mechanism that involves the formation of long polymer chains through the coordination of monomers to a metal catalyst center. This process is distinct from other forms of polymerization, such as free-radical polymerization, as it relies on the unique properties of metal complexes, typically transition metals like titanium or zirconium, to facilitate the polymerization process.
The Importance of Catalysts
Catalysts are the unsung heroes of coordination polymerization. They determine the efficiency, speed, and control of the polymerization process. The most commonly used catalysts in coordination polymerization are Ziegler-Natta catalysts and metallocene catalysts. Ziegler-Natta catalysts, discovered in the 1950s, revolutionized the polymer industry by enabling the production of highly regular polymer structures. These catalysts typically consist of titanium compounds used in conjunction with organoaluminum compounds.
Metallocene catalysts, on the other hand, are a more recent development, offering even greater control over the polymerization process. They consist of a transition metal sandwiched between two cyclopentadienyl anions, providing a versatile platform for producing polymers with specific molecular architectures and properties.
HDPE and PP: Products of Coordination Polymerization
High-Density Polyethylene (HDPE)
HDPE is one of the most widely used plastics in the world. Its production through coordination polymerization results in a polymer with high strength, stiffness, and resistance to chemicals and moisture. The structured and linear chains formed during the polymerization process give HDPE its characteristic density and toughness. This makes it an ideal material for applications such as plastic bottles, piping, and even plastic lumber.
Polypropylene (PP)
Polypropylene, another product of coordination polymerization, is known for its versatility and economic efficiency. It is produced through a similar process as HDPE but with different catalysts and conditions that yield a polymer with unique properties. PP boasts excellent chemical resistance, fatigue resistance, and a high melting point. Its ability to be easily molded and shaped makes it a popular choice for automotive parts, textiles, and consumer goods.
The Role of Polymerization Conditions
The conditions under which coordination polymerization occurs—such as temperature, pressure, and the concentration of catalysts—can significantly affect the properties of the resulting polymer. By manipulating these parameters, chemists can tailor the polymerization process to produce materials with specific characteristics suited to various applications. For instance, altering the catalyst system or reaction conditions can control the polymer's molecular weight, tacticity, and crystallinity.
Environmental and Economic Considerations
While coordination polymerization has enabled the production of versatile and durable plastics, it also raises important environmental and economic considerations. The widespread use of HDPE and PP has led to increased plastic waste and environmental pollution. As such, there is a growing emphasis on developing more sustainable polymerization processes, recycling methods, and biodegradable alternatives to address these challenges.
Conclusion
Coordination polymerization is a remarkable process that underpins the production of some of the most commonly used plastics in the modern world. Through the careful selection of catalysts and control of polymerization conditions, scientists can produce polymers with a wide range of properties and applications. As the demand for sustainable and efficient materials continues to grow, advancements in coordination polymerization will undoubtedly play a pivotal role in shaping the future of material science and engineering.
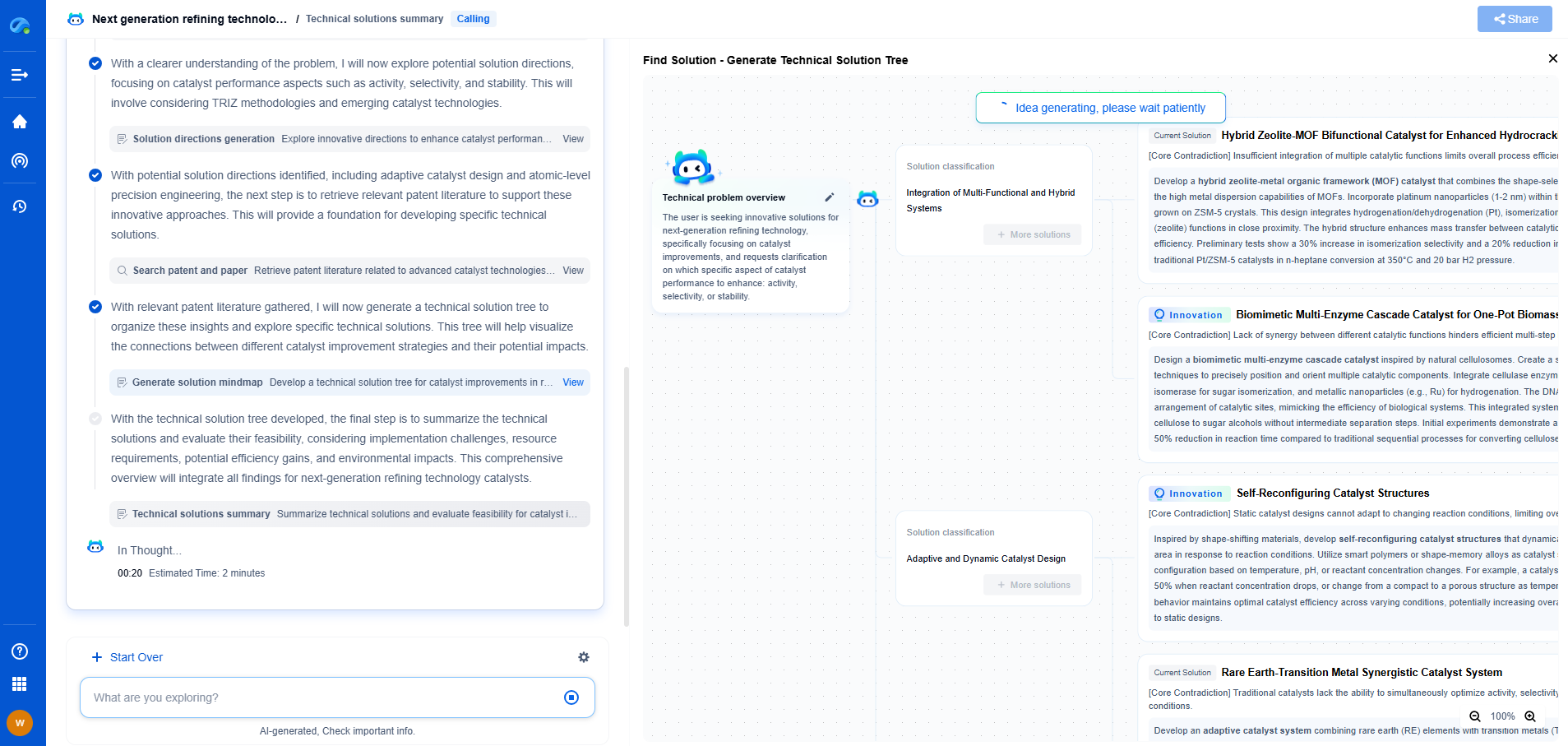
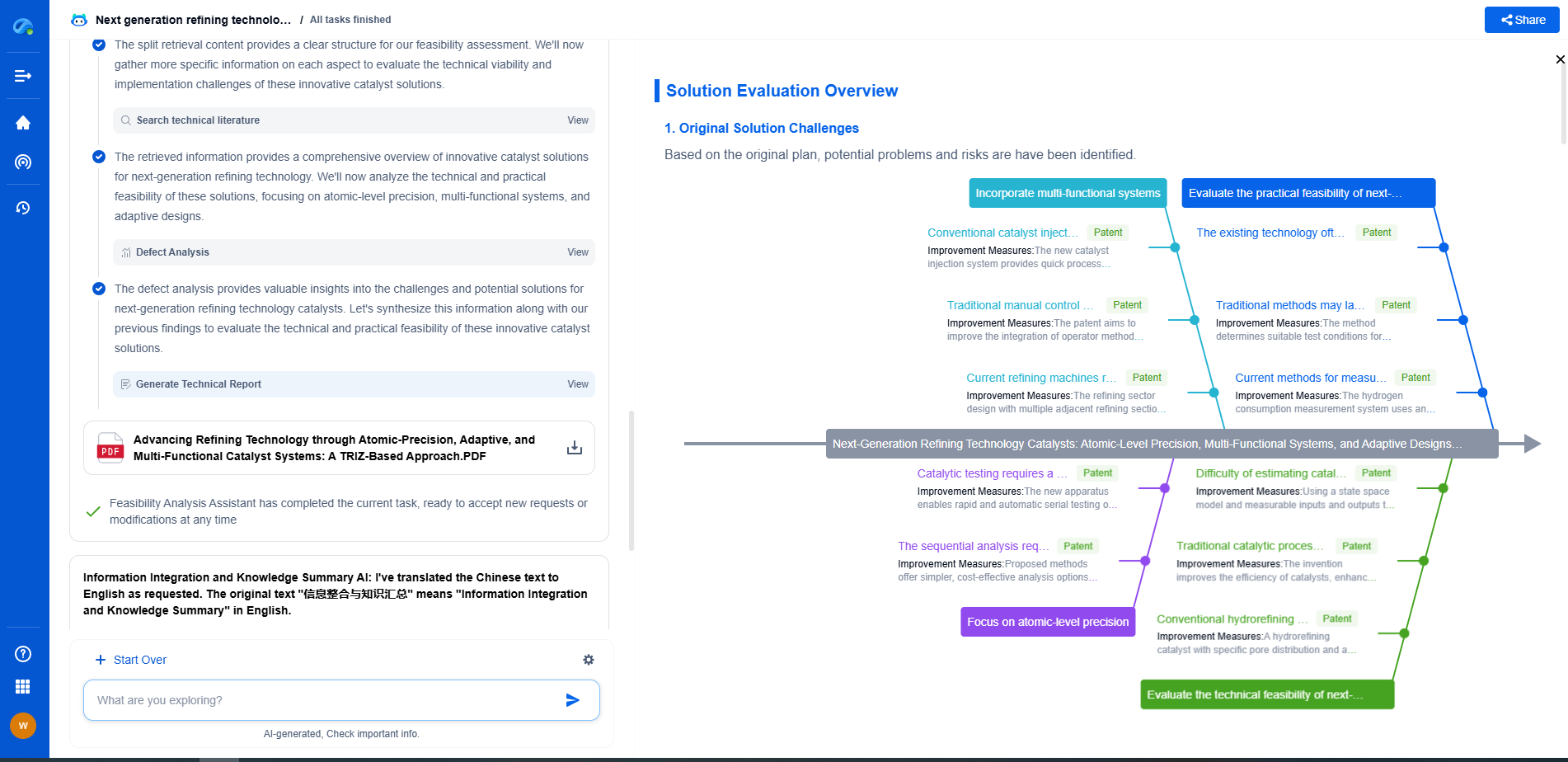
Transform Polymeric Innovation with Patsnap Eureka
From biodegradable polymers to high-performance composites, the world of polymeric compounds is evolving faster than ever—driven by the demands of sustainability, functional customization, and global IP competition. Whether you're exploring novel copolymer architectures, optimizing polymerization techniques, or tracking material patents in bioplastics, time-to-insight is everything.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're working on next-gen packaging films, bio-based resins, smart polymers for electronics, or new thermal-resistant composites, Eureka accelerates your journey from idea to patent to product—with unmatched clarity and speed.
🔍 Experience how Eureka can power your polymer R&D with AI intelligence—start your free trial today and unlock the future of materials innovation.