Differences in Reaction Mechanisms: PEMFC vs SOFC Fuel Cells
JUN 20, 2025 |
Fuel cells are devices that convert chemical energy into electrical energy through electrochemical reactions. They are heralded for their efficiency, environmental benefits, and potential to provide clean energy solutions. Among the many types, Proton Exchange Membrane Fuel Cells (PEMFC) and Solid Oxide Fuel Cells (SOFC) are prominent, each having distinct reaction mechanisms, advantages, and limitations.
Proton Exchange Membrane Fuel Cells (PEMFC)
PEMFCs are widely used for applications requiring quick start-ups, like automobiles and portable devices. At their core is a proton-conducting polymer electrolyte membrane, which facilitates the electrochemical reactions by allowing protons to flow while blocking electrons.
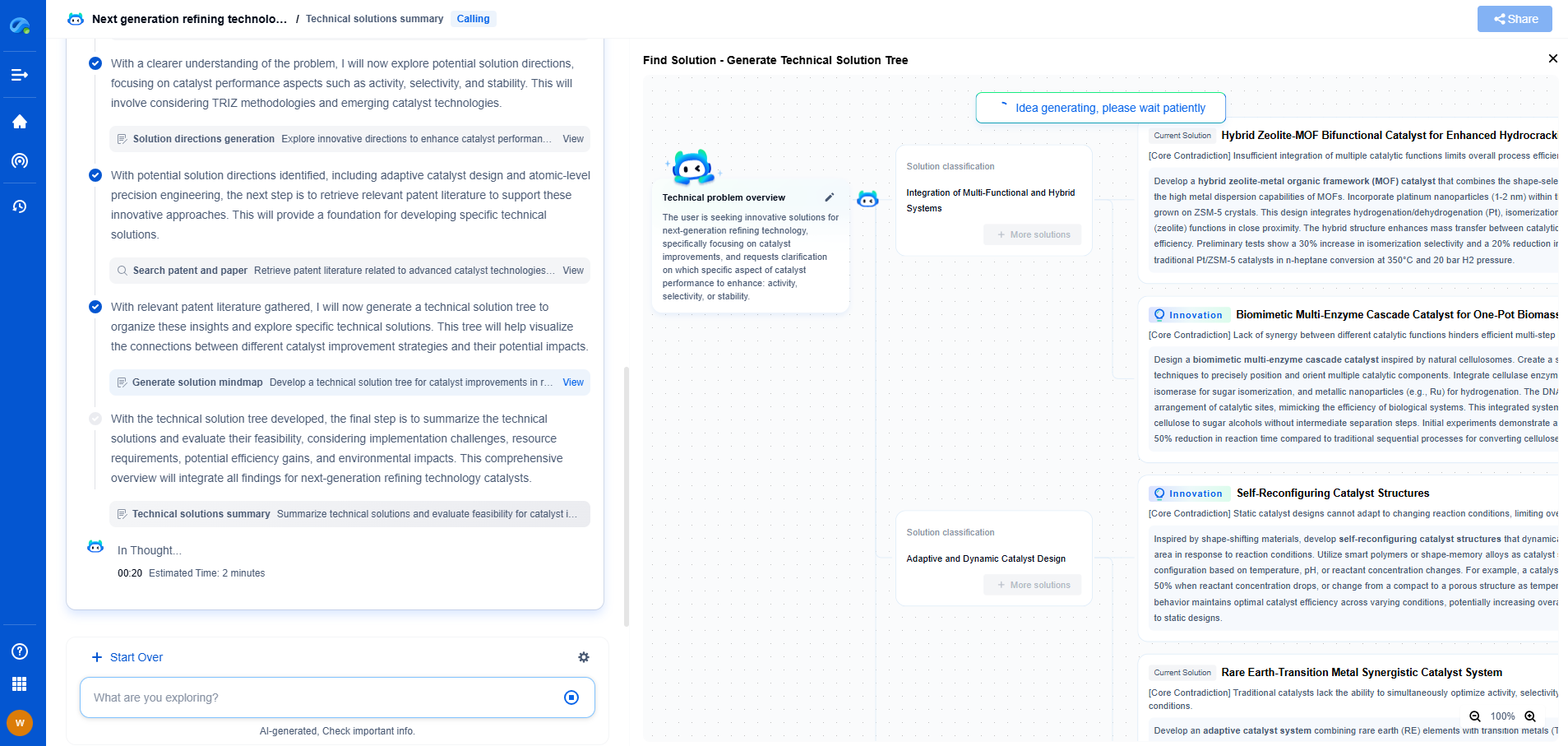
Reaction Mechanism of PEMFC
The PEMFC operates at relatively low temperatures, typically between 50°C to 100°C. The reactions in a PEMFC involve hydrogen and oxygen. At the anode, hydrogen gas is oxidized, releasing electrons and producing protons. These protons migrate through the electrolyte membrane to the cathode. Simultaneously, electrons travel through an external circuit providing electrical power.
At the cathode, oxygen gas reacts with the protons and electrons arriving from the external circuit, forming water. The overall reaction can be summarized as:
Anode: \( \text{H}_2 \rightarrow 2\text{H}^+ + 2\text{e}^- \)
Cathode: \( \text{O}_2 + 4\text{H}^+ + 4\text{e}^- \rightarrow 2\text{H}_2\text{O} \)
Overall: \( \text{2H}_2 + \text{O}_2 \rightarrow 2\text{H}_2\text{O} \)
Advantages and Limitations of PEMFC
The primary advantage of PEMFCs lies in their rapid start-up capability and operation at low temperatures, making them suitable for transportation applications. However, they require pure hydrogen as fuel and are sensitive to impurities, particularly carbon monoxide. The cost associated with the platinum catalyst and membrane materials also poses challenges.
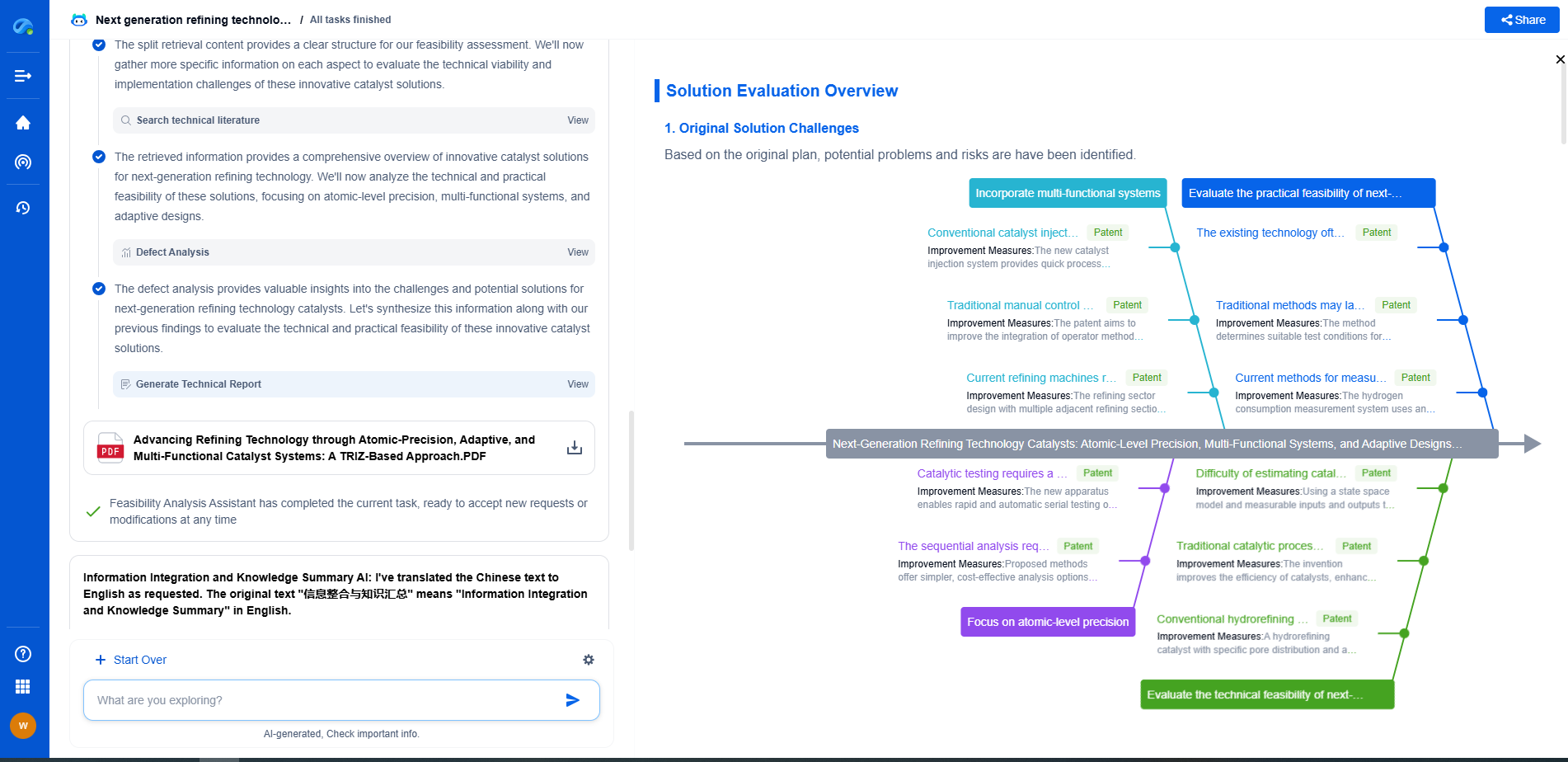
Solid Oxide Fuel Cells (SOFC)
SOFCs are high-temperature fuel cells that are versatile in fuel choice, including hydrogen, carbon monoxide, and hydrocarbons. Their unique design involves a solid ceramic electrolyte, typically made of zirconia stabilized with yttria.
Reaction Mechanism of SOFC
Operating at temperatures ranging from 600°C to 1000°C, SOFCs enable internal reforming of hydrocarbon fuels and high efficiency. The high-temperature environment facilitates the movement of oxygen ions across the electrolyte.
At the anode, hydrogen or carbon monoxide reacts with oxygen ions from the cathode, producing water, carbon dioxide, and releasing electrons. These electrons travel through an external circuit to the cathode.
At the cathode, oxygen gas acquires electrons from the external circuit and forms oxygen ions, which migrate through the electrolyte to the anode. The overall reaction can be summarized as:
Anode: \( \text{H}_2 + \text{O}^{2-} \rightarrow \text{H}_2\text{O} + 2\text{e}^- \)
Cathode: \( \text{O}_2 + 4\text{e}^- \rightarrow 2\text{O}^{2-} \)
Overall: \( \text{H}_2 + \text{O}_2 \rightarrow \text{H}_2\text{O} \)
Advantages and Limitations of SOFC
The high operating temperatures of SOFCs allow them to use a variety of fuels and achieve better efficiency compared to PEMFCs. Additionally, they do not require precious metal catalysts. However, these temperatures also present challenges in terms of material durability and long start-up times, limiting their use in mobile applications.
Comparing PEMFC and SOFC
PEMFCs and SOFCs represent two distinct technological paradigms in fuel cell design. PEMFCs are advantageous for applications requiring quick start-ups and low-temperature operations, while SOFCs excel in stationary applications due to their fuel flexibility and higher efficiency.
Despite these differences, both types of fuel cells are integral to the transition towards cleaner energy systems. Innovations in materials and design continue to enhance their viability across various sectors, paving the way for a sustainable energy future.
Conclusion
Understanding the differences in reaction mechanisms between PEMFCs and SOFCs is crucial for optimizing their use in appropriate applications. While PEMFCs offer advantages in certain sectors, SOFCs promise broader fuel compatibility and efficiency. As research progresses, both types are likely to play significant roles in meeting future energy demands.
Accelerate Breakthroughs in Fuel Cell and Battery Innovation—with the Power of AI
From solid-state battery breakthroughs to high-efficiency hydrogen fuel cells, keeping pace with fast-evolving chemistries, global patent landscapes, and emerging application pathways is an ever-growing challenge for R&D and IP professionals.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're optimizing cathode formulations, evaluating electrolyte stability, or navigating the crowded patent space around battery pack design, Eureka empowers you to move faster and with greater confidence.
Start your journey with Patsnap Eureka today—streamline your research, enhance decision-making, and power the future of energy with AI-driven clarity.