Donnan exclusion in pharmaceutical ultrafiltration processes
JUL 25, 2025 |
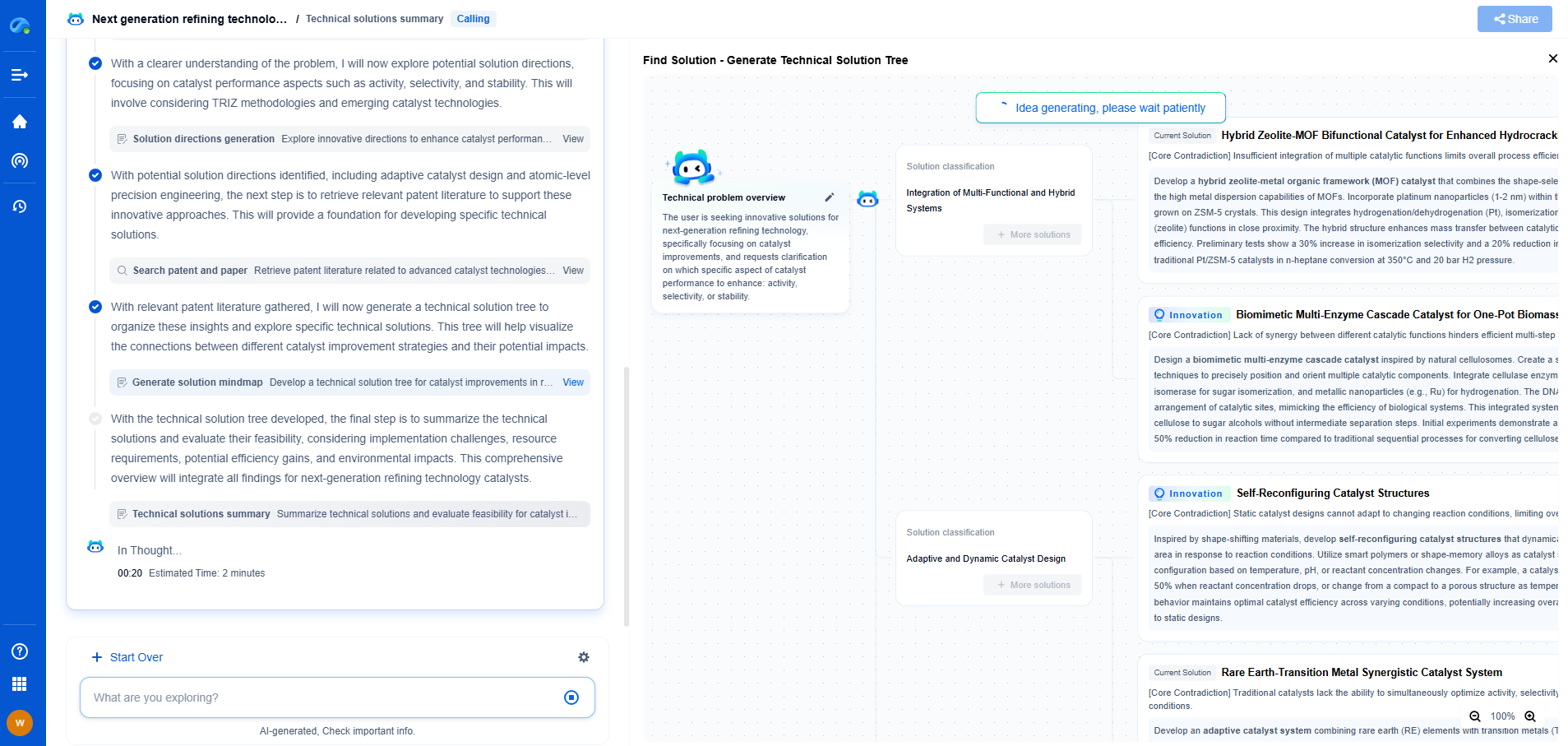
Introduction to Ultrafiltration in Pharmaceuticals
Ultrafiltration is a pivotal technology in the pharmaceutical industry, providing an efficient means for separating macromolecules from smaller molecules and solvents. This membrane-based process operates under pressure to facilitate the selective passage of solvents and small solute molecules through a semipermeable membrane, thereby retaining larger molecules. Ultrafiltration is crucial in drug formulation, purification, and protein concentration. However, an often overlooked yet significant phenomenon that impacts ultrafiltration efficiency is Donnan exclusion.
The Concept of Donnan Exclusion
Donnan exclusion, also known as the Donnan effect, arises due to the presence of charged species on either side of a semipermeable membrane. This electrochemical phenomenon dictates the distribution of ions across the membrane, thereby influencing the separation process. In pharmaceutical ultrafiltration, Donnan exclusion affects the retention and permeation rates of charged molecules, which can have profound implications for the purification of drugs and biologics.
Role of Charge in Ultrafiltration
Charged solutes in ultrafiltration interact with the membrane, which may also possess charge properties. For instance, when the membrane and the solutes bear the same charge, electrostatic repulsion occurs, leading to enhanced solute retention and reduced permeability. Conversely, if the charges are opposite, attraction may lead to increased passage through the membrane. This charge-based selectivity is central to understanding how Donnan exclusion impacts ultrafiltration processes.
Factors Influencing Donnan Exclusion
Several factors influence the extent of Donnan exclusion during ultrafiltration. The ionic strength of the solution, membrane charge density, and pH levels are primary determinants. Higher ionic strength can mitigate the effects of Donnan exclusion by shielding charge interactions, while membrane charge density and pH can alter the degree of ionization of solutes, thereby impacting their interaction with the membrane. Understanding these parameters is essential for optimizing ultrafiltration processes in pharmaceutical applications.
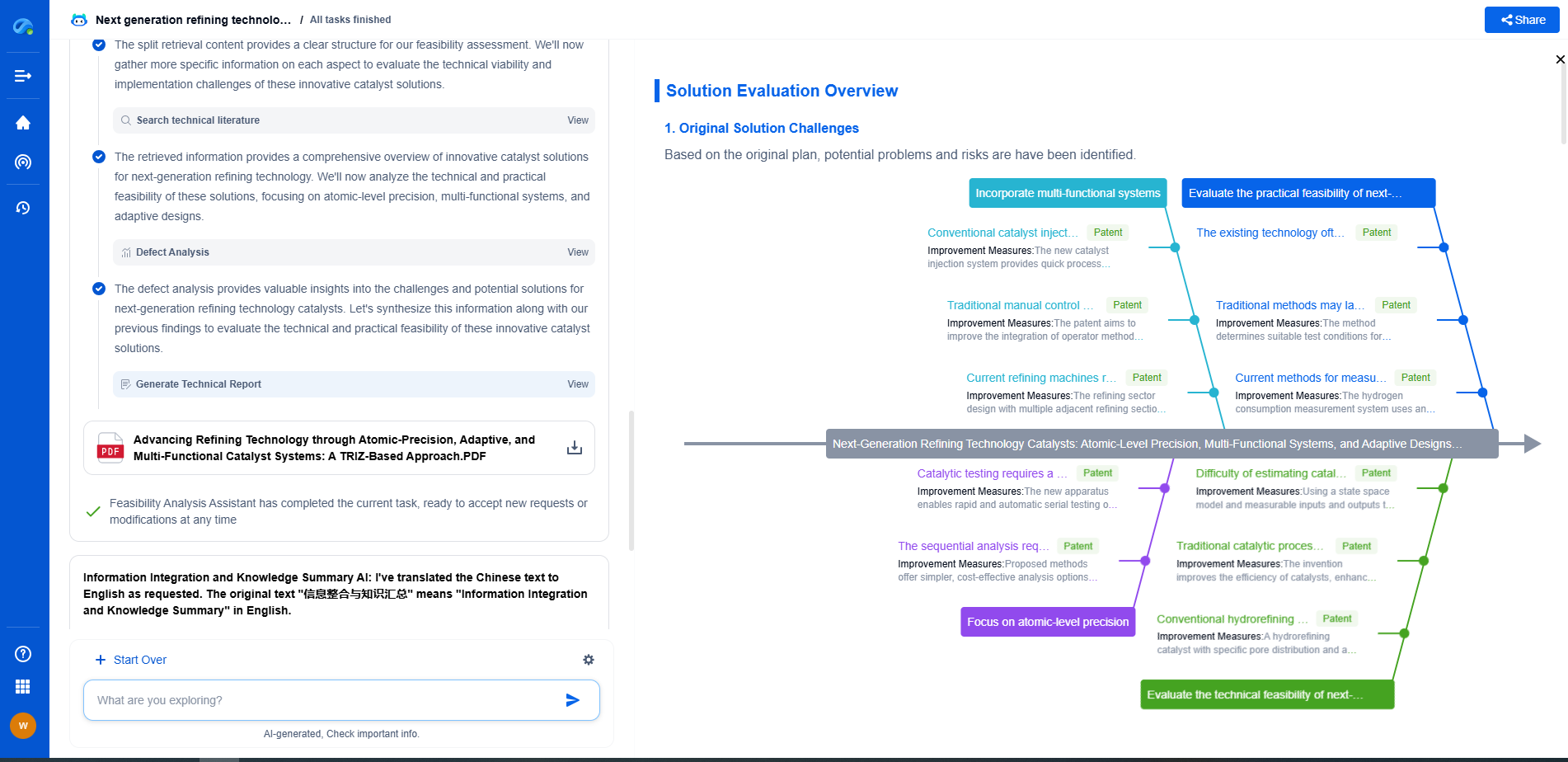
Implications for Pharmaceutical Applications
In pharmaceutical manufacturing, where purity and yield are paramount, the implications of Donnan exclusion are particularly significant. For instance, during the purification of therapeutic proteins, Donnan exclusion can influence the separation of proteins from salts and other impurities. Similarly, in drug formulation, this phenomenon can affect the concentration and stability of active pharmaceutical ingredients (APIs).
To address these challenges, pharmaceutical scientists often employ strategies to mitigate or exploit Donnan exclusion. Adjusting the pH of the solution, modifying ionic strength, or selecting membranes with specific charge properties are common tactics used to optimize ultrafiltration outcomes. By carefully considering the influence of Donnan exclusion, pharmaceutical processes can be fine-tuned to achieve desired purity and efficiency levels.
Future Directions and Research
Ongoing research in the field of ultrafiltration seeks to further elucidate the mechanisms of Donnan exclusion and its impact on membrane processes. Advances in membrane technology, such as the development of novel materials with tailored charge characteristics, hold promise for enhancing the selectivity and efficiency of ultrafiltration. Additionally, computational modeling and simulation are increasingly being used to predict and optimize the effects of Donnan exclusion in complex pharmaceutical systems.
Conclusion
Donnan exclusion is a critical factor in the ultrafiltration processes employed in pharmaceutical production. Its influence on ion distribution and separation efficiency highlights the need for careful consideration and control of charge interactions. By understanding and managing Donnan exclusion, pharmaceutical manufacturers can enhance the quality and efficacy of their products, ensuring that ultrafiltration remains a cornerstone of modern pharmaceutical processes. As research in this field continues to advance, new opportunities for innovation and optimization are likely to emerge, paving the way for more efficient and effective pharmaceutical manufacturing techniques.
From next-generation membrane materials to high-efficiency separation processes for pharmaceuticals, water treatment, food processing, or energy systems, the filtration & separation industry is rapidly evolving with a surge in material innovation, microstructure design, and process optimization.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're designing the next high-throughput filter, optimizing nanostructured surfaces, or exploring new separation media for emerging industries—Patsnap Eureka gives you AI-driven insights in seconds, helping you move from ideation to innovation with confidence.
🚀 Start your free trial today and experience how Eureka transforms filtration innovation—from reactive to predictive.