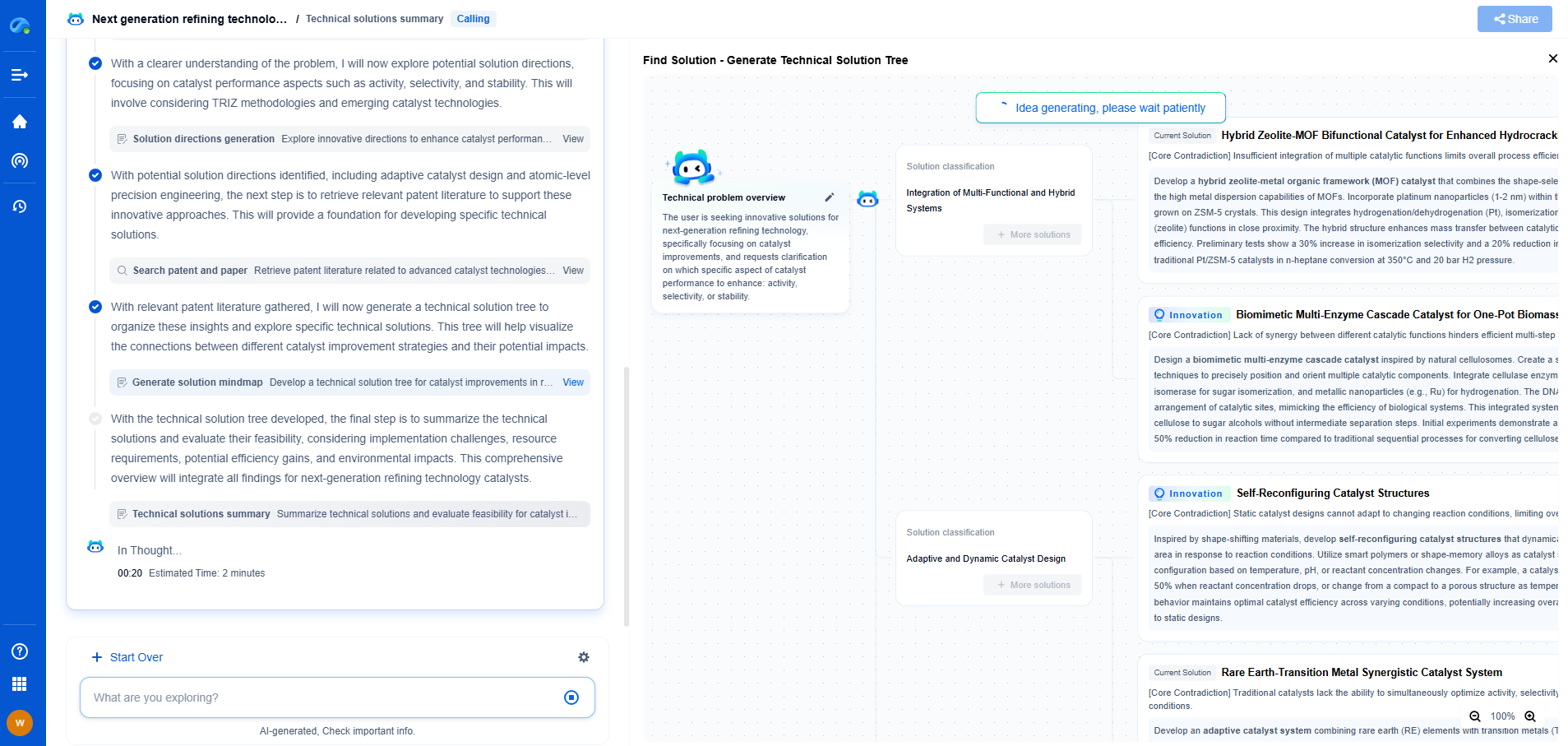
EPA Method 1603 vs 1604: Which one to use for microbial water testing?
JUL 25, 2025 |
When it comes to microbial water testing, the U.S. Environmental Protection Agency (EPA) provides standardized methods to ensure safety and accuracy in detecting harmful microorganisms. Two prominent methods in this realm are EPA Method 1603 and 1604. Each has its unique approach and applications, and choosing between them can be crucial depending on specific testing requirements. Understanding the distinctions and applications of these methods can aid in making an informed decision for water quality analysis.
Overview of EPA Method 1603
EPA Method 1603 is designed for detecting Escherichia coli (E. coli) in water samples, particularly in ambient waters and wastewater. This method employs the membrane filtration technique, which allows for the collection and analysis of bacteria present in water. A significant advantage of Method 1603 is its specificity; it uses a selective and differential medium known as Modified mTEC agar. This medium contains chromogenic substrates that result in a color change when E. coli is present, making it easy to identify and count colonies.
Another important aspect of Method 1603 is its precision in detecting E. coli, an indicator organism often associated with fecal contamination. This makes it particularly useful for assessing water body contamination and the effectiveness of wastewater treatment processes. The method's reliability is enhanced by its ability to produce results within 24 hours, facilitating timely decision-making in water safety management.
Overview of EPA Method 1604
In contrast, EPA Method 1604 is developed for the enumeration of total coliforms and E. coli in water using the membrane filtration technique on MI agar. This method is applicable to drinking water, surface water, and groundwater. The MI agar used in Method 1604 contains both chromogenic and fluorogenic substances that enable the simultaneous detection of total coliforms and E. coli. This dual detection capability provides a comprehensive assessment of microbial water quality.
One of the primary benefits of Method 1604 is its broad applicability. By targeting both total coliforms and E. coli, it provides an overall picture of microbial contamination, which can be essential in regulatory compliance and ensuring public health. Additionally, the method offers similar advantages in terms of rapid results, with the ability to detect and quantify target organisms within 24 hours.
Comparing Method 1603 and 1604: Key Differences
While both methods employ membrane filtration and are crucial for microbial water testing, their differences lie in their specific applications and target organisms. Method 1603 focuses solely on E. coli, providing high specificity in environments where fecal contamination is a primary concern. On the other hand, Method 1604 offers a broader scope by detecting both total coliforms and E. coli, making it suitable for a wider range of water types, including drinking water.
Another consideration is the choice of media: Method 1603 uses Modified mTEC agar, while Method 1604 uses MI agar. This difference in media affects the detection and identification process, with each method having its own strengths in terms of specificity and comprehensive analysis.
Choosing the Right Method for Your Needs
The decision between Method 1603 and 1604 should be guided by the specific context and requirements of the microbial water testing being conducted. For environments where identifying fecal contamination is critical, such as recreational waters or wastewater treatment facilities, Method 1603 may be more suitable due to its focus on E. coli. Meanwhile, Method 1604 is advantageous in scenarios where a broader assessment of microbial contamination is necessary, such as in drinking water systems or environmental monitoring.
Conclusion
Both EPA Method 1603 and 1604 offer robust and reliable means of assessing microbial water quality. Understanding their distinctions in scope, specificity, and application is essential in selecting the most appropriate method for your testing needs. By considering the type of water, regulatory requirements, and specific microbial targets, you can ensure that your water testing efforts are both effective and aligned with public health standards.
From next-generation membrane materials to high-efficiency separation processes for pharmaceuticals, water treatment, food processing, or energy systems, the filtration & separation industry is rapidly evolving with a surge in material innovation, microstructure design, and process optimization.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're designing the next high-throughput filter, optimizing nanostructured surfaces, or exploring new separation media for emerging industries—Patsnap Eureka gives you AI-driven insights in seconds, helping you move from ideation to innovation with confidence.
🚀 Start your free trial today and experience how Eureka transforms filtration innovation—from reactive to predictive.