Freeze Drying vs. Desublimation: Pharmaceutical Applications
JUL 25, 2025 |
Understanding Freeze Drying
Freeze drying, also known as lyophilization, is a dehydration process commonly used to preserve perishable materials and enhance the stability of pharmaceutical products. The process involves freezing the material, reducing the surrounding pressure, and then allowing the frozen water to sublime directly from the solid phase to the gas phase. This technique is highly effective in preserving the structural integrity and biological activity of heat-sensitive substances.
Freeze drying is typically employed in the production of antibiotics, vaccines, and protein-based drugs. It helps in maintaining the potency and viability of these substances without the need for refrigeration, which is crucial for ensuring patient safety and compliance. The preservation of structural integrity is particularly important for biopharmaceuticals, where slight changes can lead to reduced efficacy.
The Advantages of Freeze Drying
One of the primary advantages of freeze drying is its ability to produce a stable, easily reconstitutable product. The removal of water at low temperatures minimizes thermal degradation of the product, ensuring that heat-sensitive drugs remain effective. Additionally, the resulting product is lightweight and can be stored at room temperature, significantly reducing storage and transportation costs.
Freeze drying also offers the flexibility of producing a wide range of product formulations, from delicate biologics to robust small-molecule drugs. It provides excellent control over the final moisture content, which is crucial for maintaining product stability over extended periods.
Desublimation: A Different Approach
Desublimation, often referred to as deposition, is the process where a gas transitions directly into a solid without passing through the liquid phase. In the context of pharmaceuticals, desublimation can be used to capture volatile compounds from a gas phase and deposit them onto a solid substrate. While less commonly used than freeze drying, desublimation can be a valuable technique for specific applications.
Desublimation is particularly useful in capturing and purifying volatile compounds that might be challenging to handle through conventional methods. This process can also be instrumental in the development of certain drug delivery systems where the controlled deposition of active ingredients onto a substrate is required.
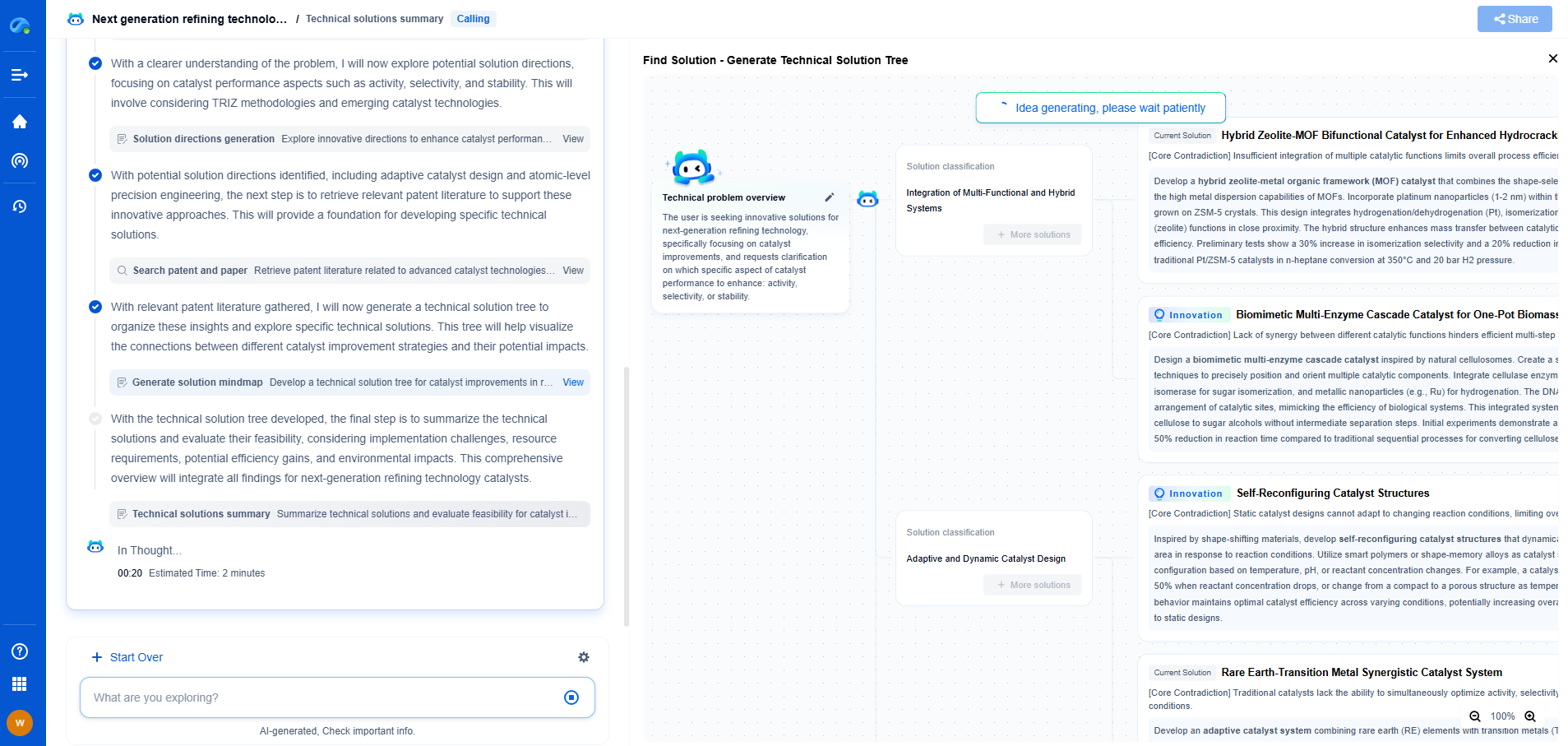
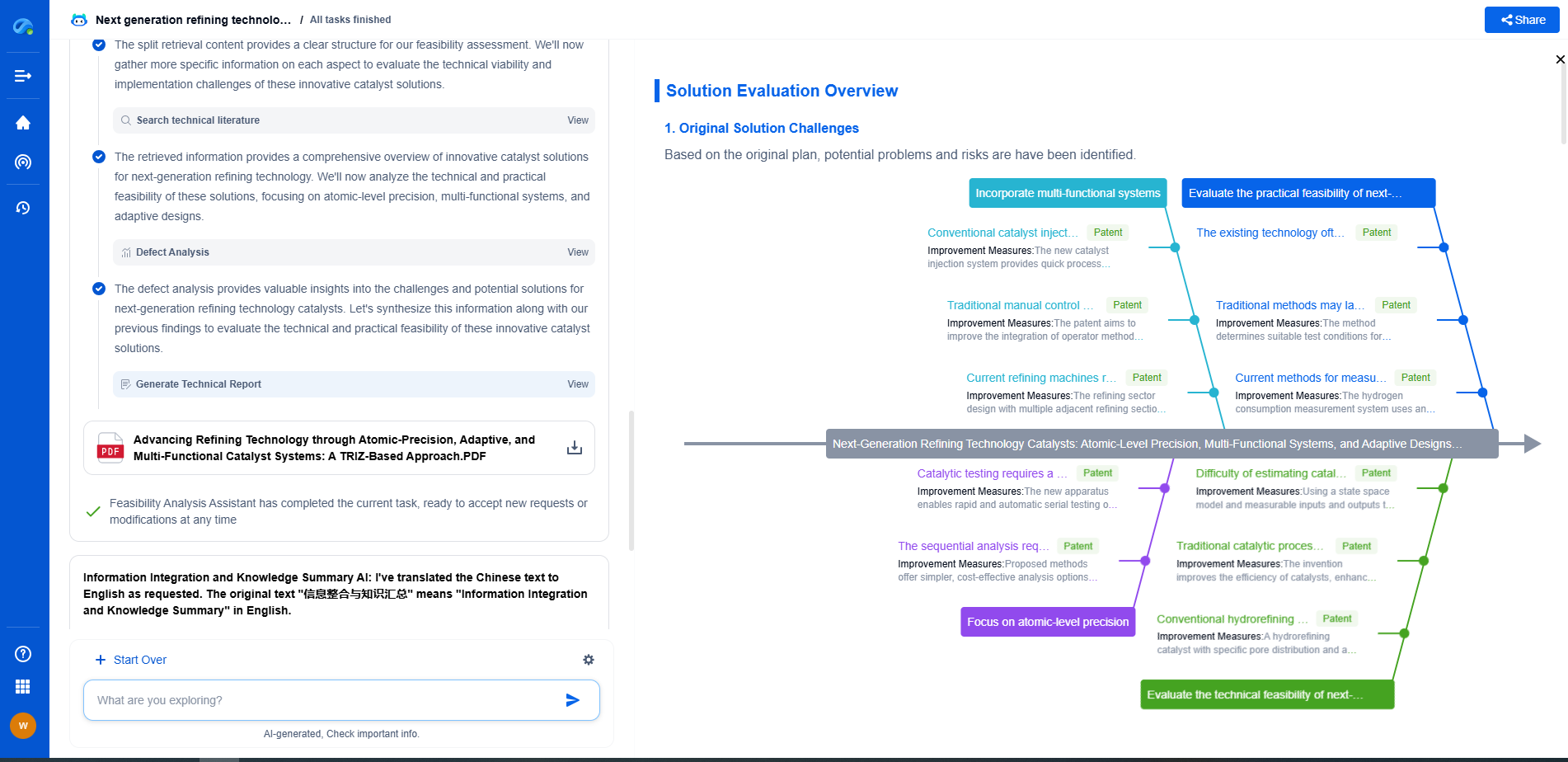
Comparing the Two Processes
While both freeze drying and desublimation involve phase changes, they are fundamentally different processes with unique applications. Freeze drying is broadly applicable in the pharmaceutical industry, particularly for products sensitive to heat and moisture. Its ability to produce stable, long-lasting formulations makes it indispensable for many biopharmaceuticals.
Desublimation, on the other hand, is more niche, often used for capturing specific volatile compounds or in specialized drug delivery systems. Its application is generally more limited, but in scenarios where it is applicable, it can offer precise control over the deposition and purity of the active ingredient.
Applications in Drug Development
The choice between freeze drying and desublimation largely depends on the specific requirements of the pharmaceutical product. For instance, large-scale production of stable, dry formulations of vaccines or biologics typically relies on freeze drying due to its proven track record and scalability.
In contrast, desublimation might be employed in the research and development phase, where precise control over the purity and deposition of volatile compounds is crucial. This could include the formulation of inhalable drugs, where active ingredients are deposited onto carriers in a controlled manner.
Conclusion
Both freeze drying and desublimation have their place in the pharmaceutical industry, each offering distinct advantages for different applications. Freeze drying remains the go-to process for preserving the stability and efficacy of a wide array of pharmaceuticals, while desublimation serves niche applications requiring the precise capture and deposition of volatile substances. As pharmaceutical technology continues to evolve, the integration and optimization of these processes will be pivotal in developing innovative drugs that meet the emerging needs of global healthcare.
From next-generation membrane materials to high-efficiency separation processes for pharmaceuticals, water treatment, food processing, or energy systems, the filtration & separation industry is rapidly evolving with a surge in material innovation, microstructure design, and process optimization.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're designing the next high-throughput filter, optimizing nanostructured surfaces, or exploring new separation media for emerging industries—Patsnap Eureka gives you AI-driven insights in seconds, helping you move from ideation to innovation with confidence.
🚀 Start your free trial today and experience how Eureka transforms filtration innovation—from reactive to predictive.