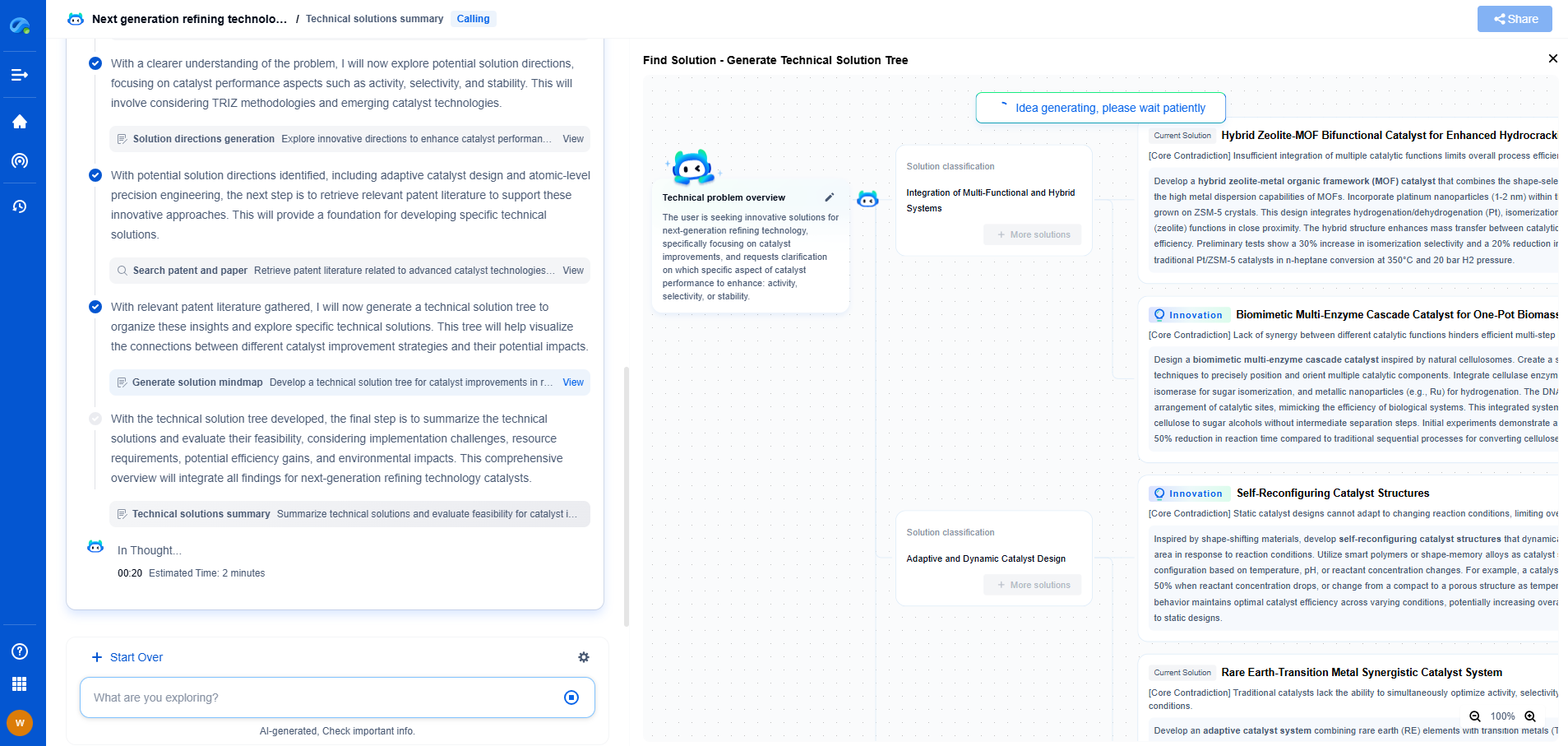
How CO2 Contamination Kills Fuel Cell Performance
JUN 20, 2025 |
Fuel cells are a promising technology for clean energy production, converting chemical energy directly into electrical energy through electrochemical reactions. Unlike conventional combustion engines, fuel cells emit only water and heat as byproducts when using hydrogen as a fuel source. This makes them an attractive option for reducing our carbon footprint and transitioning to sustainable energy solutions. However, their performance can be significantly impaired by certain contaminants, with carbon dioxide (CO2) being one of the critical culprits.
The Basics of CO2 Contamination
Carbon dioxide contamination in fuel cells, particularly in proton exchange membrane fuel cells (PEMFCs), is a well-documented challenge. When hydrogen fuel is produced through reforming processes, such as steam methane reforming, it often contains impurities, including CO2. Although CO2 is not as harmful as carbon monoxide (CO), which can poison the fuel cell's catalyst, it nonetheless impacts the efficiency and durability of fuel cells.
Mechanisms of Degradation
CO2 affects fuel cell performance through several mechanisms. One of the primary concerns is the electrode's potential shift, which can alter the electrochemical environment within the fuel cell. When CO2 is present, it can lead to the formation of carbonic acid (H2CO3) when it reacts with water. This weak acid can lower the pH of the electrolyte, impacting the proton conductivity and thus the overall efficiency of the fuel cell.
Furthermore, CO2 can participate in side reactions that lead to the formation of carbonate species within the cell. These carbonate formations can obstruct the ionic pathways, reducing the membrane's effectiveness and disrupting the delicate balance necessary for optimal fuel cell operation. This not only diminishes performance but also accelerates the degradation of the fuel cell components.
Impact on Catalyst Performance
The catalyst layer in a PEMFC is crucial for facilitating the electrochemical reactions that generate electricity. CO2 contamination can adversely affect catalyst performance. While the primary concern is often CO, CO2 can still contribute to catalyst poisoning by promoting the agglomeration or sintering of catalyst particles. This physical alteration of the catalyst layer can reduce its active surface area, making the fuel cell less efficient in catalyzing hydrogen oxidation and oxygen reduction reactions.
Moreover, the presence of CO2 can alter the adsorption-desorption dynamics of other species on the catalyst surface. The increased competition for active sites can hinder the fuel cell's ability to maintain high reaction rates, resulting in decreased power output and efficiency.
Mitigation Strategies
Given the detrimental effects of CO2 on fuel cell performance, it is essential to implement strategies to mitigate its impact. One of the most effective approaches is improving the hydrogen purification process to minimize CO2 content before it enters the fuel cell. Technologies such as pressure swing adsorption (PSA) and membrane separation can be employed to enhance hydrogen purity.
Additionally, advancements in fuel cell design can play a role in mitigating CO2 effects. Developing more robust catalyst materials that are resistant to CO2 and other contaminants can improve fuel cell resilience. Research into alternative electrolyte materials that are less susceptible to pH changes induced by CO2 is also underway.
Future Directions
As the demand for clean energy technologies grows, overcoming the challenges posed by CO2 contamination in fuel cells will be crucial. Continued research and development efforts are necessary to enhance fuel cell durability and efficiency in the face of CO2 exposure. By improving hydrogen purification techniques and developing more resilient fuel cell components, the potential of fuel cells as a sustainable energy solution can be fully realized.
In conclusion, while CO2 contamination poses significant challenges to fuel cell performance, it is not insurmountable. Through a combination of improved purification processes and innovative fuel cell designs, the negative impacts of CO2 can be mitigated, paving the way for a cleaner and more sustainable energy future.
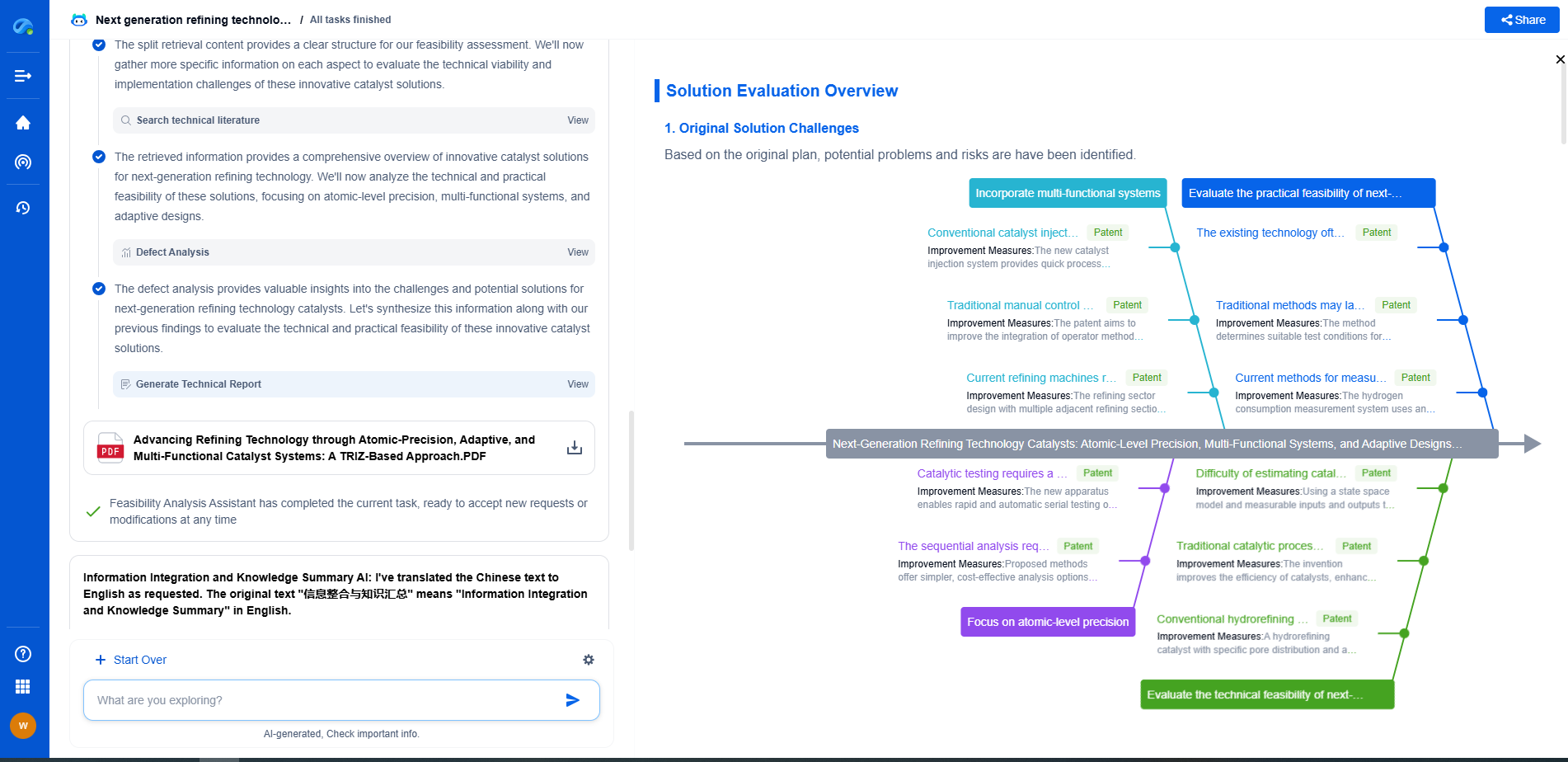
Accelerate Breakthroughs in Fuel Cell and Battery Innovation—with the Power of AI
From solid-state battery breakthroughs to high-efficiency hydrogen fuel cells, keeping pace with fast-evolving chemistries, global patent landscapes, and emerging application pathways is an ever-growing challenge for R&D and IP professionals.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're optimizing cathode formulations, evaluating electrolyte stability, or navigating the crowded patent space around battery pack design, Eureka empowers you to move faster and with greater confidence.
Start your journey with Patsnap Eureka today—streamline your research, enhance decision-making, and power the future of energy with AI-driven clarity.