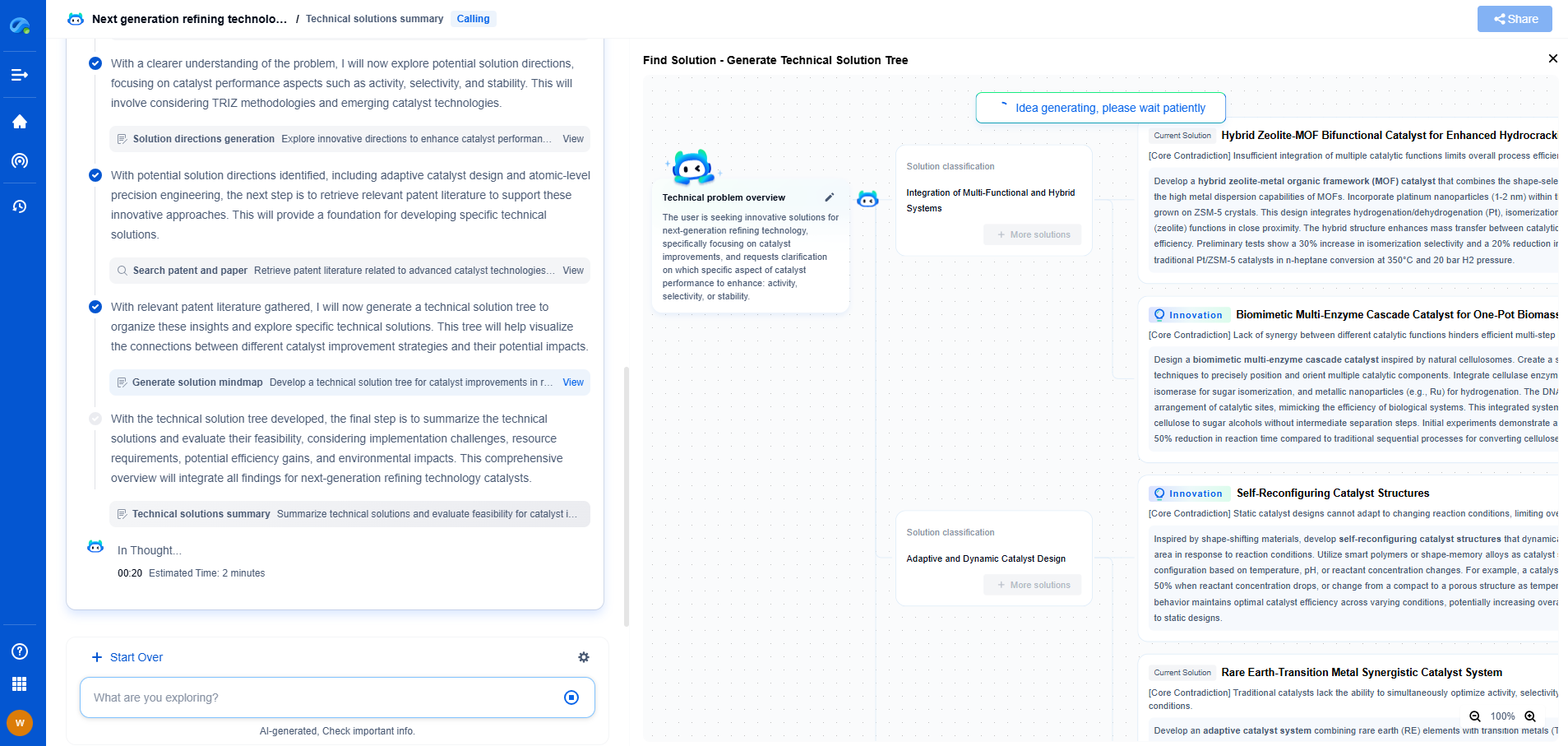
How Do Aqueous vs Non-Aqueous Electrochemical Systems Differ?
JUN 20, 2025 |
Electrochemical systems are integral to a variety of technological applications, from energy storage in batteries to chemical synthesis. These systems can be broadly categorized into aqueous and non-aqueous systems based on the solvent used. Understanding the differences between these two types is crucial for optimizing their performance in different applications.
Aqueous Electrochemical Systems
Aqueous electrochemical systems use water as the solvent. These systems are widely used due to water’s high polarity, which helps dissolve a wide range of electrolytes, and its ability to facilitate efficient ionic transport. The presence of water also allows for a stable and controlled electrochemical environment, which is ideal for many reactions.
Advantages of Aqueous Systems
1. Safety: Water-based systems are generally safer to handle due to water's non-flammable nature.
2. Cost-Effectiveness: Water is abundant and inexpensive, making aqueous systems economically attractive.
3. High Ionic Conductivity: Water's high dielectric constant allows for the dissolution of salts, leading to high ionic conductivity.
Limitations of Aqueous Systems
1. Voltage Window: One of the significant limitations is the narrow electrochemical window of water, which is approximately 1.23 volts. This limits the energy density of aqueous electrochemical systems.
2. Corrosion: The presence of water can lead to corrosion of electrodes, which can affect the longevity and efficiency of the system.
Non-Aqueous Electrochemical Systems
Non-aqueous electrochemical systems utilize organic solvents instead of water. These solvents are typically used when the reactions or applications require a broader electrochemical window or when specific solubility characteristics are needed.
Advantages of Non-Aqueous Systems
1. Wide Voltage Window: Non-aqueous systems often have a broader electrochemical window, allowing for greater energy density, which is particularly beneficial in applications like lithium-ion batteries.
2. Reduced Corrosion: The absence of water reduces the likelihood of corrosion, potentially increasing the lifespan of the electrodes and the overall system.
Limitations of Non-Aqueous Systems
1. Safety Concerns: Many organic solvents are flammable and toxic, posing significant safety risks.
2. Cost: Organic solvents can be more expensive and less environmentally friendly than water.
3. Lower Ionic Conductivity: Non-aqueous systems generally have lower ionic conductivity compared to their aqueous counterparts, which can affect the efficiency of ionic transport.
Comparative Analysis
When comparing these two systems, the choice between aqueous and non-aqueous often depends on the specific application requirements. For instance, in applications where high voltage and energy density are critical, such as electric vehicles, non-aqueous systems are typically preferred. Conversely, for safer, more environmentally benign applications, aqueous systems might be more appropriate.
Applications and Future Trends
Both aqueous and non-aqueous systems play vital roles in the advancement of technologies such as batteries, fuel cells, and electrochemical sensors. Future trends indicate a growing interest in hybrid systems that combine the benefits of both aqueous and non-aqueous systems. Moreover, ongoing research aims to develop new materials and solvents that can enhance the performance and safety of these systems.
Conclusion
Understanding the distinct characteristics of aqueous versus non-aqueous electrochemical systems allows for better design and optimization of these technologies for specific applications. By leveraging the strengths of each system and mitigating their weaknesses, researchers and engineers can continue to develop advanced electrochemical systems that meet the growing demands of modern technology.
Accelerate Breakthroughs in Fuel Cell and Battery Innovation—with the Power of AI
From solid-state battery breakthroughs to high-efficiency hydrogen fuel cells, keeping pace with fast-evolving chemistries, global patent landscapes, and emerging application pathways is an ever-growing challenge for R&D and IP professionals.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're optimizing cathode formulations, evaluating electrolyte stability, or navigating the crowded patent space around battery pack design, Eureka empowers you to move faster and with greater confidence.
Start your journey with Patsnap Eureka today—streamline your research, enhance decision-making, and power the future of energy with AI-driven clarity.