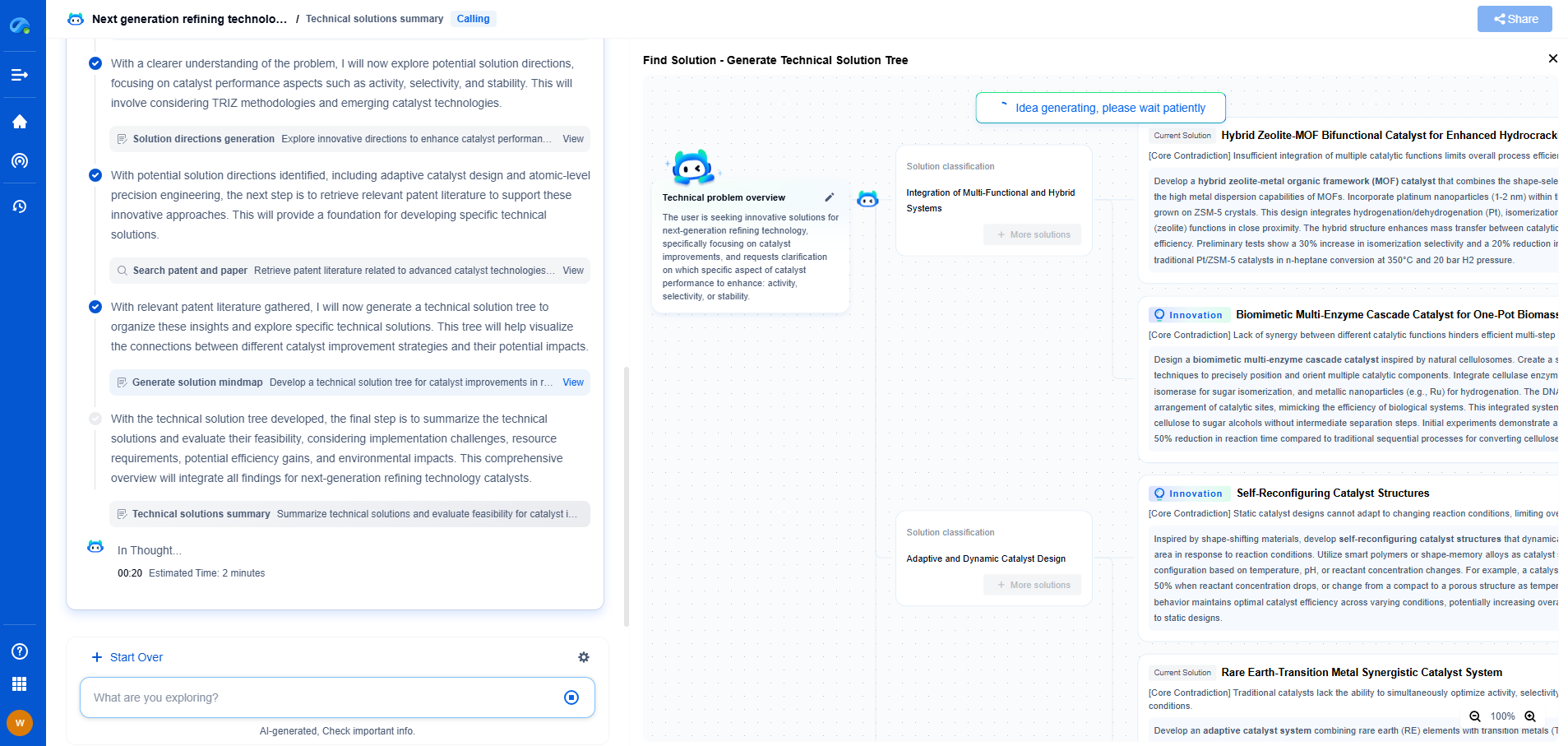
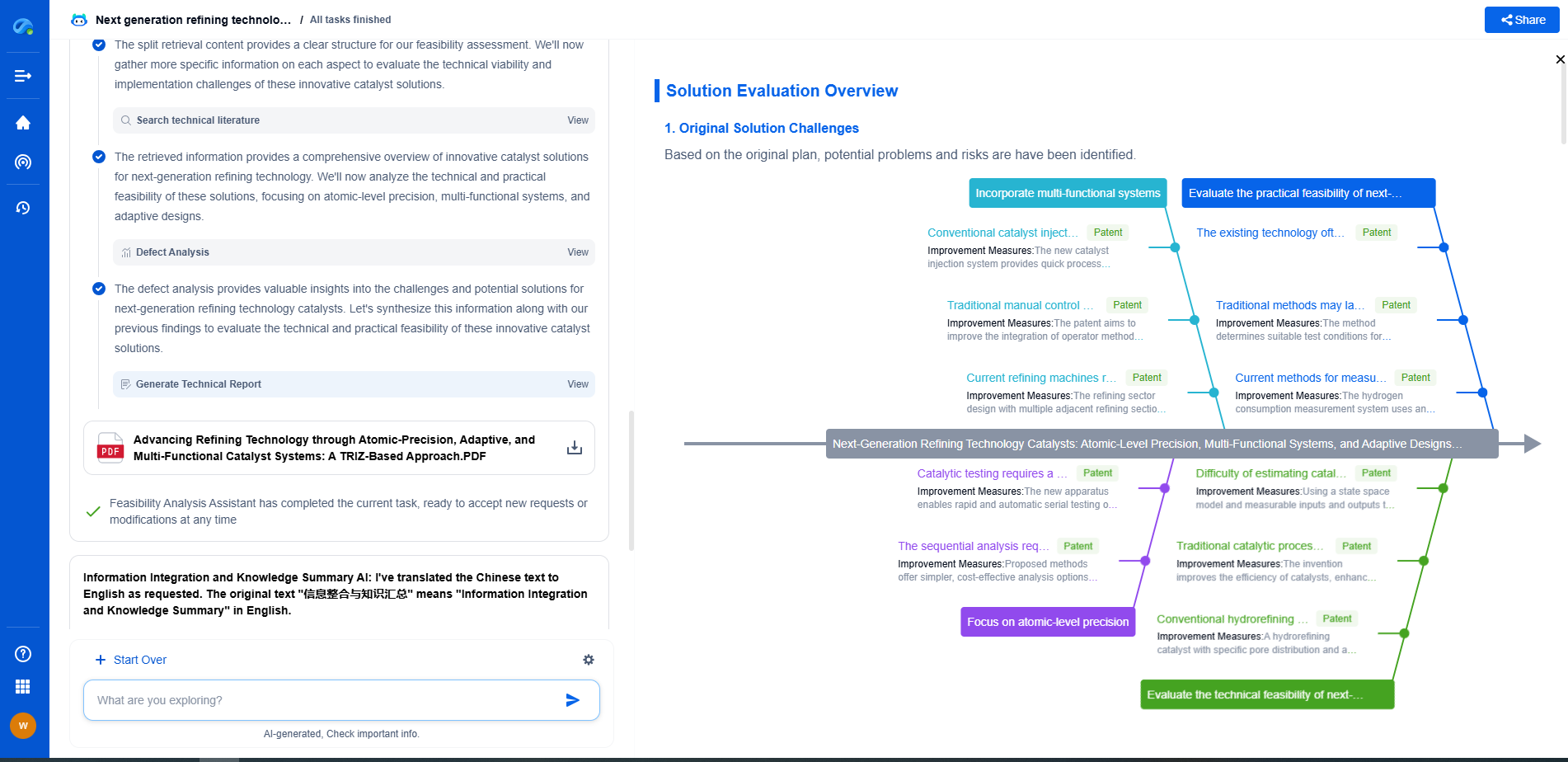
Pattern Collapse Mitigation: Supercritical CO2 Drying Techniques
JUL 28, 2025 |
Pattern collapse is a critical challenge faced in the microfabrication industry, particularly in the manufacturing of semiconductors and microelectromechanical systems (MEMS). It occurs when delicate features on a wafer, often on the nanoscale, collapse due to capillary forces during the drying process. As device geometries continue to shrink, the propensity for pattern collapse increases, necessitating advanced techniques to mitigate this problem. One such promising method is supercritical CO2 drying, which has emerged as a preferred technique due to its unique properties and effectiveness.
The Problem with Conventional Drying Methods
Traditional drying techniques, such as spin drying and oven drying, rely on the evaporation of liquid solvents. However, these methods often exacerbate pattern collapse due to the surface tension and capillary forces at play during the phase transition from liquid to vapor. As these forces exceed the mechanical strength of the nanoscale features, deformation or collapse becomes inevitable. The need for a drying method that minimizes or eliminates these forces is thus critical for preserving the integrity of microstructures.
Understanding Supercritical CO2
Supercritical CO2 (sCO2) refers to carbon dioxide that is held at or above its critical temperature and pressure, reaching a state where it exhibits properties of both a liquid and a gas. This unique phase allows it to penetrate materials like a gas while dissolving substances like a liquid. The critical point for CO2 is relatively accessible, at around 31.1 degrees Celsius and 7.38 MPa, making it an ideal candidate for industrial processes.
Advantages of Supercritical CO2 Drying
The primary advantage of using sCO2 in the drying process is its negligible surface tension. Because sCO2 can transition from its supercritical phase to a gas without crossing a liquid-vapor boundary, it eliminates the capillary forces that contribute to pattern collapse. This gentle drying process ensures the structural integrity of the finest features on a wafer.
Additionally, sCO2 is environmentally friendly and safe to handle. It is non-toxic, non-flammable, and readily available. Its use minimizes the reliance on harmful solvents and reduces the environmental footprint of the microfabrication process.
Implementing Supercritical CO2 Drying Techniques
Integrating sCO2 drying into microfabrication involves several key steps. Initially, the wafer is immersed in a compatible solvent to prevent premature drying. The solvent is then exchanged with liquid CO2, typically in a high-pressure chamber. Once the solvent has been replaced, the system is heated and pressurized to reach the supercritical phase. Careful control of temperature and pressure is crucial to ensure the CO2 remains in its supercritical state. After sufficient time, the pressure is gradually reduced, allowing the CO2 to return to its gaseous state and leaving the wafer dry without inducing capillary stress.
Challenges and Considerations
While sCO2 drying offers numerous benefits, it is not without challenges. The initial setup costs for equipment capable of maintaining the necessary temperatures and pressures can be significant. Moreover, the process itself requires precise control and understanding of supercritical fluids, necessitating specialized training and expertise.
Additionally, the compatibility of materials with sCO2 must be considered. Some materials or coatings may react adversely with CO2, necessitating additional research or process modifications to ensure compatibility.
Future Directions and Innovations
As the demand for smaller, more complex devices grows, the role of sCO2 drying in mitigating pattern collapse is likely to expand. Continued research into the behavior of sCO2 with various materials will enhance the versatility and application of this technique. Innovations in equipment design may also reduce costs and improve the accessibility of supercritical drying methods.
Moreover, hybrid approaches that combine sCO2 drying with other advanced techniques could further optimize the fabrication process, offering even greater precision and reliability.
Conclusion
Supercritical CO2 drying presents a viable and increasingly essential solution to the challenge of pattern collapse in microfabrication. By eliminating the capillary forces that plague traditional drying methods, sCO2 offers a way to preserve the intricate features necessary for the advancement of semiconductor and MEMS technologies. Although challenges remain, the benefits of sCO2 drying make it a cornerstone in the ongoing evolution of microfabrication processes. As the industry continues to innovate and adapt, supercritical drying techniques will undoubtedly play a pivotal role in shaping the future of technology.
As photolithography continues to push the boundaries of nanoscale patterning, from EUV and DUV advancements to multi-patterning and maskless lithography, innovation cycles are accelerating—and the IP landscape is becoming more complex than ever.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're optimizing lithography depth of focus or exploring new materials for sub-3nm nodes, Patsnap Eureka empowers you to make smarter decisions, faster—combining AI efficiency with domain-specific insight.
💡 Start your free trial today and see how Eureka transforms how you discover, evaluate, and act on innovation in photolithography—from idea to impact.