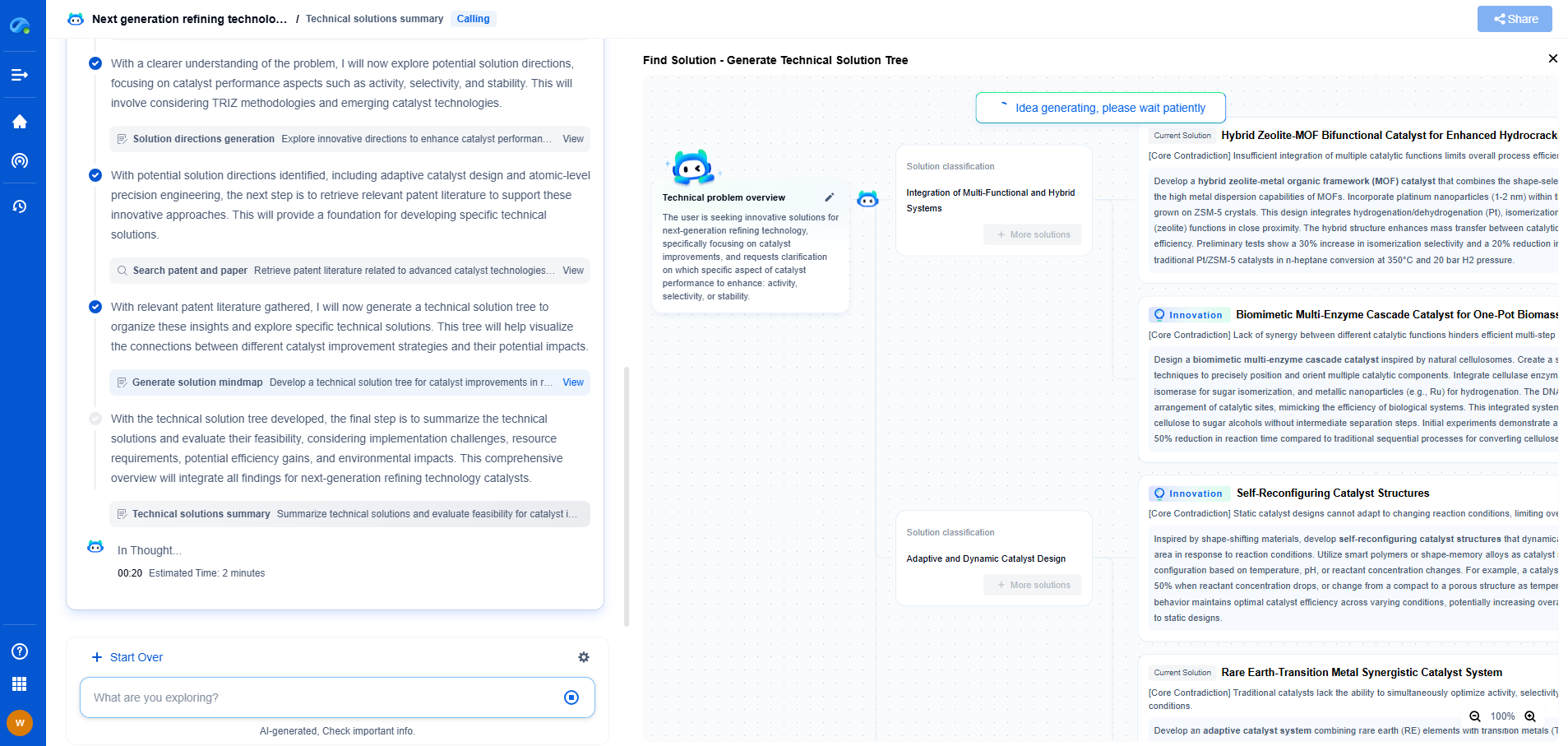
What is a catalyst regenerator and how does it function?
JUN 19, 2025 |
In the field of chemical engineering and industrial processes, the term "catalyst regenerator" holds significant importance. Catalysts are substances that increase the rate of a chemical reaction without themselves being consumed. Over time, these catalysts lose their activity due to fouling or poisoning, hence the need for regeneration. But what exactly is a catalyst regenerator, and how does it function within industrial settings?
What is Catalyst Regeneration?
Catalyst regeneration is a process that restores the activity of a spent catalyst, effectively extending its service life and maintaining the efficiency of the chemical reactions in which it participates. This process is crucial in industries such as petrochemicals, where catalysts are frequently used in cracking processes to break down large hydrocarbon molecules into smaller, more useful ones.
The regeneration process typically involves the removal of carbon deposits and other contaminants that accumulate on the catalyst surface. By cleaning and rejuvenating these catalysts, industries can significantly reduce costs associated with purchasing new catalysts and disposing of spent ones.
How Does a Catalyst Regenerator Work?
A catalyst regenerator is a device or system designed to perform the regeneration process. Its functioning can vary depending on the type of catalyst and the impurities present. However, the fundamental principles remain largely the same and can be divided into several key steps:
1. **Decoking**: In many industrial processes, catalysts become coated with a carbonaceous material known as "coke" due to the thermal decomposition of hydrocarbons. The first step in regeneration involves burning off this coke in the presence of oxygen. This is done at elevated temperatures and controlled conditions to avoid damaging the catalyst structure.
2. **Oxidation**: Once coke deposits are removed, the catalyst surface is typically oxidized to restore metal oxides that are essential for its activity. This step is crucial for catalysts used in processes like fluid catalytic cracking (FCC) in refineries.
3. **Reduction and Re-dispersion**: Some catalysts require a reduction step to restore their active metal sites. This may involve treating the catalyst with hydrogen or other reducing agents. Additionally, re-dispersion of active metals on the support may be necessary to ensure uniform activity across the catalyst.
4. **Sulfation or Sulfur Stripping**: In cases where sulfur compounds have poisoned the catalyst, a stripping step is employed to remove these impurities. This may involve treatments with steam or other chemical agents.
5. **Thermal Treatments**: Temperature control is vital throughout the regeneration process to prevent sintering, which can irreversibly damage the catalyst and reduce its surface area.
Benefits of Using Catalyst Regenerators
The use of catalyst regenerators offers several benefits to industrial operations. Firstly, they enhance the longevity of catalysts, allowing for repeated use and reducing the need for frequent replacements. This is not only cost-effective but also environmentally friendly, as it minimizes waste generation. Moreover, maintaining catalyst activity through regular regeneration ensures consistent process efficiency and product quality.
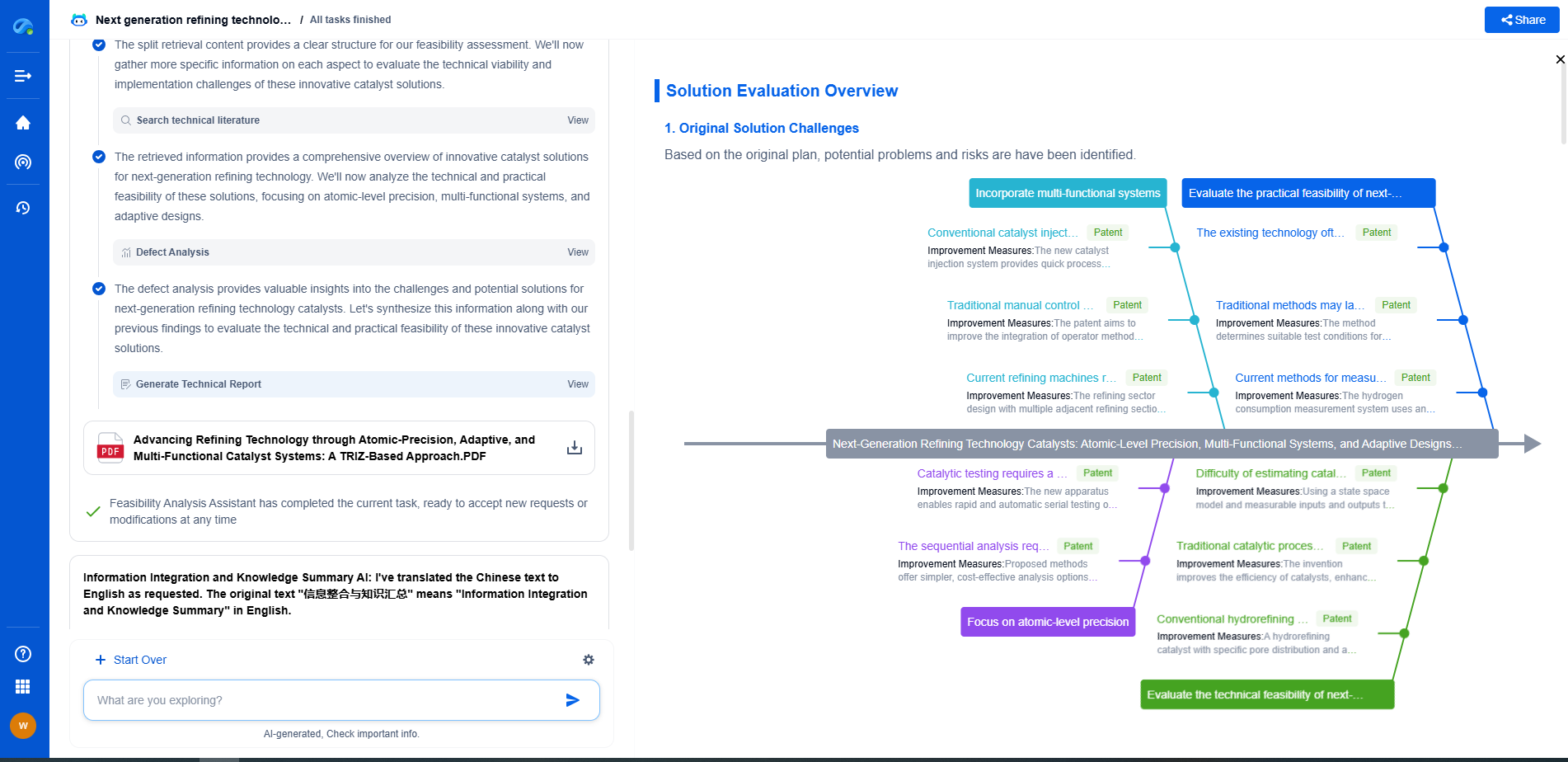
Challenges in Catalyst Regeneration
Despite the advantages, catalyst regeneration poses certain challenges. The process requires careful control of operating conditions to avoid damaging the catalyst. The choice of regeneration method must also be tailored to the specific type of catalyst and the nature of its deactivation. Additionally, the handling of potentially hazardous materials during regeneration necessitates rigorous safety protocols.
Conclusion
Catalyst regenerators play a pivotal role in modern industrial processes by restoring the activity and efficiency of catalysts. Through a series of carefully controlled steps such as decoking, oxidation, and reduction, these systems ensure that catalysts can be used repeatedly, offering economic and environmental advantages. As industries continue to rely heavily on catalytic processes, the importance of effective catalyst regeneration cannot be overstated.
Discover Patsnap Eureka: AI Agents Built for Scientific Innovation
Whether you're designing the next generation of refining technologies or analyzing catalysts and process flows, keeping up with rapidly evolving research and IP data in petroleum processing is no easy task.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Ready to accelerate your innovation process and make smarter, faster decisions? Discover Patsnap Eureka today and unlock the full power of confident, AI-driven innovation.