What Is Electrolyte Decomposition and Why Does It Matter?
JUN 20, 2025 |
Electrolyte decomposition is a critical topic in fields ranging from chemistry and materials science to energy storage systems like batteries. Simply put, electrolyte decomposition refers to the breakdown of an electrolyte into different compounds due to chemical reactions, often induced by external factors such as heat, voltage, or chemical catalysts. Understanding this process is crucial for both scientific advancement and practical applications, particularly in battery technology.
The Chemistry Behind Electrolyte Decomposition
Electrolytes are substances that contain free ions and conduct electricity. They are vital components in a variety of applications, including electrochemical cells, batteries, and even biological systems. Decomposition occurs when the electrolyte undergoes a chemical transformation, often resulting in unwanted byproducts. This breakdown can be caused by several factors, including:
1. **Thermal Instability**: Elevated temperatures can cause electrolytes to decompose, leading to the release of gases and heat, which may further accelerate the process.
2. **Electrochemical Factors**: Over-voltage or incorrect charging conditions can lead to the breakdown of electrolyte components, particularly in batteries where precise voltage conditions are necessary for safe operation.
3. **Chemical Reactions**: Interaction with other materials or impurities can initiate decomposition, leading to a chain reaction that disrupts the electrolyte's function.
Why Electrolyte Decomposition Matters
Electrolyte decomposition is not merely a scientific curiosity; it has significant practical implications, especially in the development and usage of batteries. The stability of the electrolyte is paramount to the efficiency, safety, and longevity of energy storage systems. When decomposition occurs, it can lead to:
1. **Reduced Efficiency**: Decomposition can diminish the conductivity of the electrolyte, reducing the battery’s ability to store and deliver energy effectively.
2. **Safety Hazards**: The release of gases or heat due to decomposition can pose severe safety risks, including fires or explosions, especially in high-energy-density batteries like lithium-ion cells.
3. **Shortened Lifespan**: Repeated decomposition reactions can degrade battery components, significantly shortening the lifespan of the device.
Strategies to Mitigate Electrolyte Decomposition
In response to these challenges, researchers and engineers are exploring various strategies to reduce the risks associated with electrolyte decomposition. These include:
1. **Material Innovation**: Developing new electrolyte materials that are inherently more stable under a range of conditions can help in mitigating decomposition.
2. **Protective Coatings**: Applying protective layers to battery components can minimize the interaction between the electrolyte and other materials, reducing the likelihood of decomposition reactions.
3. **Optimized Operating Conditions**: Implementing precise control over the thermal and electrochemical environment can significantly curb decomposition risks.
Implications for Future Technologies
The ongoing research into electrolyte decomposition holds promise for a wide range of future technologies. In particular, advances in this area could lead to safer, more efficient, and longer-lasting batteries, which are crucial for the advancement of electric vehicles, renewable energy storage systems, and portable electronic devices. As our reliance on these technologies grows, the importance of understanding and controlling electrolyte decomposition cannot be overstated.
In conclusion, electrolyte decomposition is a complex but highly significant phenomenon with broad implications. By continuing to explore this area, scientists and engineers hope to unlock new potential in energy storage and other applications, paving the way for technological advancements that are both innovative and sustainable.
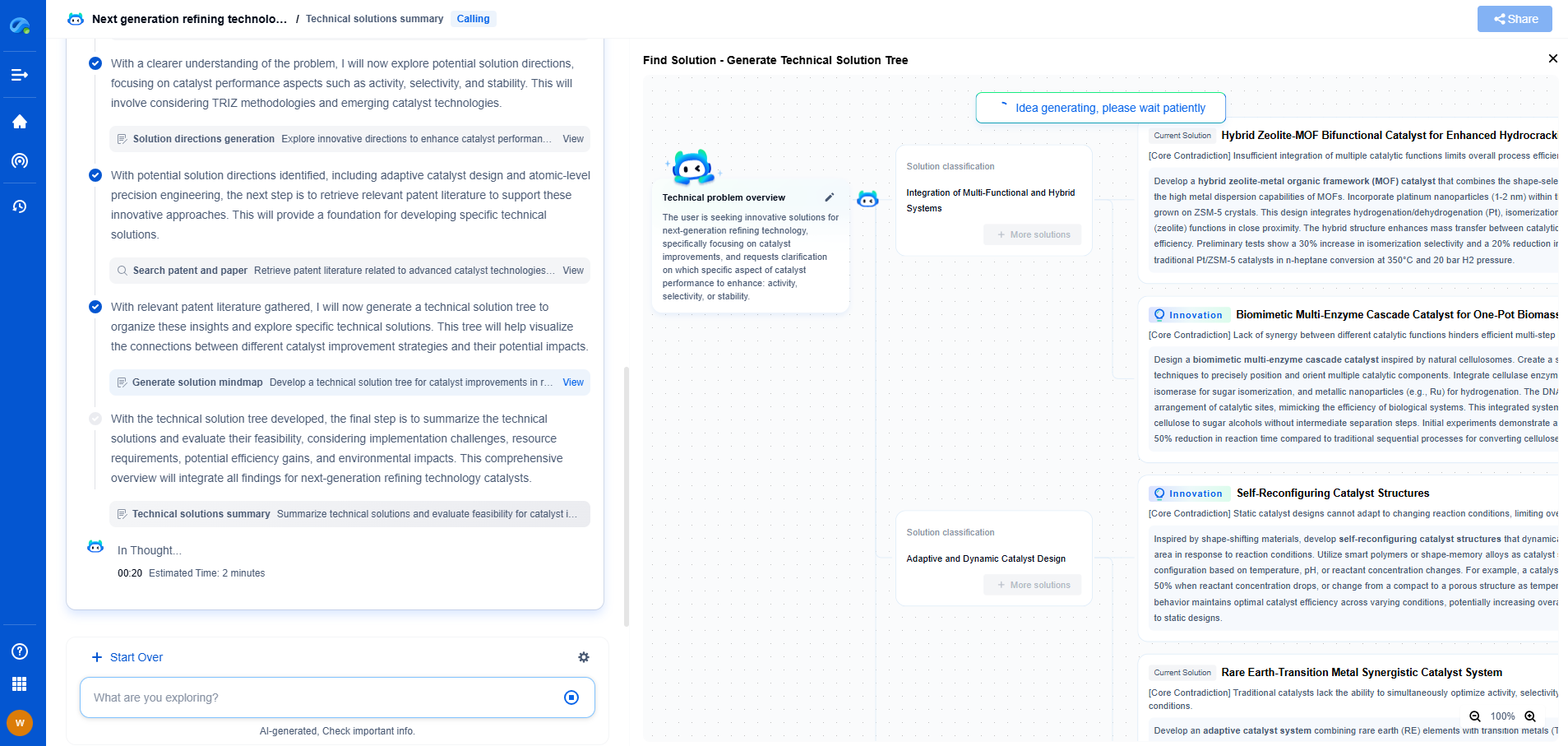
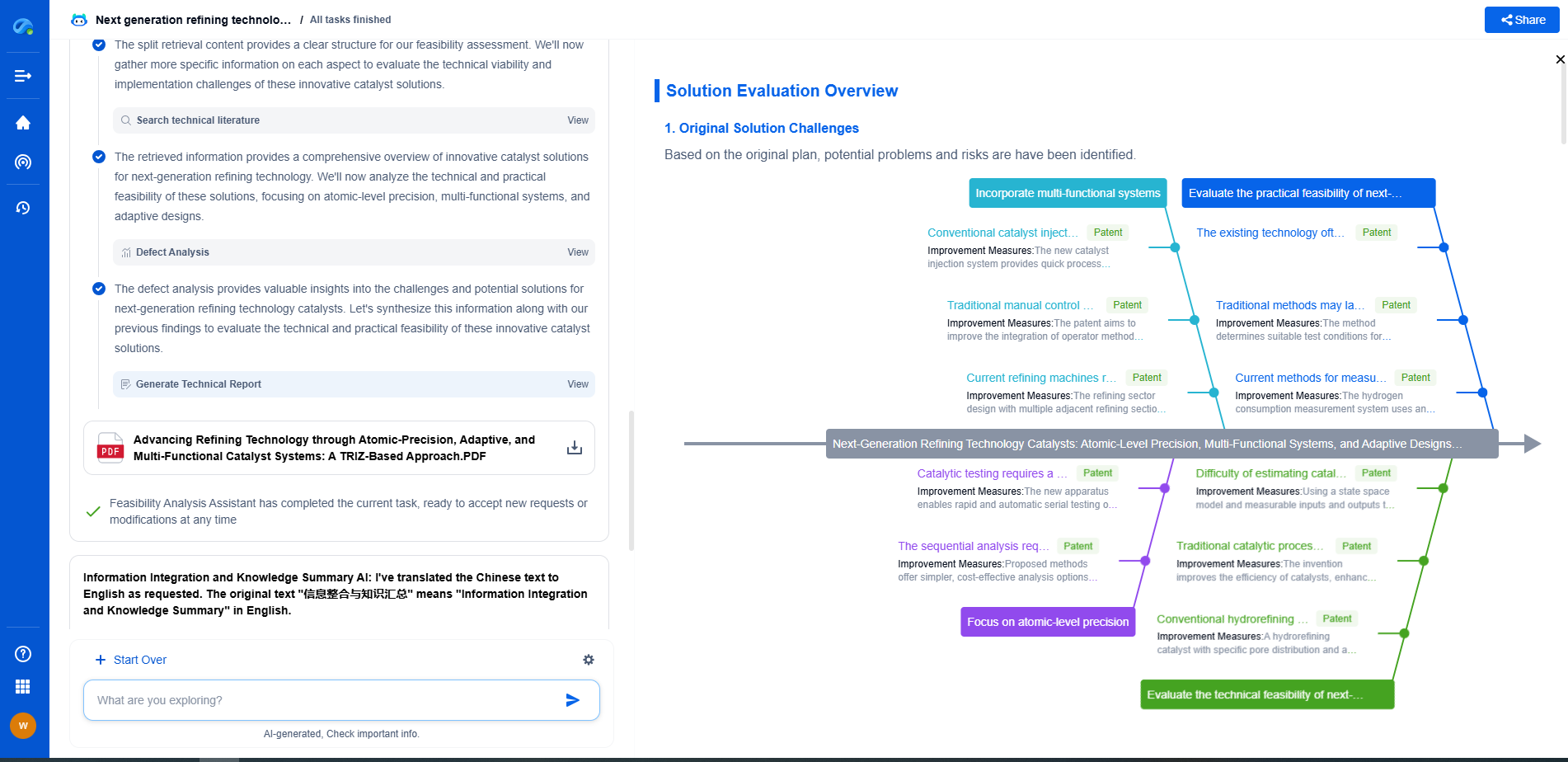
Accelerate Breakthroughs in Fuel Cell and Battery Innovation—with the Power of AI
From solid-state battery breakthroughs to high-efficiency hydrogen fuel cells, keeping pace with fast-evolving chemistries, global patent landscapes, and emerging application pathways is an ever-growing challenge for R&D and IP professionals.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Whether you're optimizing cathode formulations, evaluating electrolyte stability, or navigating the crowded patent space around battery pack design, Eureka empowers you to move faster and with greater confidence.
Start your journey with Patsnap Eureka today—streamline your research, enhance decision-making, and power the future of energy with AI-driven clarity.