What is the difference between gasoline and naphtha?
JUN 19, 2025 |
Gasoline and naphtha are both hydrocarbons derived from crude oil, but they serve different purposes and are distinct in their composition and applications. Understanding the differences between these two substances is crucial for those in the oil and gas industry, as well as for individuals interested in the energy sector.
Composition and Refining Process
Gasoline and naphtha are products of the crude oil refining process, but they are obtained at different stages and through different methods. Gasoline is a complex mixture of various hydrocarbons, primarily alkanes, cycloalkanes, and a few aromatic hydrocarbons. It is typically obtained from the fractional distillation of crude oil, followed by further processing such as reforming and blending to improve its quality and performance.
On the other hand, naphtha is a term used to describe a range of volatile, flammable liquid hydrocarbon mixtures. It is often considered a precursor to gasoline and is obtained earlier in the refining process. There are different types of naphtha, such as light and heavy naphtha, each with varying properties and uses. Light naphtha is rich in paraffinic hydrocarbons, while heavy naphtha contains more aromatic hydrocarbons.
Applications and Uses
Gasoline is primarily used as a fuel for internal combustion engines in vehicles. Its properties make it ideal for use in cars, motorcycles, and small engines due to its high energy content and volatility. The formulation of gasoline includes additives that enhance engine performance, reduce emissions, and prevent knocking.
In contrast, naphtha has a broader range of applications. It is used as a feedstock for the petrochemical industry, where it is further processed to produce ethylene, propylene, and other valuable chemicals. Naphtha is also used as a solvent in various industrial processes, including the production of paints and varnishes. Additionally, it can be utilized as a blending component in gasoline production, though it requires further refining to meet regulatory standards for road fuel.
Physical and Chemical Properties
One of the key differences between gasoline and naphtha is their boiling point range. Gasoline typically has a boiling point range of about 30°C to 200°C, which contributes to its suitability as a fuel for internal combustion engines. Its volatility is carefully controlled to ensure optimal ignition and combustion in engines.
Naphtha, by comparison, has a broader boiling point range, usually between 30°C and 210°C, depending on its type. Light naphtha boils at lower temperatures, while heavy naphtha has higher boiling points. These properties make naphtha an excellent feedstock for producing various chemicals through cracking and reforming processes.
Environmental and Safety Considerations
Both gasoline and naphtha pose environmental and safety concerns, primarily due to their flammability and potential for pollution. Gasoline engines emit greenhouse gases and other pollutants, contributing to air quality issues and climate change. Efforts to improve gasoline formulations and develop alternative fuels are ongoing to mitigate these effects.
Naphtha, while less commonly used as a direct fuel, still presents risks due to its volatility and flammability. In industrial settings, handling and storage of naphtha require stringent safety measures to prevent accidents and spills. Additionally, the chemical processes that utilize naphtha must adhere to environmental regulations to limit emissions and waste.
Conclusion
In summary, while gasoline and naphtha share some similarities as petroleum-derived products, they differ significantly in their composition, refining process, applications, and properties. Gasoline is mainly used as a vehicle fuel, whereas naphtha serves as an essential feedstock for the petrochemical industry and a solvent in various applications. Understanding these differences is key to appreciating their roles in our daily lives and the broader energy landscape.
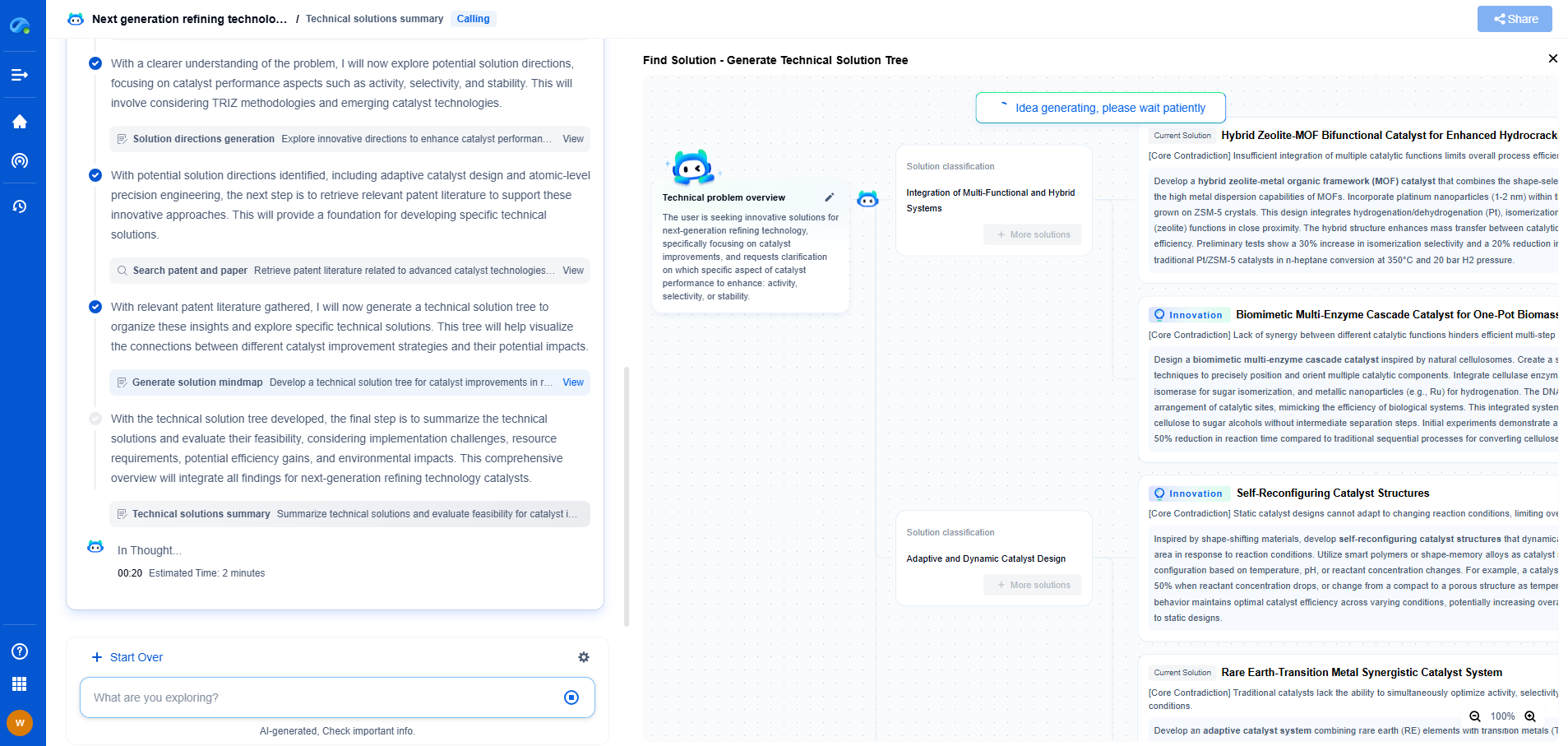
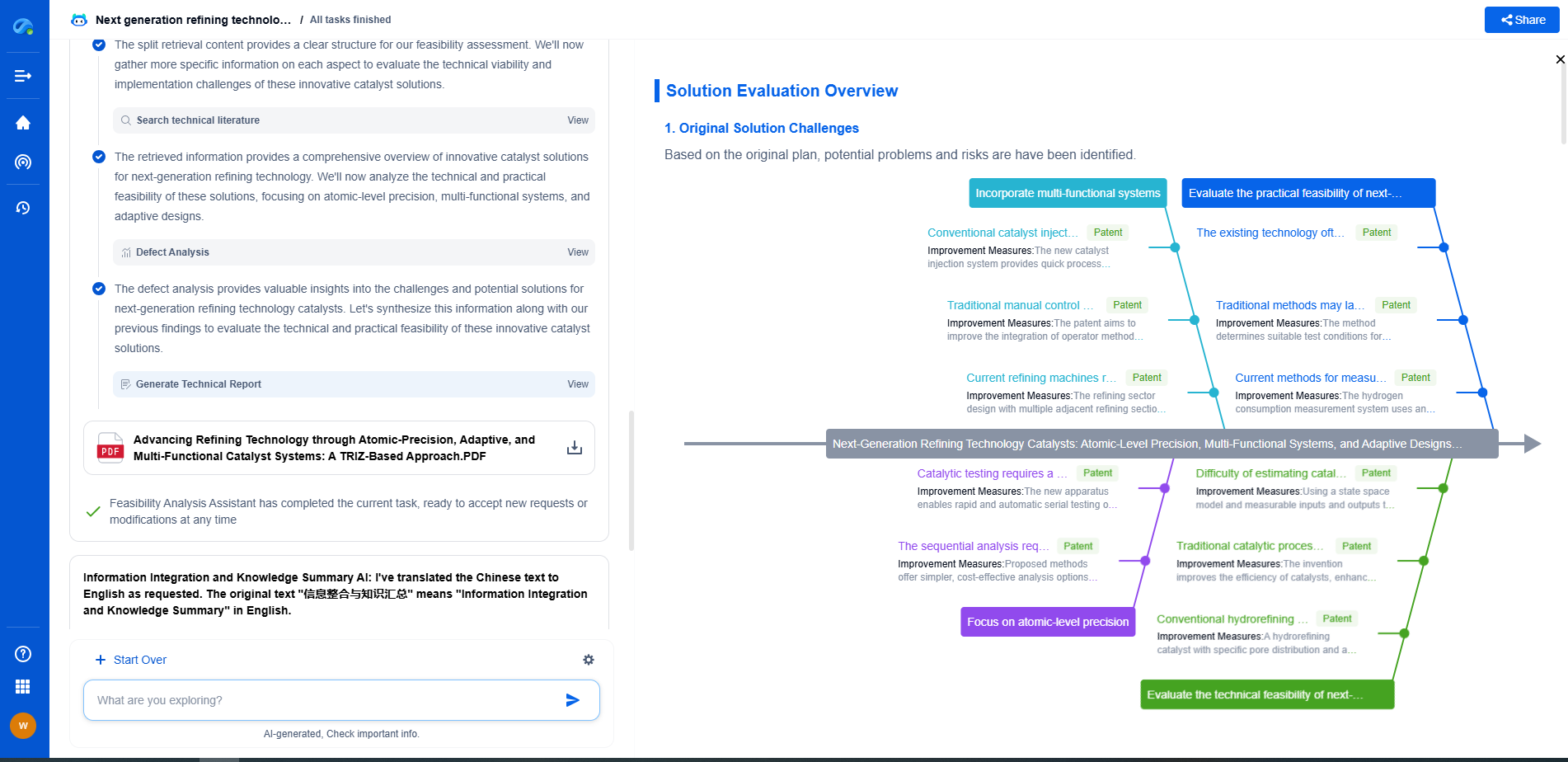
Discover Patsnap Eureka: AI Agents Built for Scientific Innovation
Whether you're designing the next generation of refining technologies or analyzing catalysts and process flows, keeping up with rapidly evolving research and IP data in petroleum processing is no easy task.
Patsnap Eureka, our intelligent AI assistant built for R&D professionals in high-tech sectors, empowers you with real-time expert-level analysis, technology roadmap exploration, and strategic mapping of core patents—all within a seamless, user-friendly interface.
Ready to accelerate your innovation process and make smarter, faster decisions? Discover Patsnap Eureka today and unlock the full power of confident, AI-driven innovation.