
The Gabriel Phthalimide Synthesis is a classical organic reaction used to prepare primary amines from alkyl halides. It is widely appreciated for its selectivity, allowing for the clean formation of primary (1°) amines without producing secondary or tertiary amines as byproducts. This article explains the step-by-step reaction mechanism, provides a detailed overview of the reagents and conditions involved, and highlights the key applications and limitations of this synthesis method.
Get Started with PatSnap Eureka AI Agent
What Is the Gabriel Phthalimide Synthesis?
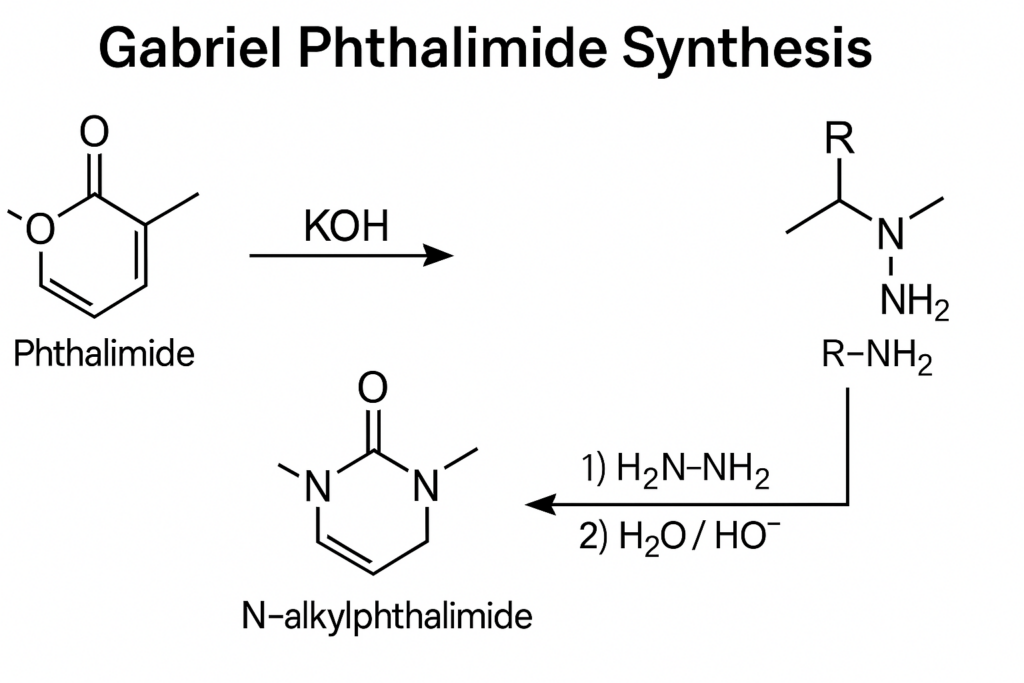
Gabriel phthalimide synthesis is a chemical process used to convert primary alkyl halides into primary amines. This method primarily involves the use of potassium phthalimide as a starting material, which undergoes a series of reactions to introduce the amine group into the alkyl chain.
What is the Gabriel Phthalimide Synthesis? Eureka Technical Q&A explains this method for preparing primary amines using potassium phthalimide, helping you understand its mechanism, advantages, and role in organic chemistry.
Hydrolysis to Primary Amine: The final step is the hydrolysis of the N-alkyl phthalimide to yield the primary amine. This is typically achieved by treating the compound with acid, which hydrolyzes the phthalimide ring, releasing the primary amine.
Formation of Potassium Phthalimide: The process begins with the reaction of phthalimide with ethanolic potassium hydroxide (KOH). This reaction forms the potassium salt of phthalimide by removing the NH proton, resulting in the imide ion.
Alkylation Reaction: The next step involves heating the potassium phthalimide with an alkyl halide. This alkylation reaction introduces the alkyl group into the phthalimide structure, forming N-alkyl phthalimide.
Key Reactants and Reagents
- Potassium Phthalimide: This is a crucial starting material in the Gabriel synthesis. It serves as the nucleophile that undergoes alkylation with alkyl halides .
- Alkyl Halides: These are used as the alkylating agents in the reaction. Examples include ethyl halides, such as ethyl bromide or ethyl chloride, which react with potassium phthalimide to form the N-alkylated product .
- Solvents: Dimethylformamide (DMF) is commonly used as a solvent in the Gabriel synthesis to facilitate the reaction and improve the yield .
- Catalysts: In some variations of the Gabriel synthesis, catalysts such as tetrabutylammonium bromide (TBAB) may be employed to enhance the reaction rate and selectivity .
- Base: Potassium hydroxide is often used to convert phthalimide into its potassium salt form, which is more reactive towards alkylation .
- Hydrazine or Ethylenediamine: These reagents can be used to remove the phthalimide group from the N-alkylated product, yielding the primary amine .
- Acids: Acidic conditions may be employed in certain steps of the synthesis, such as during the cleavage of the phthalimide group .
General Reaction Scheme
The Gabriel phthalimide synthesis is a well-established method for transforming primary alkyl halides into primary amines, utilizing potassium phthalimide as a key reactant. Here is a general reaction scheme for the Gabriel phthalimide synthesis:
- Starting Materials:
- Primary alkyl halide (e.g., 1-bromopropane)
- Potassium phthalimide
- Reaction Mechanism:
- The primary alkyl halide reacts with potassium phthalimide in a polar aprotic solvent like DMF (dimethylformamide).
- The reaction proceeds through the nucleophilic substitution of the alkyl halide by the phthalimide anion, resulting in the formation of an N-alkylphthalimide intermediate.
- Optimized Conditions:
- The reaction typically occurs at elevated temperatures, around 90°C, to enhance the reactivity and yield.
- The molar ratio of alkyl halide to potassium phthalimide is often optimized to achieve higher yields, commonly around 2:1.
- Product Formation:
- The N-alkylphthalimide product can then be hydrolyzed under acidic conditions to release the primary amine and phthalic anhydride.
- Yield and Purity:
- The overall yield of the Gabriel synthesis can vary but is often reported in the range of 70-90%, depending on the specific reactants and conditions used.
This synthesis method is advantageous for its straightforward manipulation and relatively high yield, making it a popular choice in both laboratory and industrial settings for the preparation of primary amines, which are valuable intermediates in pharmaceutical and chemical research.
Applications of Gabriel Synthesis
- Pharmaceuticals: Gabriel synthesis is crucial in drug development due to its efficiency in producing primary amines, which are fundamental building blocks for many pharmaceuticals. The method allows for the creation of diverse amine structures that are essential in developing new drugs and therapeutic agents.
- Polymer Industry: The primary amines produced via Gabriel synthesis serve as essential monomers in the synthesis of polyamides and polyurethanes. These polymers have extensive applications, ranging from textiles to automotive parts, highlighting the synthesis’s importance in material science.
- Aerospace and Automotive Fields: In these industries, primary diamines are significant for manufacturing high-performance materials. The Gabriel synthesis contributes to the production of lightweight and durable components, which are critical for aerospace applications.
- Environmental Monitoring: The Gabriel synthesis is also applied in environmental monitoring, where primary amines can be used in the development of sensors and detection devices for monitoring environmental pollutants.
- Heterocyclic Chemistry: The Gabriel synthesis is involved in the preparation of heterocyclic compounds, such as oxazoles and isoxazoles, through methods like the Robinson-Gabriel synthesis. These heterocycles are important in medicinal chemistry and agrochemicals.
Limitations of Gabriel Phthalimide synthesis
- Incompatibility with Secondary Alkyl Halides: The Gabriel method generally does not work well with secondary alkyl halides. This limitation restricts the applicability of the method to specific types of substrates.
- Hydrolysis Issues: The use of acidic or basic hydrolysis in the Gabriel Phthalimide synthesis is associated with minimal yields. Additionally, hydrazine hydrolysis, which is often employed, involves harsh conditions that can be detrimental to the reaction’s efficiency and scalability.
- Reaction Mechanism Constraints: The Gabriel Phthalimide synthesis relies on an SN2 reaction between phthalimide and an alkyl halide. This mechanism can be less effective in certain scenarios, potentially leading to lower yields or incomplete reactions.
- Scalability and Commercial Viability: Some variations of the Gabriel synthesis, such as those involving reduction of alkyl phthalimides, may face challenges in terms of scalability and commercial viability. These methods might require optimization to be suitable for industrial applications.
- Environmental and Economic Considerations: Traditional Gabriel synthesis methods often use costly reagents and harsh chemicals, which hinder large-scale commercial adoption. These methods also involve long reaction times, reducing overall process efficiency and increasing operational costs. Recent advances focus on replacing corrosive substances with safer, greener alternatives to improve sustainability. Researchers now prefer phthalimide-based approaches that offer higher yields and better cost-effectiveness. These innovations make Gabriel synthesis more suitable for scalable and environmentally responsible chemical production.
FAQs
Because the SN2 reaction step is hindered with secondary alkyl halides, and it risks elimination or side products.
Hydrazine cleaves the N–C bond in the N-alkylphthalimide to liberate the primary amine under milder conditions than acid or base hydrolysis.
Yes, benzyl halides (primary) work well because they are reactive in SN2 conditions.
Phthalimide’s rigid structure and strong electron-withdrawing groups make it a more stable nucleophile, ideal for alkylation and clean deprotection.
Conclusion
The Gabriel Phthalimide Synthesis remains a reliable, selective, and widely used method for preparing primary amines in both academic and industrial chemistry. Its straightforward mechanism and predictable outcomes make it a cornerstone reaction in organic synthesis. Whether you’re building amine-containing drugs or intermediates, Gabriel synthesis offers a clean and effective pathway to primary amines.
How PatSnap Eureka Accelerates Gabriel Phthalimide Synthesis Innovation
Gabriel Phthalimide Synthesis is a classic method for preparing primary amines, essential in pharmaceuticals, agrochemicals, and advanced materials. As the demand for selective and scalable amine synthesis grows, PatSnap Eureka empowers researchers with real-time insights into the evolving landscape of this foundational reaction.
- Patent Intelligence: Eureka scans global patents to reveal innovations in Gabriel Phthalimide Synthesis, including new reagents and catalysts. It helps research teams discover faster, avoid duplication, and optimize chemical process development more effectively.
- Competitive Landscape Tracking: Benchmark how leading companies apply Gabriel methods in drug discovery, fine chemical synthesis, and green chemistry. Track key players’ strategies and stay competitive in both pharmaceutical and sustainable chemistry sectors.
- Trend Forecasting: Eureka’s AI tools detect emerging Gabriel synthesis uses, including bioactive molecule development and feedstock conversion.
- Technical Clustering: Visual clustering highlights key research areas, including reaction scalability, regioselectivity enhancements, and integration into multistep synthetic routes.
From academic labs to industrial pipelines, Eureka drives faster, smarter innovation in Gabriel Phthalimide Synthesis by turning complex data into actionable insight.
To get detailed scientific explanations of Gabriel Phthalimide Synthesis, try Patsnap Eureka.


