
Balance redox reactions can be one of the trickiest parts of high school or college chemistry. Redox (short for reduction-oxidation) reactions involve the transfer of electrons between species, meaning both mass and charge must be balanced. If that sounds intimidating, don’t worry—this guide will break it down into manageable steps and share practical tips to make balancing redox reactions much easier.
Struggling with redox reactions? Eureka Technical Q&A makes balancing redox reactions simple with proven tricks like the half-reaction method, oxidation number changes, and step-by-step examples—helping you master this key chemistry skill with confidence.
What Is a Redox Reaction?
A redox reaction, short for reduction-oxidation reaction, is a chemical reaction in which the transfer of electrons occurs between two species. This transfer results in the simultaneous occurrence of oxidation and reduction processes. In a redox reaction, one substance is oxidized, meaning it loses electrons, while another substance is reduced, meaning it gains electrons. This exchange of electrons leads to changes in the oxidation states of the involved species.
A redox reaction involves two processes:
These always occur together. One species loses electrons (oxidized), while another gains them (reduced). The key to balancing redox reactions is making sure the number of electrons lost equals the number gained.
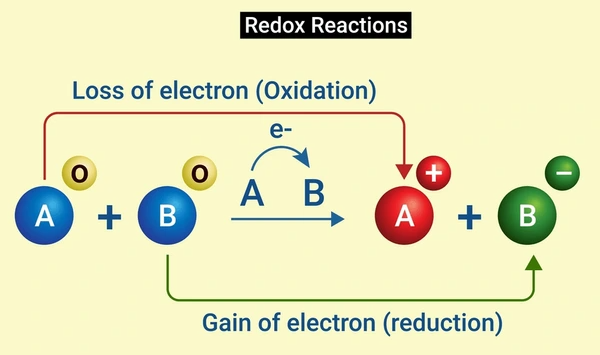
Step-by-Step Guide to Balancing Redox Reactions
- Identify and write the half-reactions: The first step is to separate the redox reaction into two half-reactions. One half-reaction involves oxidation, where a molecule, atom, or ion loses electrons, and the other half-reaction involves reduction, where a molecule, atom, or ion gains electrons. Write these half-reactions separately.
- Balance the atoms other than O and H: In the half-reactions, balance all atoms except for oxygen and hydrogen. This can be done by adding or subtracting electrons to one side of the equation to make the number of atoms on both sides equal.
- Balance the oxygen atoms by adding H2O: To balance the oxygen atoms in the half-reactions, add water molecules (H2O) to the side with fewer oxygen atoms. For each oxygen atom added, add two hydrogen atoms (as H2O) to the same side.
- Balance the hydrogen atoms by adding H+: To balance the hydrogen atoms, add hydrogen ions (H+) to the side with fewer hydrogen atoms. For each hydrogen ion added, add one electron to the same side.
- Equalize the number of electrons in both half-reactions: Make sure that the number of electrons lost in the oxidation half-reaction equals the number of electrons gained in the reduction half-reaction. If the electrons are not equal, multiply one or both half-reactions by appropriate integers to balance them.
- Add the half-reactions: Once the electrons are balanced, add the two half-reactions together. Combine like terms and cancel out any common factors on both sides of the equation.
- Check your work: Finally, check your balanced redox reaction to ensure that the number of atoms of each element is equal on both sides and that the charge is balanced.
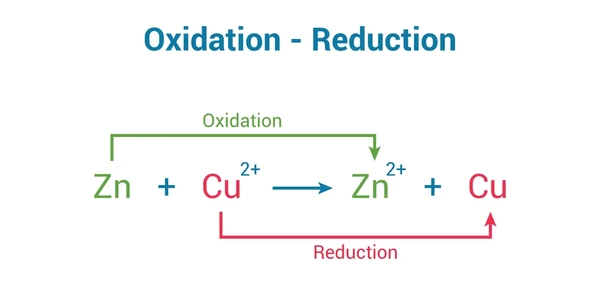
2. Oxidation Number Method
Step 1: Assign oxidation numbers to all atoms
Use standard rules to find what is oxidized and reduced.
Step 2: Identify the atoms that change oxidation state
Determine how many electrons are gained or lost.
Step 3: Multiply to equalize electron transfer
Make the total increase and decrease in oxidation number equal.
Step 4: Balance remaining atoms
Balance the rest of the atoms in the equation (O, H, etc.) using H₂O, H⁺, and OH⁻ as needed.
Tricks and Tips for Success
Use Mnemonics
- OIL RIG: Oxidation Is Loss, Reduction Is Gain (of electrons)
- LEO the lion says GER: Lose Electrons = Oxidation, Gain Electrons = Reduction
Balance in Acid or Base?
- Use H⁺ and H₂O in acidic solutions
- Use OH⁻ and H₂O in basic solutions
Always Check:
- Atom counts for each element
- Total charges on both sides
- Electrons lost = electrons gained
Practice Common Redox Reactions
- Metal displacement: Zn + Cu²⁺ → Zn²⁺ + Cu
- Combustion: CH₄ + O₂ → CO₂ + H₂O
- Permanganate reactions in acid or base
Example: Balancing in Acidic Solution
Unbalanced Reaction:
MnO₄⁻ + Fe²⁺ → Mn²⁺ + Fe³⁺
Step 1: Split into half-reactions
MnO₄⁻ → Mn²⁺
Fe²⁺ → Fe³⁺
Step 2: Balance atoms
MnO₄⁻ → Mn²⁺ (add 4 H₂O to right)
Add 8 H⁺ to left: 8H⁺ + MnO₄⁻ → Mn²⁺ + 4H₂O
Step 3: Balance charge
Left: 8H⁺ (+8) + MnO₄⁻ (–1) = +7
Right: Mn²⁺ = +2
Add 5 electrons to left
Fe²⁺ → Fe³⁺ + 1 electron
Step 4: Multiply Fe half-reaction by 5
5Fe²⁺ → 5Fe³⁺ + 5e⁻
Step 5: Combine
8H⁺ + MnO₄⁻ + 5Fe²⁺ → Mn²⁺ + 4H₂O + 5Fe³⁺
FAQs
The half-reaction method is generally more systematic and easier to follow, especially in acidic or basic solutions.
Only if the reaction occurs in an aqueous solution. In acidic media, use H⁺. In basic media, use OH⁻.
Yes. Redox is about electron transfer, not just oxygen. Many redox reactions don’t involve oxygen at all.
They’re the “currency” of redox. One species loses them (oxidation), another gains them (reduction).
Conclusion
Balancing redox reactions may seem challenging, but with a clear process and a few helpful tricks, it becomes much more manageable. Whether you’re a student studying for exams or a chemistry enthusiast, mastering redox balancing will strengthen your understanding of chemical reactions and their underlying principles. Practice a few examples, follow the steps, and soon it’ll feel simple.
To get detailed scientific explanations of Balancing Redox Reactions, try Patsnap Eureka.


