
Alkyl halide, also known as haloalkanes, are organic compounds in which one or more hydrogen atoms in an alkane are replaced by halogen atoms like fluorine, chlorine, bromine, or iodine. These compounds play a central role in synthetic organic chemistry and serve as key intermediates in the manufacture of pharmaceuticals, agrochemicals, polymers, and refrigerants.
This article explores the structure, classification, and important reactions of alkyl halides, offering insight into their practical applications and reactivity trends.
Structure of Alkyl Halides
What is an alkyl halide? Eureka Technical Q&A explains that alkyl halides are organic compounds containing a halogen bonded to an alkyl group. Learn about their structure, classification (primary, secondary, tertiary), and how they undergo substitution and elimination reactions in organic chemistry.
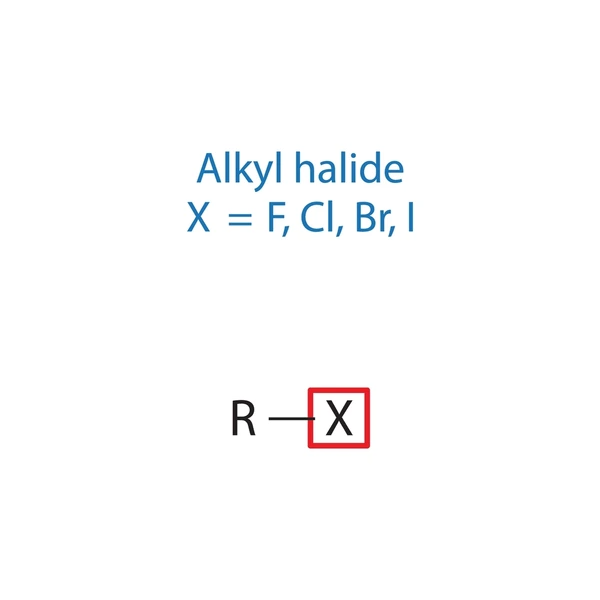
An alkyl halide consists of a hydrocarbon chain (alkyl group) attached to a halogen atom (X). The general formula is:
R–X
Where:
- R = alkyl group (such as methyl, ethyl, propyl)
- X = halogen atom (F, Cl, Br, or I)
The carbon-halogen bond is polar covalent due to the difference in electronegativity between carbon and the halogen. This polarity influences both the physical properties and chemical reactivity of alkyl halides.
Classification of Alkyl Halides
1. Based on the Type of Carbon Atom
- Primary (1°) Alkyl Halide: Halogen is attached to a carbon bonded to one other carbon.
Example: CH₃CH₂Cl (ethyl chloride) - Secondary (2°) Alkyl Halide: Halogen is attached to a carbon bonded to two other carbons.
Example: CH₃CHClCH₃ (isopropyl chloride) - Tertiary (3°) Alkyl Halide: Halogen is attached to a carbon bonded to three other carbons.
Example: (CH₃)₃CCl (tert-butyl chloride)
2. Based on the Number of Halogen Atoms
- Mono-halides: One halogen atom (e.g., methyl bromide)
- Di-halides: Two halogen atoms (e.g., 1,2-dibromoethane)
- Tri-halides: Three halogen atoms (e.g., chloroform – CHCl₃)
Physical Properties of Alkyl Halides
- Boiling points increase with molecular weight and decrease with branching.
- Solubility: Alkyl halides are insoluble in water but soluble in organic solvents.
- Density: Most are denser than hydrocarbons but less dense than water, except for polyhalogenated compounds.
Reactions of Alkyl Halides
Alkyl halides are reactive due to the electrophilic carbon bonded to the halogen, making them ideal substrates for nucleophilic reactions.
1. Nucleophilic Substitution Reactions
SN1 (Unimolecular)
- Favors tertiary halides
- Involves carbocation intermediate
- Solvent: polar protic (e.g., water, alcohol)
Example:
(CH₃)₃CBr + H₂O → (CH₃)₃COH + HBr
SN2 (Bimolecular)
- Favors primary halides
- One-step backside attack with inversion of configuration
- Solvent: polar aprotic (e.g., acetone, DMSO)
Example:
CH₃CH₂Br + OH⁻ → CH₃CH₂OH + Br⁻
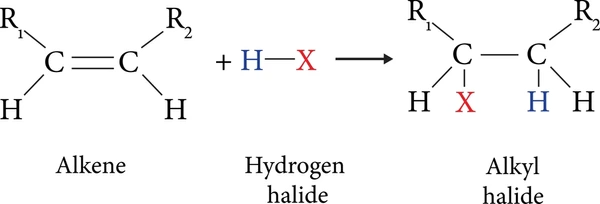
2. Elimination Reactions
Alkyl halides can undergo β-elimination to form alkenes, especially in the presence of strong bases like KOH or NaOEt.
Example:
CH₃CH₂CHBrCH₃ + KOH → CH₃CH=CHCH₃ + KBr + H₂O
3. Reaction with Metals (Wurtz Reaction)
In dry ether, alkyl halides react with sodium metal to form alkanes.
Example:
2 CH₃Br + 2 Na → CH₃–CH₃ + 2 NaBr
4. Formation of Grignard Reagents
When reacted with magnesium in dry ether, alkyl halides form Grignard reagents, useful in carbon-carbon bond formation.
Example:
CH₃Br + Mg → CH₃MgBr
Industrial and Laboratory Uses
- Solvents: Such as chloroform and carbon tetrachloride
- Refrigerants: Fluorinated haloalkanes like Freon (now phased out)
- Pharmaceuticals: Starting materials for drug synthesis
- Pesticides and Herbicides: Alkyl halides form key components
- Synthetic Intermediates: In the production of plastics and dyes
Application Cases
| Product/Project | Technical Outcomes | Application Scenarios |
|---|---|---|
| Carbonate Absorption/Stripping Process KC8 Capture Technologies Ltd. | Utilizes potassium chloride to produce potassium hydroxide and hydrogen, capturing carbon dioxide and generating valuable by-products | Industrial emissions reduction and carbon capture in fertilizer industry |
| Chemosensors for Alkylating Agents Technion Research & Development Foundation Ltd. | Enables sensitive and selective identification of alkylating agents, including weakly electrophilic ones | Detection of alkylating agents in various phases for environmental and safety monitoring |
| Amino-terminated Polymer Additives Atotech Deutschland GmbH & Co. KG | Controls molecular weight and halogen content to improve deposition quality and efficiency in zinc electrodeposition | Zinc and zinc alloy electroplating processes in industrial manufacturing |
| Isotopically Labeled Reagents Revvity Health Sciences, Inc. | Enhanced stability formulation as L – (aCbH2)πaC H3(I) for extended storage and usage | Isotopic labeling reactions in pharmaceutical research and development |
| NC=S Compound Additive Nippon Soda Co., Ltd. | Prevents decomposition during distillative purification by converting residual palladium into an inactive form | Large-scale industrial production of aryl-, heteroaryl-, or alkenyl-substituted unsaturated hydrocarbons |
Safety and Environmental Considerations
Many alkyl halides, especially polyhalogenated compounds, are toxic, carcinogenic, or ozone-depleting. Examples include:
- Chlorofluorocarbons (CFCs): Banned due to ozone depletion
- Chloroform: Potential carcinogen with CNS depressant effects
- Carbon tetrachloride: Highly toxic to the liver
Proper handling, ventilation, and disposal protocols are critical.
FAQs
Yes, due to the polar carbon-halogen bond, they are considered polar molecules, although their overall polarity depends on structure and symmetry.
The electronegative halogen withdraws electron density from the carbon, making it an electrophile that attracts nucleophiles.
Haloalkanes have the halogen attached to an aliphatic carbon, while aryl halides have it attached to an aromatic ring, which influences reactivity.
No. SN2 reactions are favored by primary alkyl halides and are hindered in tertiary structures due to steric effects.
On hydrolysis, certain alkyl halides can form alcohols, which can be further oxidized to form acids, but the halide itself does not directly produce an acid.
Conclusion
Alkyl halides are versatile and important organic compounds characterized by a carbon-halogen bond. Their classification, reactivity, and use in various chemical transformations make them foundational in both academic and industrial chemistry. Understanding how they behave in substitution, elimination, and metalation reactions is key to mastering organic synthesis and exploring their vast applications.
How PatSnap Eureka Accelerates Innovation in Alkyl Halides Research
Alkyl halides are fundamental building blocks in organic synthesis, widely used in pharmaceuticals, agrochemicals, and materials science. PatSnap Eureka enables researchers to stay ahead by uncovering how innovations involving alkyl halides are evolving across industries.
- Patent Intelligence: Eureka scans global patent databases to identify cutting-edge developments in alkyl halide synthesis, functionalization, and applications in cross-coupling or substitution reactions.
- Competitive Landscape Tracking: Benchmark how leading organizations are leveraging alkyl halides in scalable synthetic routes, drug design, and industrial chemistry processes.
- Trend Forecasting: Eureka’s AI-driven tools highlight emerging uses of alkyl halides in areas like green chemistry, fluorinated compound development, and advanced material synthesis.
- Technical Clustering: Visual clustering reveals innovation hotspots in alkyl halide chemistry, such as novel halogenation methods, catalytic transformations, and bio-based alternatives.
With PatSnap Eureka, teams can quickly access strategic insights and technical intelligence to drive forward-thinking innovation in alkyl halides research and development.
To get detailed scientific explanations of alkyl halide, try Patsnap Eureka.

