
In organic chemistry, stereochemistry plays a crucial role in how molecules behave and interact. One key concept is the racemic mixture, a blend of two enantiomers—molecules that are mirror images of each other. Though identical in composition, these enantiomers can have very different biological or chemical behaviors.
This article explains what a racemic mixture is, how it forms, its properties, and why it’s important in pharmaceuticals, synthesis, and chemical analysis.
What Is a Racemic Mixture?
What is a racemic mixture? Eureka Technical Q&A explains that it’s a 50:50 blend of two enantiomers—molecules that are mirror images—resulting in no net optical activity due to their equal and opposite effects on plane-polarized light.
A racemic mixture (also called a racemate) is a 50:50 mixture of two enantiomers of a chiral molecule. Enantiomers are non-superimposable mirror images—just like your left and right hands. A racemic mixture contains equal amounts of these left- and right-handed forms.
Notation
- Represented as (±)- or rac-
For example: (±)-lactic acid or rac-lactic acid
Properties of Racemic Mixtures
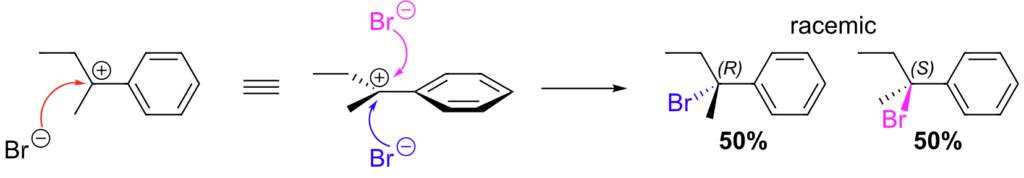
1. Optical Inactivity
Although each enantiomer can rotate plane-polarized light (one clockwise, one counterclockwise), a racemic mixture shows no net optical rotation because the effects cancel each other out. This is a defining feature of racemates.
2. Physical Properties
Racemic mixtures may have different melting points, boiling points, or solubility compared to the pure enantiomers.
- The melting point of a racemate can be lower than that of the pure enantiomers (a phenomenon called racemic melting point depression).
- Crystallization behavior may also differ—some racemates form racemic crystals, while others may form separate crystals of each enantiomer.
3. Biological Activity
In biological systems, enantiomers can have dramatically different effects. In a racemic drug, typically only one enantiomer is active. The other may be inactive or even harmful.
How Racemic Mixtures Are Formed

1. Chemical Synthesis
Most chemical reactions that create chiral molecules from achiral reactants produce racemic mixtures unless chemists use a chiral catalyst or environment.
Example:
Hydrogenation of prochiral alkenes using an achiral catalyst typically gives a 1:1 mixture of enantiomers.
2. Racemization
A pure enantiomer can convert to a racemic mixture through a process called racemization, where environmental factors (like heat or pH) cause one enantiomer to convert into the other.
Examples of Racemic Mixtures
1. Racemic Ibuprofen
The over-the-counter pain reliever ibuprofen is commonly sold as a racemic mixture. However, only the S-enantiomer is biologically active. The R-form is less effective but can be converted in the body to the active form.
2. Racemic Thalidomide
One of the most infamous examples, thalidomide was sold as a racemic mixture. The R-enantiomer was effective against morning sickness, while the S-enantiomer caused severe birth defects—highlighting the importance of stereochemistry in pharmaceuticals.
3. Racemic Lactic Acid
Naturally occurring lactic acid in muscles is L-lactic acid, but in lab synthesis, lactic acid is often produced as a racemate containing both D- and L-forms.
Applications of Racemic Mixtures
Liquid Crystals
Researchers add racemic mixtures to liquid crystals to enhance the antiferroelectric phase range and reduce rotational viscosity. These enhancements improve optical device performance and expand their range of applications.
Chiral Separation
Scientists separate racemic mixtures into enantiomers to ensure proper drug activity and safety. Techniques like chiral chromatography and crystallization allow precise separation for pharmaceutical use.
Biocatalysis
Industries use biocatalysts, such as specific fungi, to resolve racemic mixtures into pure enantiomers. This green approach supports sustainable and cost-effective chemical production.
Material Science
Engineers use racemic mixtures to develop advanced chiral polymers and hybrid materials. These materials improve performance in separation membranes and catalytic applications.
Pharmaceuticals
Pharmaceutical companies use racemic intermediates to develop enantiomerically pure drugs. Accurate resolution ensures drug safety, potency, and regulatory compliance.
Catalysis
Chemists employ racemic compounds as catalysts in asymmetric synthesis to favor one enantiomer over another. This selectivity increases reaction efficiency and product value.
Natural Product Synthesis
Scientists start with racemic mixtures to synthesize complex natural products. Organisms selectively consume one enantiomer, producing bioactive compounds like D-amino acids.
Application Cases
| Product/Project | Technical Outcomes | Application Scenarios |
|---|---|---|
| GAL-021 Galleon Pharmaceuticals, Inc. | Enhanced respiratory stimulation with minimized side effects through isolation of (+)-enantiomer of doxapram. | Treatment of breathing disorders and respiratory depression. |
| Chiral Polymer Resolution System Celanese Corp. | Resolves racemic compounds into optically active isomers without chemical transformation using polymers with chiral moieties. | Separation of enantiomers in pharmaceutical and chemical industries. |
| Optically-Active Amphiphilic Copolymers The Dow Chemical Co. | Achieves high enantiomeric excess and improved yields through diastereomeric interactions and crystallization. | Efficient separation of enantiomers in racemic mixtures for pharmaceutical and fine chemical applications. |
| Nanoporous Silica Chiral Separation Council of Scientific & Industrial Research | High enantiomeric purity and loading capacity through sol-gel protocol with tetraethylorthosilicate and amino alkylsilane. | Cost-effective resolution of racemic compounds in pharmaceutical and chemical industries. |
| Graphene-Based Chiral Separation Materials UNM Rainforest Innovations | Efficient chiral separation of racemic mixtures using graphene-based hybrid materials with enantiospecific organic molecules. | Cost-effective isolation of pure enantiomers for improved pharmacological efficacy. |
Separation of Enantiomers from a Racemic Mixture
This process is called resolution. Common techniques include:
- Chiral chromatography: Using a chiral stationary phase
- Diastereomeric salt formation: Reacting with a chiral acid or base to form separable compounds
- Biocatalysis: Using enzymes that selectively react with one enantiomer
These methods are essential in the production of enantiopure pharmaceuticals.
FAQs
Because the two enantiomers rotate light in opposite directions with equal magnitude, their effects cancel out, resulting in no net rotation.
Yes. Techniques like chiral chromatography, crystallization, or enzymatic resolution can isolate individual enantiomers.
By definition, yes. If the ratio is not equal, the mixture is called scalemic or enantiomerically enriched.
Unless a chiral environment or reagent is used, most reactions that produce chiral molecules generate both enantiomers in equal amounts.
Yes, if both enantiomers exist and can be synthesized or converted under suitable conditions, a racemic form can be produced.
Conclusion
A racemic mixture is an equal blend of two enantiomers of a chiral molecule. Though it may appear simple, the racemate has unique chemical, physical, and biological characteristics that differ from its individual components. Understanding racemic mixtures is essential in fields like drug development, stereochemistry, and asymmetric synthesis, where molecular orientation directly affects function and safety.
How PatSnap Eureka Accelerates Research on Racemic Mixtures
Racemic mixtures are central to pharmaceutical and chemical synthesis, where enantiomeric purity directly impacts efficacy, safety, and regulatory outcomes. PatSnap Eureka empowers researchers with deep insights into how racemic mixtures are being studied, resolved, and applied across industries.
- Patent Intelligence: Eureka scans global patent databases to uncover innovations in racemic mixture resolution, asymmetric synthesis, and chiral separation techniques—streamlining discovery and reducing duplication.
- Competitive Landscape Tracking: Analyze how pharmaceutical and chemical companies handle racemic mixtures in drug development, material design, and stereoselective manufacturing.
- Trend Forecasting: Eureka’s AI-driven analytics reveal where racemic mixture applications are growing, from enantiopure drug synthesis to green chemistry approaches in chiral technologies.
- Technical Clustering: Visual clustering highlights research hotspots related to racemic mixtures, such as novel catalysts, solvent systems, and scalable enantioselective processes.
With PatSnap Eureka, scientists and R&D teams can make faster, more informed decisions in the development and application of racemic mixtures across competitive innovation landscapes.
To get detailed scientific explanations of racemic mixture, try Patsnap Eureka.


