
In every chemical reaction, one reactant is used up before the others. This substance is called the limiting reactant—and it’s the key to determining how much product a reaction can actually produce. Without knowing the limiting reactant, it’s impossible to predict reaction yields accurately.
This article explains what a limiting reactant is, how to identify it, and how to calculate it using simple steps and real-world examples.
What Is a Limiting Reactant?
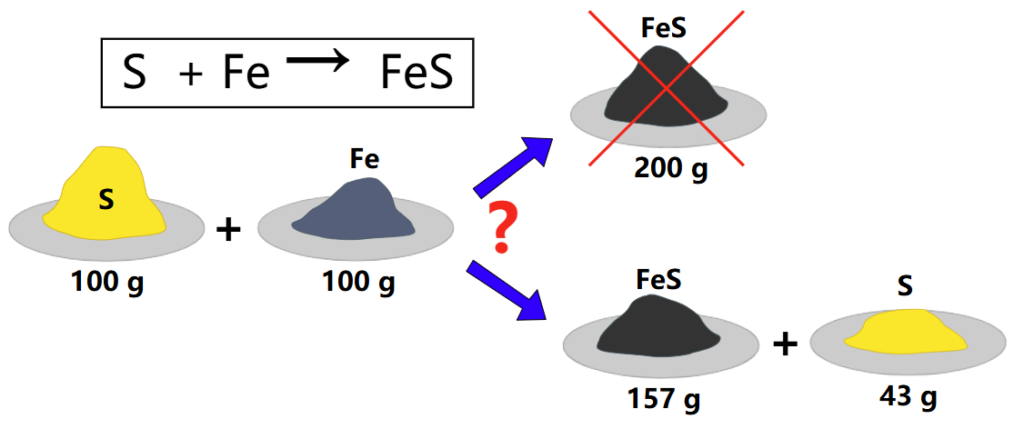
What is a limiting reactant? Eureka Technical Q&A explains that it’s the substance in a chemical reaction that runs out first, determining the maximum amount of product that can be formed.
The limiting reactant (or limiting reagent) is the reactant that runs out first during a chemical reaction, limiting the amount of product that can form. Once this reactant is completely consumed, the reaction stops, even if other reactants are still available.
In contrast, excess reactants are those that remain unused after the reaction is complete.
Why Is It Important?
Knowing the limiting reactant allows you to:
- Predict the maximum amount of product formed
- Avoid wasting excess reactants
- Optimize industrial and lab-scale reactions
- Perform accurate stoichiometric calculations
Step-by-Step: How to Identify the Limiting Reactant
Step 1: Write a Balanced Chemical Equation
A balanced equation ensures you use the correct mole ratio between reactants and products.
Example:
2 H₂ + O₂ → 2 H₂O
Step 2: Convert Quantities to Moles
Use molar mass or given mole values to determine how much of each reactant you have.
Example:
Given: 5 moles of H₂ and 3 moles of O₂
Step 3: Divide by Coefficients (Mole Ratio Test)
Divide the available moles by the stoichiometric coefficient from the balanced equation.
- H₂: 5 mol ÷ 2 = 2.5
- O₂: 3 mol ÷ 1 = 3
Step 4: Identify the Smallest Result
The smallest value determines the limiting reactant. In this case, H₂ is limiting (2.5 < 3).
Example Problem: Limiting Reactant Calculation
Reaction:
N₂ + 3 H₂ → 2 NH₃
Given:
- 1 mole of N₂
- 2 moles of H₂
Step 1: Convert to Mole Ratio
- N₂: 1 ÷ 1 = 1
- H₂: 2 ÷ 3 ≈ 0.67
Step 2: Compare Values
Since 0.67 < 1, H₂ is the limiting reactant.
Step 3: Calculate Product Formed
H₂ is limiting and follows a 3:2 ratio to NH₃.
2 mol H₂ → (2/3) × 2 = 1.33 mol NH₃
Shortcut Method Using Stoichiometry
You can also find the limiting reactant using a stoichiometry table or by calculating how much of one reactant is needed to completely react with the other. The one that requires more than available is in excess; the other is limiting.
Real-World Applications
Chemical Reactions and Manufacturing
In chemical processes, the substance that gets used up first controls the maximum yield of the final product. This concept plays a key role in manufacturing, where precise reactant proportions are needed to maximize efficiency and minimize waste.
Pharmaceuticals
In drug production, using the correct reactant ratios ensures accurate dosage and minimizes byproducts. Identifying which substance will run out first helps maintain consistency and safety in medication batches.
Everyday Scenarios
This principle applies to common tasks like cooking and fuel usage. For example, in baking, the smallest available ingredient sets the limit for how many cookies you can make.
Stoichiometry in Problem Solving
Calculating which component is in short supply is a key part of stoichiometry. It helps chemists and engineers solve real-world problems by predicting outcomes based on reactant quantities.
Application Cases
| Product/Project | Technical Outcomes | Application Scenarios |
|---|---|---|
| Catalysis Technology BASF | Optimized use of reactants, increased yield and efficiency in chemical reactions | Large-scale industrial chemical production |
| Drug Formulation Process Pfizer | Precise control of reactant ratios, ensuring accurate medication production | Pharmaceutical manufacturing and drug development |
| Food Processing Technology Nestle | Improved ingredient utilization, reduced waste in food production | Large-scale food manufacturing and product development |
| Fuel Cell Technology Toyota | Enhanced fuel efficiency through optimized reactant usage in hydrogen fuel cells | Automotive industry, particularly in hydrogen-powered vehicles |
| Chemistry Lab Equipment Vernier Software & Technology | Accurate measurement and analysis of limiting reactants in educational experiments | Chemistry education and laboratory research |
Common Mistakes to Avoid
- Not using a balanced equation
- Forgetting to convert mass to moles
- Assuming the smaller mass = limiting reactant (not always true)
- Ignoring the mole ratio in stoichiometry
FAQs
No. In any given reaction, only one reactant limits the formation of the product.
They remain unreacted after the reaction is complete.
Only reactions with unequal reactant quantities have a limiting reactant. In ideal cases with exact mole ratios, no limiting reactant exists.
Not necessarily. It depends on the mole ratio and the stoichiometry of the reaction, not just the quantity.
Conclusion
Understanding the limiting reactant is essential for mastering chemical reactions. It determines the maximum yield and affects how efficiently a process runs. By following a few simple steps—balancing equations, converting to moles, and applying stoichiometry—you can accurately identify the limiting reagent and make better predictions in both academic and practical chemistry settings.
How PatSnap Eureka Accelerates Research on Limiting Reactant Applications
The concept of the limiting reactant is fundamental in reaction optimization, process efficiency, and yield prediction across chemical manufacturing and pharmaceuticals. PatSnap Eureka empowers researchers with actionable insights to explore how this principle is being applied, optimized, and patented across industries.
- Patent Intelligence: Eureka scans global patent databases to identify innovations that involve limiting reagent strategies, including stoichiometric control, scalable synthesis methods, and efficiency-driven process designs.
- Competitive Landscape Tracking: Analyze how top companies are incorporating limiting reagent optimization into industrial chemistry, continuous flow systems, and green synthesis techniques.
- Trend Forecasting: Eureka’s AI-driven analytics reveal where limiting reagent applications are gaining ground—such as in sustainable chemistry, waste reduction, and high-throughput experimentation.
- Technical Clustering: Visualimiting reagent management in multi-step synthesis, catalyst usage, and resource-efficient production pathways.
With data-driven insights from Eureka, R&D teams can make smarter decisions around limiting reagent usage, enabling more efficient, innovative, and sustainable chemical processes.
To get detailed scientific explanations of limiting reactant, try Patsnap Eureka.


