
A neutralization reaction is a fundamental concept in chemistry that plays a vital role in everything from medicine to agriculture to wastewater treatment. At its core, a neutralization reaction occurs when an acid reacts with a base to form water and a salt. This process neutralizes the acidic and basic properties of the reactants, making the resulting solution more stable or neutral in pH. In this article, we’ll break down how neutralization reactions work, explore real-life examples, and show you how to write and balance these reactions effectively.
What is a neutralization reaction? Eureka Technical Q&A explains that it’s a chemical reaction where an acid and a base react to form water and a salt, helping you understand its role in pH balance, titrations, and real-world applications like antacids and wastewater treatment.
Definition of Neutralization Reaction
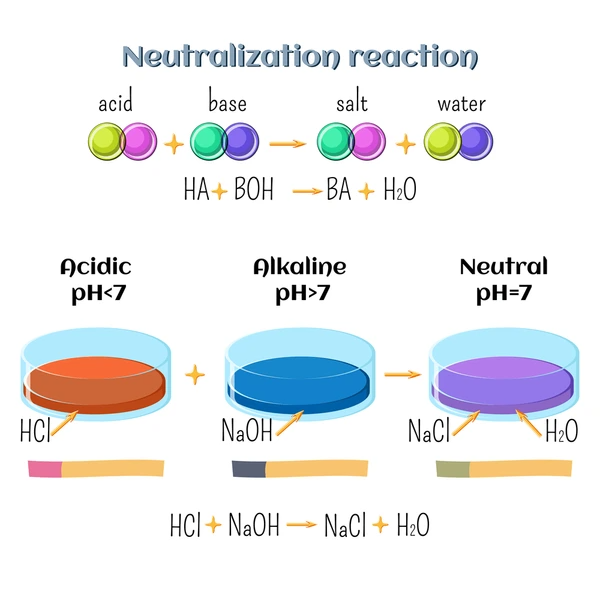
A neutralization reaction is a chemical reaction between an acid and a base, producing a salt and water as products.
General Formula:
Acid + Base → Salt + Water
This reaction typically involves the combination of hydrogen ions (H⁺) from the acid and hydroxide ions (OH⁻) from the base to form H₂O.
Common Examples
- Hydrochloric acid + Sodium hydroxide HCl + NaOH → NaCl + H₂O
- Sulfuric acid + Potassium hydroxide H₂SO₄ + 2KOH → K₂SO₄ + 2H₂O
- Acetic acid + Ammonia CH₃COOH + NH₃ → CH₃COONH₄
Even weak acids and weak bases can participate in neutralization, though the resulting pH may not be exactly.
Types of Neutralization Reactions
1. Strong Acid + Strong Base
Produces a neutral solution with a pH of ~7.
Example: HNO₃ + KOH → KNO₃ + H₂O
2. Strong Acid + Weak Base
The resulting solution is slightly acidic.
Example: HCl + NH₃ → NH₄Cl
3. Weak Acid + Strong Base
Produces a slightly basic solution.
Example: CH₃COOH + NaOH → CH₃COONa + H₂O
4. Weak Acid + Weak Base
Often produces a neutral salt but may require more analysis to predict pH.
Applications of Neutralization Reactions
- Industrial Applications:
- Chemical Production: Chemical manufacturers widely use neutralization reactions to produce essential compounds. For example, fertilizer producers neutralize acidic and basic substances to create products ideal for agriculture.
- Pesticide and Explosives Production: Scientists also apply these reactions in synthesizing pesticides and explosives, where they must precisely control the chemical processes.
- Environmental Applications:
- Soil Treatment: In agriculture, neutralization reactions help improve soil quality by adjusting its pH levels, making it more conducive for plant growth.
- Waste Management: Industries use neutralization to treat acidic or basic waste streams, making them less harmful before disposal.
- Everyday Life:
- First Aid: Antacids, which are used to relieve heartburn and indigestion, work through neutralization reactions. They neutralize stomach acid to provide relief.
- Household Cleaners: Many household cleaners and detergents rely on neutralization reactions to break down stains and grime effectively.
- Technological Applications:
- Ion Trap Mass Spectrometry: In this technology, neutralization reactions are controlled to analyze and separate ions, which is crucial for accurate mass spectrometry.
- Electrodialytic Cells: These cells generate electrical energy through neutralization reactions, showcasing a unique application of this chemical principle.
- Biological and Medical Applications:
- Allergy Treatment: Research has explored the use of neutralization reactions in developing treatments for allergic reactions by targeting specific proteins involved in these responses.
Application Cases
| Product/Project | Technical Outcomes | Application Scenarios |
|---|---|---|
| Ammonium Nitrate Reactor Yara International ASA | Coaxial acid sparger and injection pipe design controls pH and temperature, optimizing acid-base reaction and preventing hot spots. | Efficient and safe production of ammonium nitrate fertilizer. |
| Ion Trap Mass Spectrometer Hitachi High-Tech America, Inc. | Dipolar DC fields control ion/ion reactions, inhibiting charge transfer between positive and negative ions. | Enhanced analysis and processing of high mass molecules like proteins. |
| Bipolar Membrane Cell The Graver Co. | Generates electrical potential difference from acid-base neutralization, enabling regenerative energy storage. | Production of commercially significant electrical energy from chemical reactions. |
| Structured Liquid Catalyst American Technologies Group, Inc. | Enhances reaction rates and biological growth using structured liquids with induced electric dipole moments. | Catalyzing chemical and biological reactions with increased efficiency and cost reduction. |
How to Identify a Neutralization Reaction
- Presence of Acid and Base: Neutralization reactions occur when an acid and a base come into contact. The acid typically contains hydrogen ions (H+), while the base contains hydroxide ions (OH-).
- Formation of Salt and Water: One of the main products of a neutralization reaction is water (H2O), and a salt is also formed. This is because the hydrogen ions from the acid react with the hydroxide ions from the base to form water, while the remaining ions combine to form a salt.
- Double Displacement Reaction: Neutralization reactions are often classified as double displacement reactions, where the ions of the reactants switch places to form new products.
- Quantitative Reaction: Neutralization reactions are typically quantitative, meaning they occur in a specific ratio. For example, one molecule of acid reacts with one molecule of base to produce one molecule of salt and one molecule of water.
- pH Change: As the reaction proceeds, the pH of the solution will change. Initially, the solution may be acidic if the acid is in excess, or basic if the base is in excess. As the reaction progresses, the pH will approach neutral (pH 7) as the acid and base are consumed.
- Heat Release: Some neutralization reactions may release heat, which can be observed as a temperature increase in the reaction mixture.
FAQs
Most acid-base reactions are neutralization reactions, especially if water is formed. However, some may produce other products depending on the reactants.
Only when a strong acid reacts with a strong base. If one or both are weak, the final pH can be slightly acidic or basic.
Yes. Some reactions release gases like CO₂, especially when carbonates are involved.
Neutralization is the reaction itself; titration is a technique that uses neutralization to determine concentration.
Conclusion
Neutralization reactions are a cornerstone of chemistry, occurring whenever an acid and a base interact to form a salt and water. Understanding this process is not only key to grasping chemical principles, but also essential for real-world applications in healthcare, environmental science, and industry. Whether you’re neutralizing stomach acid or balancing soil pH, the concept remains the same: bringing opposites together to achieve balance.
To get detailed scientific explanations of neutralization reactions, try Patsnap Eureka.


