
Have you ever mixed two clear solutions and seen a cloudy solid suddenly appear? That’s the magic of a precipitation reaction in action. These reactions are common in chemistry labs, water treatment plants, and even inside your body. But what exactly is happening when a solid forms out of a liquid? In this article, we’ll explain what a precipitation reaction is, how to recognize one, provide clear examples, and show where these reactions appear in real life.
What is a precipitation reaction? Eureka Technical Q&A explains that it’s a chemical reaction where two soluble solutions combine to form an insoluble solid, or precipitate—commonly used in labs, water treatment, and chemical analysis.
Definition of a Precipitation Reaction
A precipitation reaction is a type of chemical reaction in which dissolved substances, typically ions in a solution, combine to form an insoluble solid product, known as a precipitate. This process occurs when the solubility of the products in the solution is exceeded, leading to the formation of solid particles that can be separated from the solution.
General Formula:
AB (aq) + CD (aq) → AD (s) + CB (aq)
The solid (AD) is the precipitate, and it forms because the product is insoluble in water.
Key Features of a Precipitation Reaction
- Involves aqueous ionic compounds
- Produces an insoluble solid
- Occurs due to double displacement
- Can be predicted using solubility rules

Common Examples of Precipitation Reactions
1. Silver Nitrate and Sodium Chloride
AgNO₃ (aq) + NaCl (aq) → AgCl (s) + NaNO₃ (aq)
Silver chloride (AgCl) is the white precipitate that forms.
2. Barium Chloride and Sulfate
BaCl₂ (aq) + Na₂SO₄ (aq) → BaSO₄ (s) + 2NaCl (aq)
Barium sulfate (BaSO₄) is the insoluble precipitate.
3. Lead(II) Nitrate and Potassium Iodide
Pb(NO₃)₂ (aq) + 2KI (aq) → PbI₂ (s) + 2KNO₃ (aq)
Lead(II) iodide (PbI₂) forms a bright yellow precipitate.
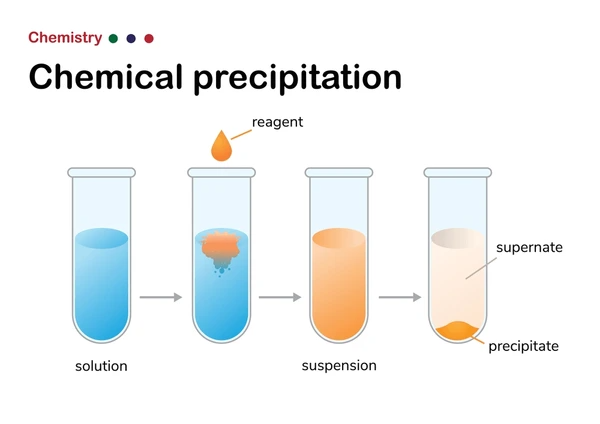
How to Identify a Precipitation Reaction
- Identify the reactants and products: Look at the chemical equation and determine what substances are being combined (reactants) and what new substance is formed (product).
- Check for a solid product: Precipitation reactions typically produce an insoluble solid, known as a precipitate. This solid will usually settle out of the solution and can be collected by filtration.
- Examine the solubility of the products: Use solubility rules to determine if the products are likely to form a precipitate. Generally, precipitation occurs when one of the products is insoluble in the solvent.
- Look for a change in appearance: During the reaction, you may notice a change in the solution’s appearance, such as the formation of a cloudy or milky substance, which indicates the presence of a precipitate.
- Perform a simple test: You can test for the presence of a precipitate by adding a few drops of a solution to a test tube containing the reaction mixture and observing any changes in appearance.
Example:
From AgNO₃ + NaCl → AgCl (s) + NaNO₃
Net ionic equation: Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s)
Solubility Rules Overview (Quick Guide)
- General Solubility Guidelines:
- Compounds of Group 1 elements (Li⁺, Na⁺, K⁺, Rb⁺, Cs⁺, and Fr⁺) or ammonium (NH₄⁺) are generally soluble in water.
- Nitrates (NO₃⁻), chlorates (ClO₃⁻), and perchlorates (ClO₄⁻) are usually soluble.
- Binary compounds of halogens (Cl⁻, Br⁻, or I⁻) with metals are generally soluble, except for fluoride (F⁻), silver (Ag⁺), lead (Pb²⁺), and mercury (Hg²⁺) salts.
- Solubility in Organic Solvents:
- Solubility can vary significantly depending on the solvent used. Polar solutes tend to dissolve better in polar solvents, while nonpolar solutes are more soluble in nonpolar solvents.
- The solubility of a substance is also influenced by temperature and pressure, with solubility generally increasing with heat, except for gases.
- Special Cases and Exceptions:
- Some compounds may dissolve through chemical reactions rather than simple dissolution. For example, zinc dissolves in hydrochloric acid by reacting to form zinc chloride.
- The solubility of a substance can be affected by the presence of other compounds. The common-ion effect, for instance, can decrease solubility.
| Ion/Compound | Solubility in Water |
|---|---|
| Nitrates (NO₃⁻) | Soluble |
| Alkali metal salts | Soluble |
| Halides (Cl⁻, Br⁻, I⁻) | Soluble (except with Ag⁺, Pb²⁺, Hg₂²⁺) |
| Sulfates (SO₄²⁻) | Soluble (except Ba²⁺, Pb²⁺, Ca²⁺) |
| Carbonates, phosphates | Insoluble (except with Na⁺, K⁺) |
| Hydroxides (OH⁻) | Insoluble (except with Ba²⁺, Ca²⁺, Sr²⁺) |
Real-Life Applications of Precipitation Reactions
Water Treatment and Purification
Water treatment facilities use precipitation reactions to remove impurities. For example, they treat hard water by converting calcium and magnesium ions into insoluble calcium carbonate or magnesium hydroxide, which they then filter out.
Industrial Processes
Industries apply precipitation reactions to separate and purify substances. Chemical manufacturers rely on this technique to eliminate impurities and ensure high product quality.
Environmental Monitoring
Scientists use precipitation reactions in environmental monitoring to detect specific ions in water samples. By adding selective reagents, they induce precipitate formation, which they analyze visually or through quantitative methods.
Medical Diagnostics
Medical professionals perform precipitation tests to identify antibodies in patient serum. When they add a specific antigen, it reacts with any corresponding antibody to form a visible precipitate.
Soil and Water Analysis
Agricultural and environmental scientists conduct soil and water analyses using precipitation reactions. These tests reveal the presence of key ions and compounds, helping them assess soil fertility and water quality.
Material Science
Researchers in material science use precipitation reactions to control how precipitates form within materials. These reactions influence material properties, helping scientists design substances with targeted performance traits.
Pharmaceuticals
Pharmaceutical manufacturers use precipitation reactions to isolate and purify active ingredients. This process ensures the safety, efficacy, and consistency of the final drug products.
Food Industry
Food technologists employ precipitation reactions in cheese production. They induce the precipitation of casein from milk, which forms curds essential for cheese making.
FAQs
A precipitate is an insoluble solid that forms when two soluble substances react in a solution.
They primarily happen in water, but similar principles can apply in other solvents if solubility limits are exceeded.
Not always. It could be suspended particles or emulsions. A true precipitate is formed through a chemical reaction and typically settles over time.
No. A precipitate forms only if one of the products is insoluble in the reaction medium.
Some can, under specific conditions, but generally, once a solid forms, it doesn’t readily redissolve unless chemically treated.
Conclusion
Precipitation reactions are a key concept in both classroom chemistry and real-world applications. Whether you’re filtering water or identifying ions in a lab, recognizing how and why a precipitate forms can help you understand broader principles of solubility, reactivity, and chemical change. With the help of solubility rules and ionic equations, these reactions become easy to identify and balance.
To get detailed scientific explanations of Precipitation Reaction, try Patsnap Eureka.


