
Introduction to the Diels-Alder Reaction
The Diels-Alder reaction ranks among organic chemistry’s most versatile tools, serving as the classic [4+2] cycloaddition. Otto Diels and Kurt Alder first reported this transformation in 1928, earning them the 1950 Nobel Prize in Chemistry. The reaction builds six-membered rings through concerted cycloaddition between a conjugated diene (electron-rich) and a dienophile (electron-deficient), making it indispensable for constructing complex molecular architectures in natural product synthesis and materials science.
Patsnap Eureka Q&A explains the Diels-Alder Reaction—a diene and dienophile combining into a six-membered ring with perfect precision—revolutionizing drug development, materials science, and synthetic chemistry!
This reaction’s exceptional value lies in its ability to create up to four stereocenters in one step with predictable stereoselectivity. It typically occurs under mild conditions without catalysts, though Lewis acids can accelerate the process. The reaction’s perfect atom economy aligns with modern green chemistry principles.
Detailed Reaction Mechanism
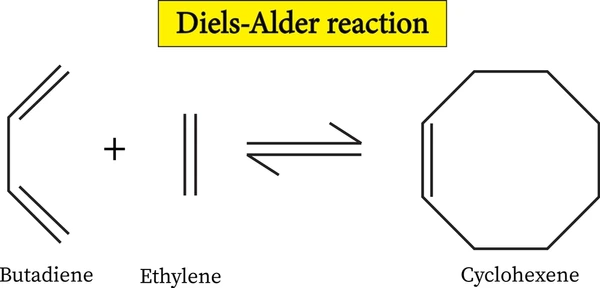
Mechanistic Details
The Diels-Alder reaction usually follows a concerted mechanism with a single transition state where new C-C bonds form simultaneously. However, certain conditions or substituents can favor a stepwise mechanism through diradical or zwitterionic intermediates.
Concerted Pathways
We classify concerted mechanisms as synchronous (bonds forming together) or asynchronous (sequential bond formation). The aromatic transition state explains the reaction’s high regioselectivity and stereoselectivity.
Stepwise Pathways
Some reactions proceed stepwise when substituent effects or catalysts stabilize intermediates. Lewis acid catalysts often promote this pathway by lowering activation energy.
Electronic Effects
Substituents on reactants significantly influence reaction rates by modifying frontier molecular orbital energies. Electron-donating groups on dienes and electron-withdrawing groups on dienophiles typically accelerate reactions.
Modern Insights
Quantum mechanical calculations and DFT studies now provide detailed understanding of electronic effects, charge transfer, and orbital interactions in these transformations. Researchers use these tools to predict reaction outcomes and design new Diels-Alder variants.

Expanded Scope of Diels-Alder Reactions
Catalytic and Enantioselective Variants
Chemists now design chiral catalysts to control Diels-Alder stereochemistry precisely. These systems produce optically active compounds efficiently, including vital antiviral agents.
Inverse Electron Demand Reactions
Researchers employ electron-rich dienophiles with electron-poor dienes for inverse demand variants. This approach yields unique cycloadducts under milder reaction conditions.
Natural Product Synthesis
The reaction constructs complex natural product frameworks efficiently. Its precise regio- and stereoselectivity builds intricate bioactive molecular architectures reliably.
Material Science Applications
Scientists functionalize carbon nanotubes and fullerenes using Diels-Alder chemistry. These modifications tailor nanomaterial properties for specific technological applications.
Biomedical Innovations
The reaction’s mild conditions enable safe biomolecule conjugation. Researchers leverage this for creating diagnostic tools and targeted therapeutics.
Green Chemistry Advances
New catalytic systems operate effectively in water. This eliminates organic solvents, reducing the reaction’s environmental footprint significantly.
Apllication of Diels-Alder Reactions
Polymer and Material Science
Chemists use Diels-Alder reactions to build precision polymers. These reactions create self-healing materials and smart hydrogels efficiently.
Chiral Synthesis Advances
Researchers developed chiral diamine catalysts for asymmetric Diels-Alder reactions. These enable efficient antiviral drug synthesis.
Natural Product Construction
The reaction assembles complex natural product frameworks. It forms six-membered rings with perfect stereocontrol.
Biomedical Innovations
Scientists apply Diels-Alder chemistry to drug delivery systems. It functionalizes biomaterials for tissue engineering.
Industrial Scale-Up
Engineers now adapt Diels-Alder reactions for agrochemical production. The process shows promise for large-scale applications.
Application Cases
| Product/Project | Technical Outcomes | Application Scenarios |
|---|---|---|
| MaDA Enzyme Peking University | Solves stereoselectivity and low yield issues in Diels-Alder reactions, enabling high-efficiency synthesis of complex natural products | Catalyzing reactions between non-natural dienes and chalcones in organic synthesis |
| Diels-Alder Hydrogels University of Regensburg | Enables preparation of functionalized surfaces and bioconjugation techniques without metal catalysts | Drug delivery systems and tissue engineering applications |
| Total Synthesis Platform The Scripps Research Institute | Facilitates complex molecule construction through programmed cascade sequences | Total synthesis of natural products and pharmaceuticals |
| Chemical Masking System Tagworks Pharmaceuticals BV | Enables selective chemical masking and unmasking in vitro using inverse electron-demand Diels-Alder reaction | Complex chemical reactions and biomolecule synthesis, particularly on solid supports |
| Masked Ketene Dienophiles University of Münster | Allows for Diels-Alder reactions in settings with functional group incompatibility | Complex organic synthesis and natural product synthesis |
Emerging Trends and Future Directions
Computational Breakthroughs
Researchers now harness DFT calculations to predict Diels-Alder outcomes precisely. These digital tools slash lab time while boosting success rates dramatically.
Bioorthogonal Chemistry Wins
Scientists exploit tetrazine-alkene reactions for live-cell imaging. This breakthrough enables real-time tracking of biological processes safely.
Green Chemistry Revolution
Biomass-derived furanics power sustainable Diels-Alder reactions. These eco-friendly processes create renewable polymers and specialty chemicals.
Why This Matters Today
The Diels-Alder reaction keeps transforming modern chemistry. Its perfect blend of efficiency and precision drives innovation across industries.
Cutting-Edge Applications
From drug discovery to smart materials, this reaction delivers. Researchers continue finding groundbreaking uses in unexpected fields.
The Future Looks Bright
New catalytic systems and AI-powered design tools promise exciting advances. This century-old reaction still holds untapped potential.
Key Takeaways
- Computational tools predict reactions with pinpoint accuracy
- Bioorthogonal chemistry enables breakthrough medical imaging
- Sustainable versions use plant-based starting materials
- Versatility ensures ongoing relevance across industries
- Continuous innovation expands applications
Conclusion
The Diels-Alder reaction remains one of organic chemistry’s most powerful transformations nearly a century after its discovery. Its unique combination of efficiency, selectivity, and versatility continues to inspire new applications across chemical synthesis, materials science, and biological chemistry. As this overview demonstrates, ongoing innovations in catalytic systems, computational design, and sustainable applications ensure that this classic reaction will remain at the forefront of synthetic methodology for years to come. From its fundamental orbital-controlled mechanism to its cutting-edge applications in biomedicine and green chemistry, the Diels-Alder reaction exemplifies how deep understanding of fundamental chemical principles can lead to transformative practical applications.
To get detailed scientific explanations of Diels-Alder Reaction , try Patsnap Eureka.


