
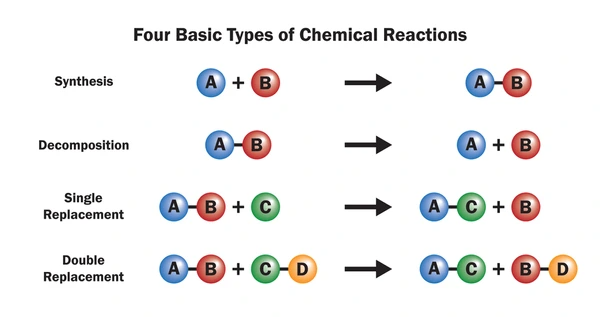
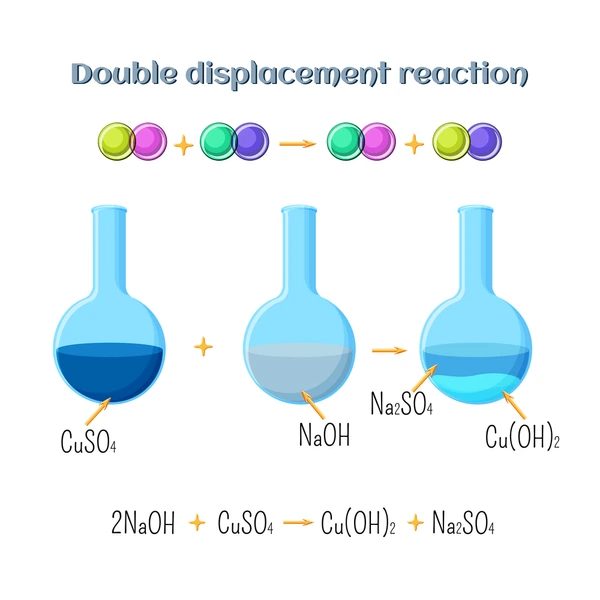
A double replacement reaction, also known as a double displacement or metathesis reaction, is a common type of chemical reaction that occurs between two ionic compounds in solution. During this process, the positive and negative ions of two different compounds exchange places, forming two new products. These reactions are frequently observed in precipitation, neutralization, and gas evolution processes.
This article explains the definition, general formula, reaction types, and real-world examples of double replacement reactions, along with tips on identifying and balancing them.
Definition of a Double Replacement Reaction
What is a limiting reactant? Eureka Technical Q&A explains that it’s the substance in a chemical reaction that runs out first, determining the maximum amount of product that can be formed.
A double replacement reaction involves two reactants (usually ionic compounds dissolved in water) that swap ions to form two new compounds. These reactions typically occur in aqueous solutions, and at least one of the products is often a precipitate, gas, or water.
General Reaction Format:
AB + CD → AD + CB
- A and C are cations (positive ions)
- B and D are anions (negative ions)
- The cations exchange their anions to form new compounds
Types of Double Replacement Reactions
1. Precipitation Reactions
These occur when an insoluble solid (precipitate) forms as one of the products.
Example:
Na₂SO₄ (aq) + BaCl₂ (aq) → BaSO₄ (s) + 2 NaCl (aq)
BaSO₄ is insoluble in water and forms a solid precipitate.
2. Neutralization Reactions
This type happens when an acid reacts with a base, producing water and a salt.
Example:
HCl (aq) + NaOH (aq) → NaCl (aq) + H₂O (l)
The hydrogen ion from the acid reacts with the hydroxide ion from the base to form water.
3. Gas Formation Reactions
Some double replacement reactions produce a gas, which bubbles out of the solution.
Example:
Na₂CO₃ (aq) + HCl (aq) → CO₂ (g) + H₂O (l) + NaCl (aq)
Carbon dioxide gas is released in this reaction.
How to Identify a Double Replacement Reaction
Examine the Chemical Equation
Start by checking if the equation follows the pattern AB + CD → AD + CB. This pattern indicates ion exchange.
Check for Two Reactants and Two Products
Next, confirm the reaction has two reactants and two products. This balance strongly suggests a double replacement reaction.
Identify Ion Exchange
Then, look for cations and anions switching partners. This ion exchange confirms the reaction follows the double replacement mechanism.
Consider the Types of Compounds Involved
Most often, this reaction type involves ionic compounds. These compounds allow ions to separate and recombine freely.
Look for Precipitate Formation
Also, check if a solid forms during the reaction. Precipitate formation is a classic sign of a double replacement reaction.
Check for Gas Evolution
Sometimes, the reaction produces gas like hydrogen or chlorine. Gas evolution can also signal a double replacement process.
Tip: Use a solubility chart to predict whether a product is insoluble and forms a precipitate.
Balancing a Double Replacement Reaction
- Write correct formulas for all reactants and products
- Use coefficients to balance the number of atoms of each element on both sides
- Confirm that the total charge is balanced for ionic compounds
Example:
AgNO₃ (aq) + NaCl (aq) → AgCl (s) + NaNO₃ (aq)
This reaction is already balanced as written.
Real-Life Applications
Water Softening
Hard water contains calcium and magnesium ions that reduce soap effectiveness. Adding sodium compounds replaces them, forming removable precipitates.
Medicine
Pharmaceutical manufacturers use double replacement reactions to synthesize compounds. Neutralization between acids and bases forms salts used in medications.
Industrial Processes
Industries use double replacement reactions to create fertilizers, dyes, and pigments. For example, sodium nitrate reacts with ammonium chloride to form ammonium nitrate.
Environmental Remediation
These reactions help treat pollution. Sodium hydroxide reacts with sulfuric acid to produce sodium sulfate, neutralizing acidic wastewater effectively.
Food Industry
Baking relies on double replacement reactions. Sodium bicarbonate reacts with citric acid to release carbon dioxide, making dough rise.
Application Cases
| Product/Project | Technical Outcomes | Application Scenarios |
|---|---|---|
| Pyramid™ Fragment Screening Platform Astex Therapeutics Ltd. | Enables rapid and cost-effective ligand identification without purification or characterization using X-ray crystallography | Drug discovery and development, particularly in identifying ligands for target macromolecules |
| PyriSynth™ Huaqiao University | Achieves efficient synthesis of multi-substituted pyridine derivatives with mild reaction conditions and high yields | Pharmaceutical industry, particularly in the synthesis of pyridine-based drug compounds |
| OxaPrep™ Sunshine Lake Pharma Co., Ltd. | Achieves efficient and safe preparation of oxazolidinone compounds using copper catalysts and mixed solvents, suitable for industrial production | Pharmaceutical manufacturing, especially in the production of oxazolidinone-based antibiotics |
| CycloPenta-Pro™ China Petroleum & Chemical Corp. | Achieves high selectivity and conversion rate in synthesizing multi-substituted cyclopentadienes and substituted indenes using Grignard reagents | Petrochemical industry, particularly in the production of specialty chemicals and advanced materials |
| EnolAce™ Nippon Soda Co., Ltd. | Achieves high yields and retains enolic double bond configuration in the production of enol O-acyl compounds | Agrochemical and pharmaceutical industries, specifically in the synthesis of intermediates for pesticides and drugs |
FAQs
In a single replacement reaction, one element replaces another in a compound. In a double replacement reaction, two compounds exchange ions.
Not all. Some produce water (as in neutralization) or gas, but at least one product must leave the solution in some form.
These reactions typically occur in aqueous solutions, where ions are free to move and react.
Common signs include formation of a solid, gas bubbles, or heat release when neutralization happens.
Conclusion
A double replacement reaction is a key concept in chemistry where two ionic compounds exchange ions to form new products. These reactions are vital in many biological, environmental, and industrial processes. By recognizing the structure and outcomes of double replacement reactions, students and professionals can better understand how substances interact in aqueous environments.
How PatSnap Eureka Accelerates Innovation in Double Replacement Reactions
The double replacement reaction is a key mechanism in inorganic and analytical chemistry, with wide applications in material synthesis, environmental remediation, and industrial processing. PatSnap Eureka helps researchers uncover how this reaction type is being applied and refined across the innovation landscape.
- Patent Intelligence: Eureka scans global patent databases to identify developments involving double replacement reactions, including novel compound synthesis, precipitation techniques, and ion-exchange processes.
- Competitive Landscape Tracking: Explore how companies and research institutions utilize double replacement strategies in sectors like water treatment, battery chemistry, and pharmaceuticals.
- Trend Forecasting: Eureka’s AI-powered analytics highlight growing use cases of double replacement reactions in sustainable materials, diagnostics, and clean energy technologies.
- Technical Clustering: Visual clustering reveals R&D hotspots focused on optimizing double replacement mechanisms, such as reaction condition tuning, selectivity control, and solid-state applications.
With PatSnap Eureka, researchers can accelerate discovery and make informed decisions by uncovering the latest insights in double replacement reaction innovation worldwide.
To get detailed scientific explanations of Double Replacement Reaction, try Patsnap Eureka.


