Phenyl benzylamine compound and application thereof
A technology of phenylbenzylamine and compounds, applied in the field of biopharmaceuticals, can solve the problems of high toxicity and side effects, high treatment costs, and poor curative effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0081]
[0082] Preparation of N-benzyl-4-methyl-o-nitroaniline (II)
[0083] Add 4-methyl-o-nitroaniline (200g, 1.32mol) to the four-necked bottle, K 2 CO 3 (20g, 0.5mol) and benzyl bromide (200ml), DMF (1.2ml) were slowly added dropwise to the reaction solution. Raise the temperature to 190°C to 200°C. The degree of reaction was detected by TLC (developing solvent: PE:EA=1:9). The reaction product was poured into distilled water, and a solid precipitated out. Suction filtration, filtrate with CH 2 Cl 2 Extract 3 (200ml*3) times, combine CH 2 Cl 2 layer and suction filtered the resulting solid. It was washed once more with water, dried and concentrated. Finally, the pure product was obtained by recrystallization from ethyl acetate with a yield of 58%.
[0084] Preparation of N1-benzyl-4-methyl-o-phenylenediamine (III)
[0085] Add N-benzyl-4-methyl-o-nitroaniline (10g, 0.04mol) in a 500ml eggplant-shaped bottle, SnCl 2 .2H 2 O (95g, 0.44mol), ethanol (80ml), s...
Embodiment 2
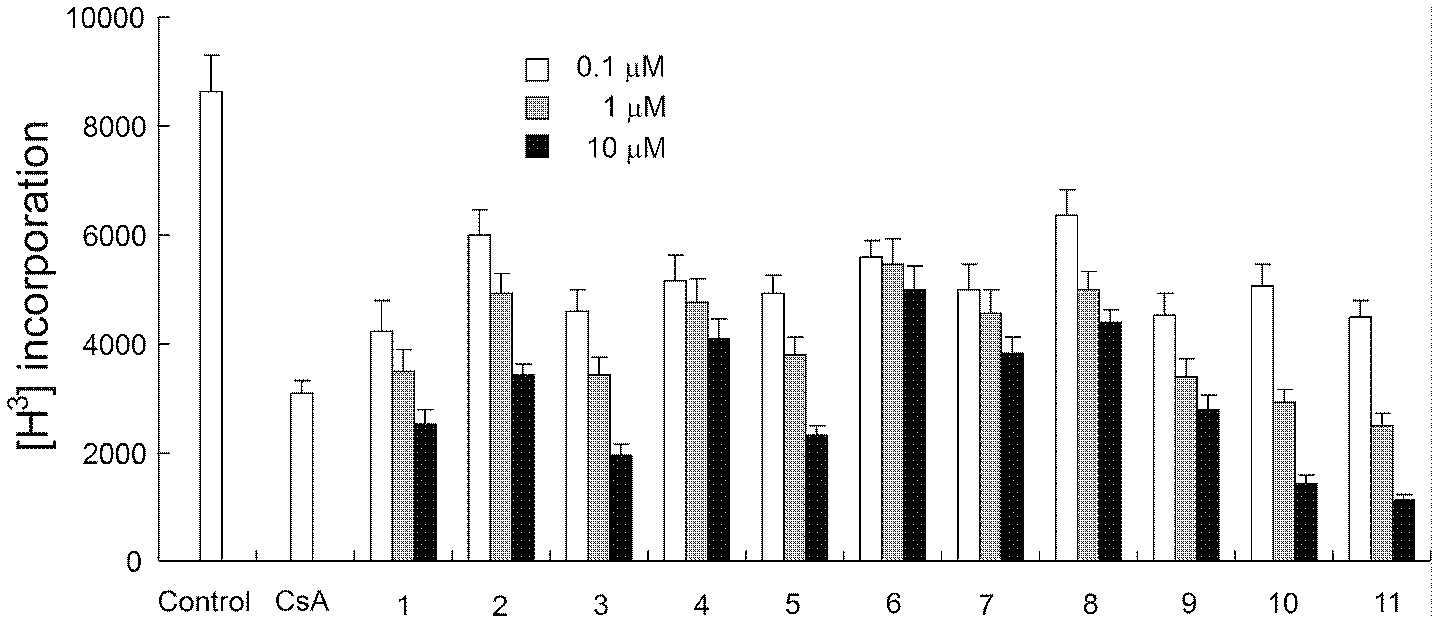
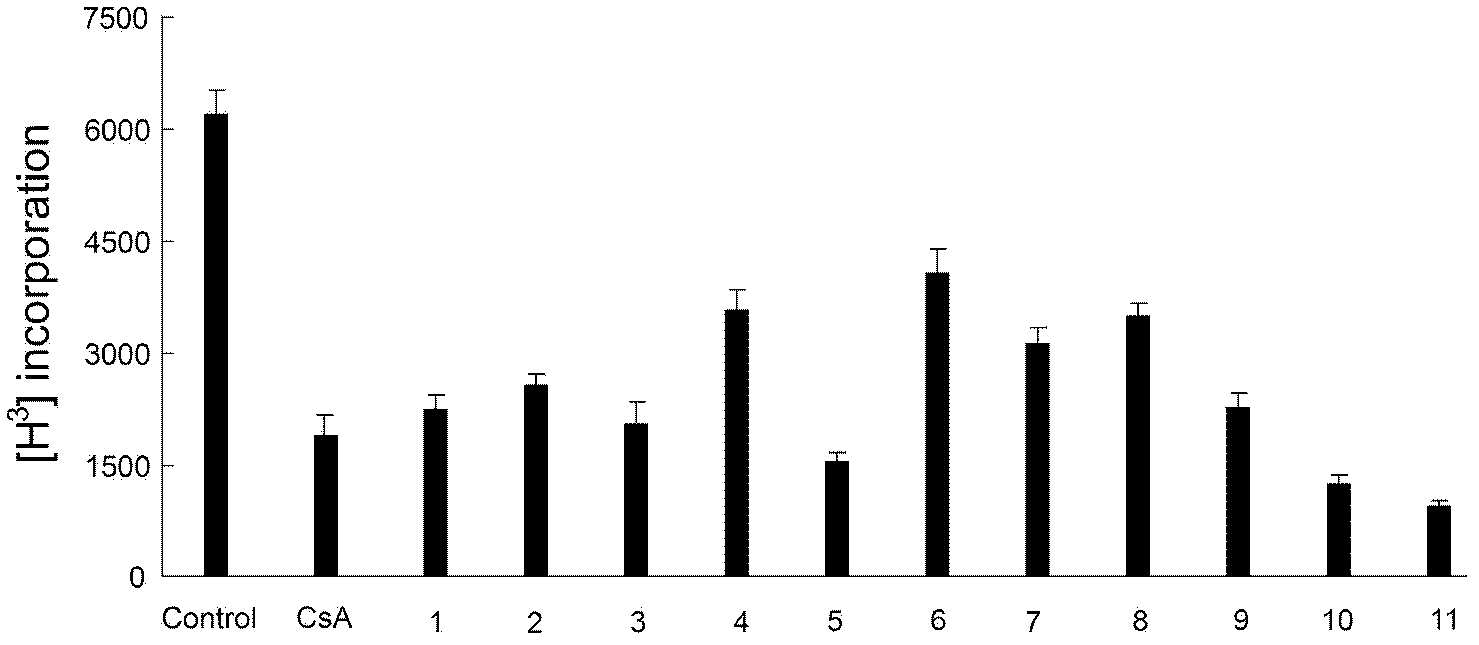
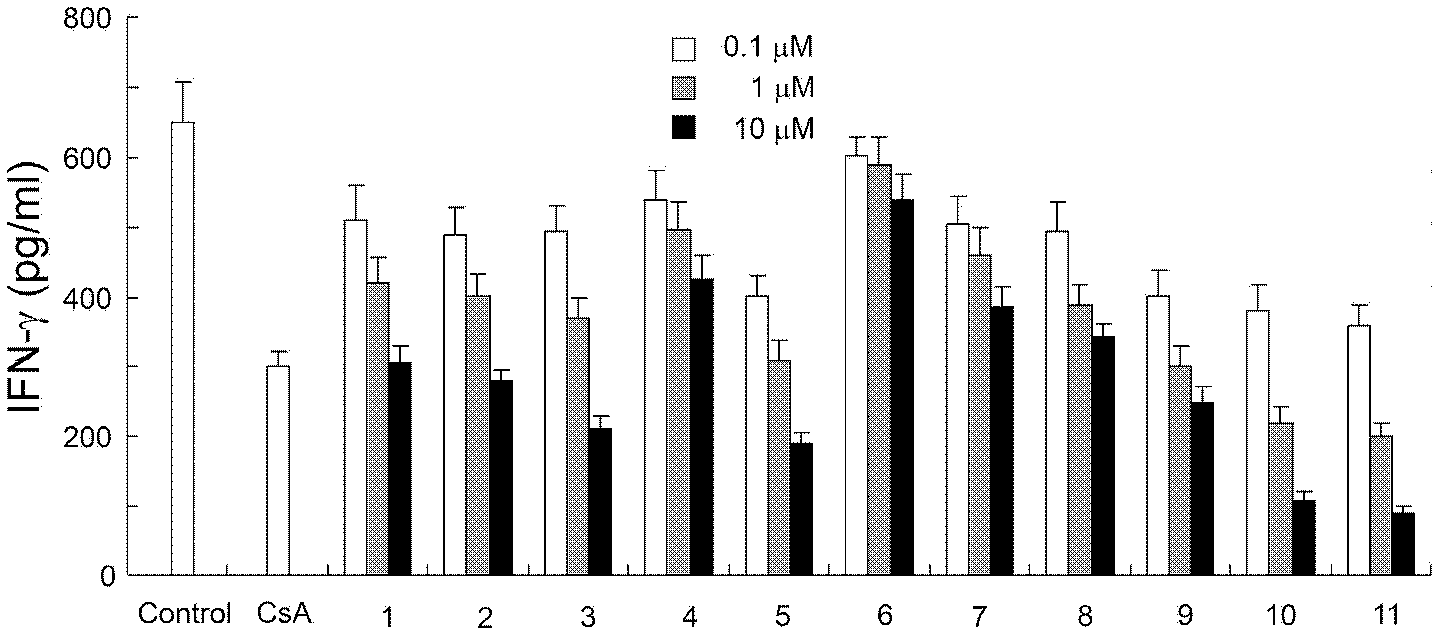
[0182] 1. Pharmacological tests of phenylbenzylamine compounds
[0183] 1) Proliferation test of mouse T cells activated by anti-CD3 / anti-CD28
[0184] Four lymph nodes in the groin and armpit of the mice were taken to prepare a single cell suspension. Make 3×10 with complete medium RPMI-1640 6 / ml of cell suspension, seeded in 96-well plate, 3×10 5 Cells per well were stimulated with a final concentration of 10 μg / ml anti-CD3 and 1 μg / ml anti-CD28 (purchased from BD PharMingen) for 72 hours, incubated with compounds, and the proliferation of lymphocytes was detected by isotope tritium labeling.
[0185] 2) MOG 35-55 T-lymphocyte proliferation and cytokine release test in EAE-induced model mice
[0186] Take MOG 35-55 (purchased from Shanghai Qiangyao Biotechnology Co., Ltd.) and complete Freund's adjuvant (purchased from sigma company) emulsion sensitized for 10 days, four lymph nodes in the inguinal and axilla of mice were prepared into a single cell suspension. Make 3...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com