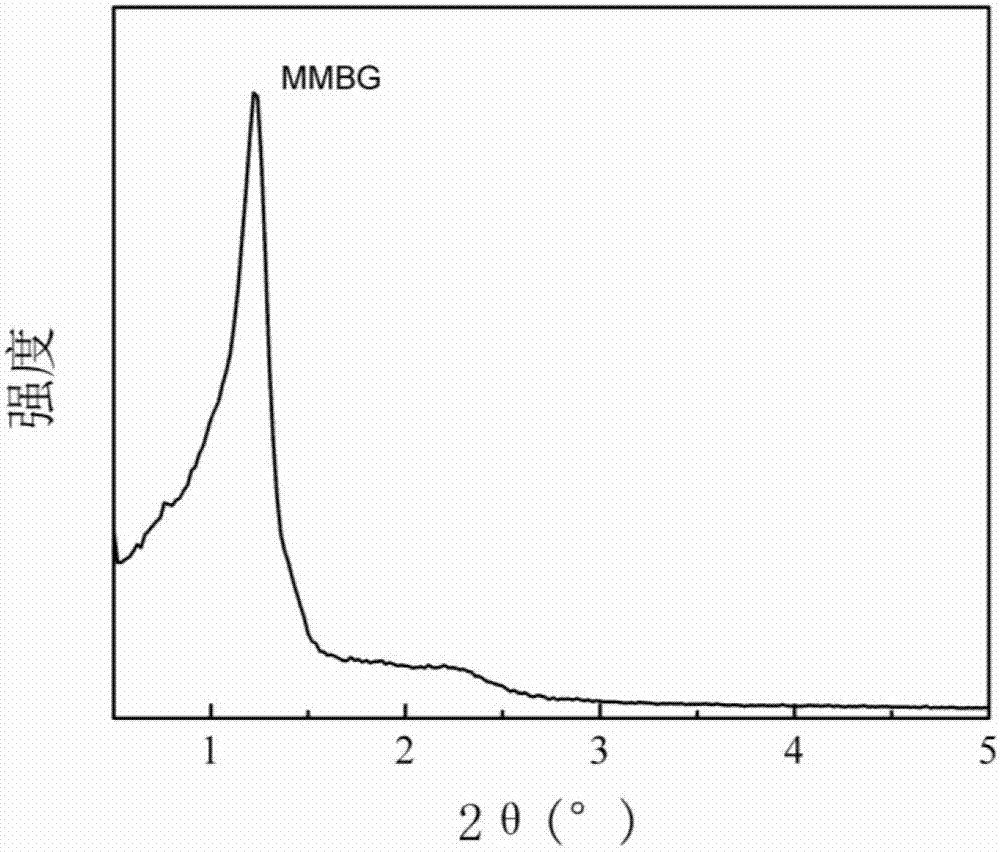
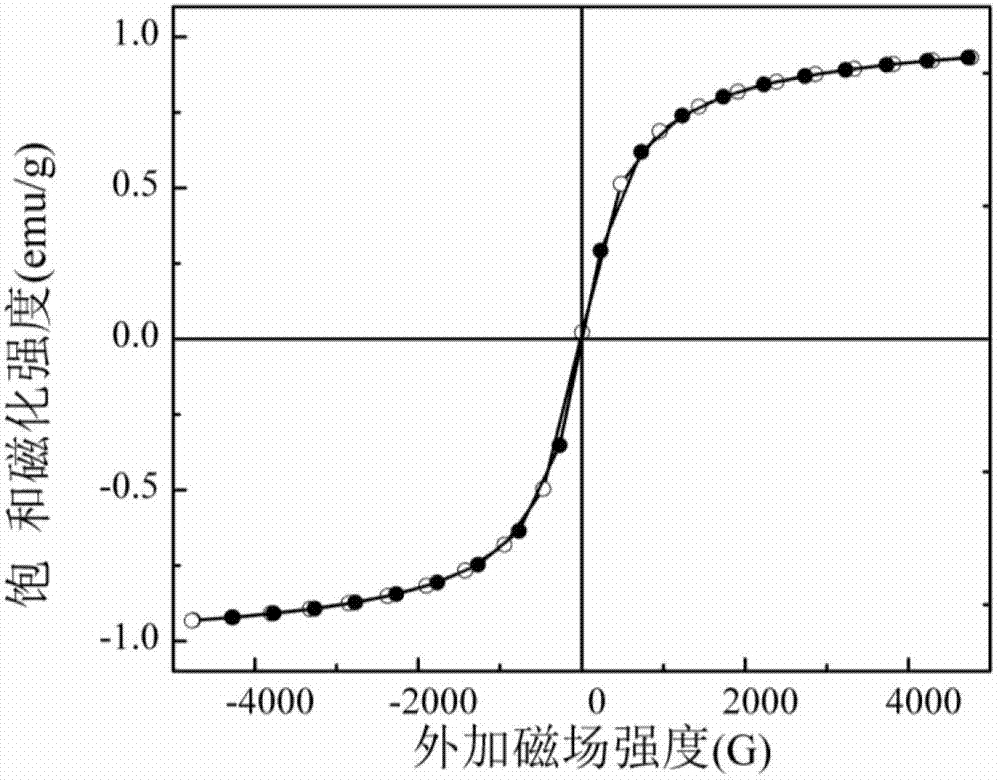
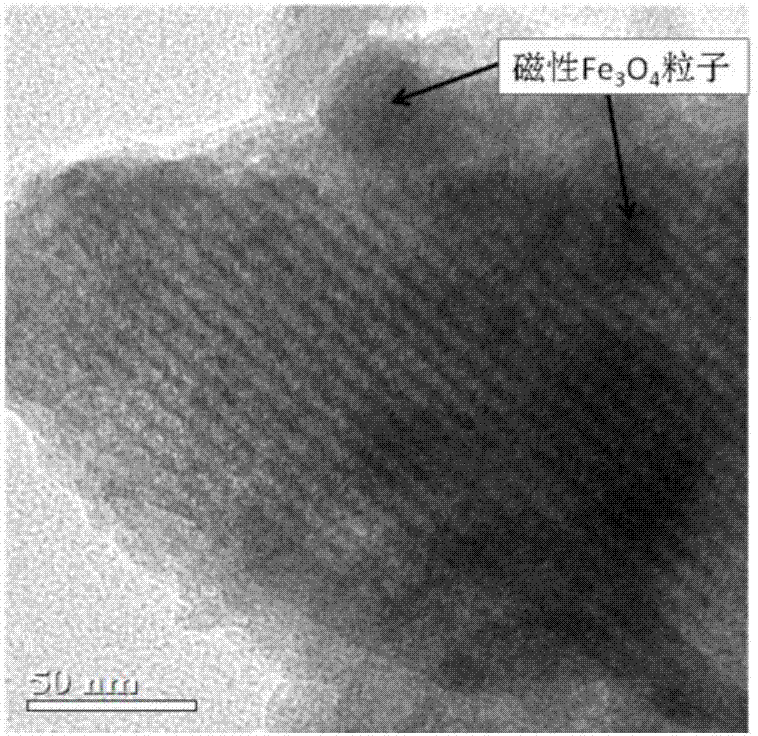
Magnetic mesoporous bioactive glass drug delivery system and preparation method thereof
A bioglass and delivery system technology, applied in medical science, prosthesis, inorganic non-active ingredients, etc., can solve the problems of increased cytotoxicity and biocompatibility, and achieve increased drug loading, excellent drug loading and sustained release performance , the effect of simple equipment
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
[0024] Example 1-6
[0025] First place 4g of P123 in 50mL of ethanol and stir vigorously to obtain a homogeneous ethanol dispersion solution of surfactant; then prepare inorganic precursors to obtain SiO as shown in Table 1. 2 :CaO:P 2 O 5 :Fe 3 O 4 In the magnetic mesoporous bioglass drug delivery system with a mass ratio of, the inorganic substances are added in order, and the first group of embodiments are specifically: 1.25g calcium nitrate tetrahydrate, 8.6mL (8g) tetraethylorthosilicate (TEOS) , 0.35mL (0.37g) triethyl phosphate (TEP), 0.142g nano Fe 3 O 4 Particles, then add 1-3mL dilute HNO 3 (1mol / L) The solution is catalyzed and hydrolyzed at a reaction temperature of 40℃; after the inorganic precursor is hydrolyzed and dispersed uniformly, the surfactant ethanol dispersion solution is added, the reaction is fully mixed, and the soft template is sealed and self-assembled into a sol-gel. The product is 60 Drying in an oven at ℃ for 1-3 days, calcining the gel obtained af...
Example Embodiment
[0028] Example 7
[0029] According to the steps in Example 1, no magnetic nanoparticles were added, and other processes and conditions were the same as in Example 1. Finally, a mesoporous bioglass drug delivery system without magnetism was obtained, which was a blank control group.
Example Embodiment
[0030] Example 8
[0031] The magnetic mesoporous bioglass drug delivery system prepared in Example 1 was selected for drug loading and sustained release experiments.
[0032] (1) Drug load: weigh 30 mg of powdered gentamicin sulfate under the protection of inert gas in a glove box, and dissolve it in 50 mL of deionized water to prepare a 600 μg / mL gentamicin sulfate solution; take 0.5 g of sample and place it in the solution Stir vigorously for 24h at a constant temperature at 37°C. Centrifuge at 8000r / min, dry the lower sediment in a vacuum drying oven at 37°C, and leave the upper sediment for drug load testing.
[0033] (2) Drug release: Weigh 0.2g of the drug-loaded sample, press it into a sheet with a diameter of 2-3 cm with a tablet press, and soak the sample in 100 mL of deionized water. At different time points, use UV- The visible spectrophotometer detects the concentration of the drug in the solution. The drug release curve is as Figure 4 Shown.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Specific surface area | aaaaa | aaaaa |
| Mesopore diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap