Method for establishing fritillary bulb medicinal material alkaloids fingerprint
A technology of fingerprints and establishment methods, which is applied in the field of establishment of high-performance liquid chromatography fingerprints of Fritillaria chinensis medicinal materials, can solve the problems of incomplete reflection of the overall characteristics of medicinal materials, difficulty in multi-factor detection limitations, and low content of fritillaria. problem, to achieve the effect of simple identification method, ensuring accuracy and good separation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
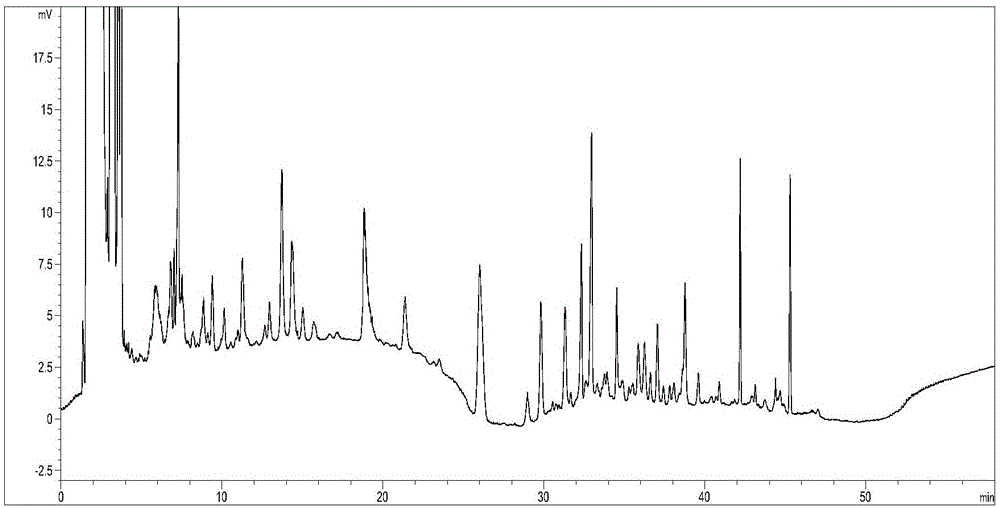
[0058] Example 1: Establishing the fingerprint of alkaloids in Fritillaria sibiricus medicinal material
[0059] 1. Instruments, reagents and test samples
[0060] Instrument: high performance liquid chromatograph: Aglient1200, evaporative light diffuser: Alltech 3300;
[0061] Electronic balance: Model: CP214, manufactured by Ohaus Instrument Shanghai Co., Ltd.;
[0062] Reference substance: Peimusin reference substance (China Food and Drug Control Study, batch number: 11892-201201); Peimusu A reference substance (China Food and Drug Control Study, content 98.3%, batch number: 110750-201110); Peimousin B reference substance (China Food and Drug Control Study, content is 99.9%, batch number: 110751-201110); Sibeimine reference substance (China Food and Drug Control Study, content is 99.1%, batch number: 110767-201107).
[0063] Sample: Ping Fritillaria medicinal materials (supplied by the Purchasing Department of Wuzhou Pharmaceutical Co., Ltd., batch numbers: 13082101, 13082102, 13082...
Example Embodiment
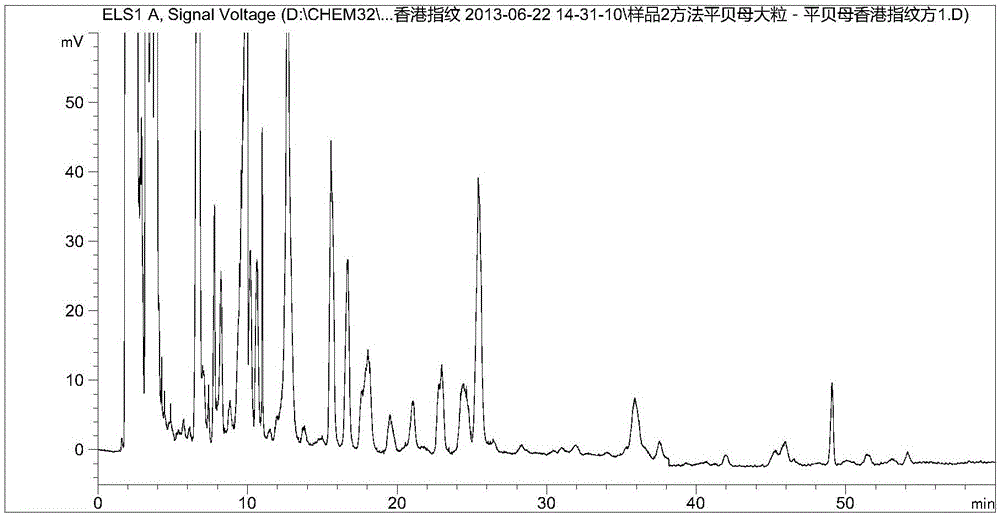
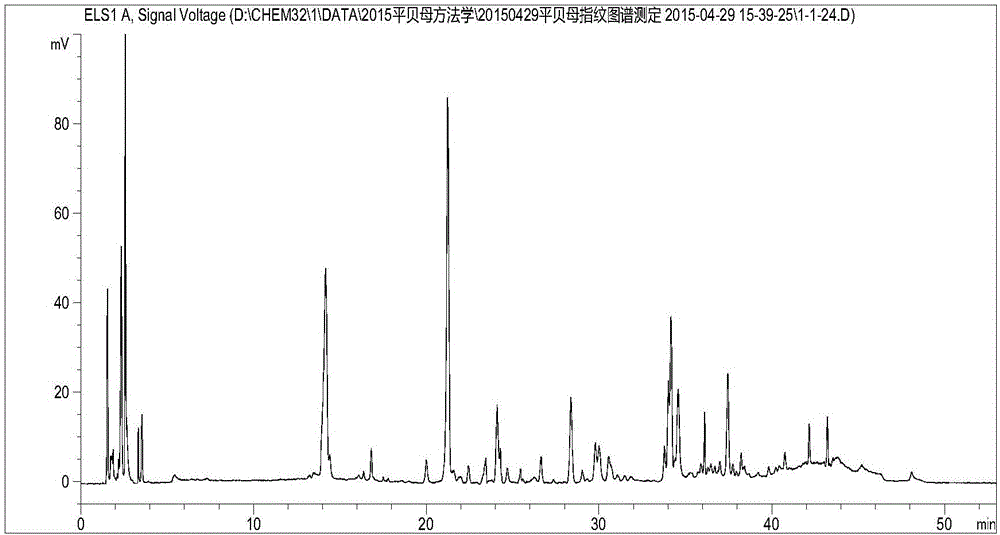
[0126] Example 2: Establishment of HPLC characteristic profile of Fritillaria sibiricus:
[0127] Fritillaria medicinal materials are provided by the purchasing department of Guangxi Wuzhou Pharmaceutical (Group) Co., Ltd.
[0128] According to high-performance liquid chromatography ("Chinese Pharmacopoeia" 2010 edition an appendix VID) determination:
[0129] Use octadecylsilane-bonded silica gel as filler, mobile phase A is 0.1% triethylamine (containing 1mmol / L ammonium acetate) aqueous solution, mobile phase B is acetonitrile solution; flow rate is 1.0ml / min, using gradient Elution, the elution procedure is as follows,
[0130] 0~8min, mobile phase A is 70%~65%, mobile phase B is 30%~35%;
[0131] 8-25min, mobile phase A is 65%-55%, mobile phase B is 35%-45%;
[0132] 25~40min, mobile phase A is 55%~5%, mobile phase B is 45%~95%;
[0133] 40~43min, mobile phase A is 5%, mobile phase B is 95%;
[0134] 43~48min, mobile phase A is 5%~70%, mobile phase B is 95%~30%
[0135] 48~58min, mobi...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Mobile phase a | aaaaa | aaaaa |
| Mobile phase a | aaaaa | aaaaa |
| Mobile phase a | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap