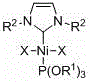Mixed nickel (II) complex based on phosphite and unsaturated n-heterocyclic carbene as well as preparation method and application thereof
A nitrogen-heterocyclic carbene and phosphite technology, which is applied in the field of nickel complexes and their organic synthesis, can solve the problems of no hydrogenation addition reaction, etc., and achieve the effect of improving operability, low toxicity, and facilitating coordination
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0028] Embodiment one: Ni[P(OR 1 ) 3 ][(R 2 NCHCHNR 2 )C]X 2 (R 1 = ethyl, R 2 =2,4,6-trimethylphenyl, X=bromine) synthesis
[0029] The unsaturated nitrogen heterocyclic carbene (R 2 NCHCHNR 2 )C (0.3042g, 1.0 mmol) was added into a tetrahydrofuran solution of bis(triethylphosphite)nickel(II) bromide (0.5500 g, 1.0 mmol), reacted at room temperature for 2 hours, and removed the solvent in vacuo, Wash the residue with n-hexane, extract the residue with toluene, transfer the clear liquid and remove the solvent toluene to obtain a red solid with a yield of 68%.
[0030] The product was subjected to elemental analysis, and the results are shown in Table 1:
[0031] Table 1 Elemental analysis results
[0032] C:(%) H:(%) N:(%) theoretical value 46.99 5.84 4.06 actual value 47.16 5.93 4.01
[0033] The product was characterized by NMR, and the results are as follows:
[0034] Dissolve the product in C 6 D. 6 Medium (about 0.4 mL), seal...
Embodiment 2
[0035] Embodiment two: Ni[P(OR 1 ) 3 ][(R 2 NCHCHNR 2 )C]X 2 (R 1 = isopropyl, R 2 = 2,4,6-trimethylphenyl, X = bromine) synthesis
[0036] The unsaturated nitrogen heterocyclic carbene (R 2 NCHCHNR 2 )C (0.3042g, 1.0 mmol) was added to a tetrahydrofuran solution of bis(triisopropyl phosphite)nickel(II) bromide (0.6350 g, 1.0 mmol), reacted at room temperature for 3 hours, and removed the solvent in vacuo , wash the residue with n-hexane, extract the residue with toluene, transfer the clear liquid and remove the solvent toluene to obtain a red solid with a yield of 72%.
[0037] Product is carried out elemental analysis, and the result is as shown in table 2:
[0038] Table 2 Elemental analysis results
[0039] C:(%) H:(%) N:(%) theoretical value 49.21 6.33 3.83 actual value 49.43 6.39 3.72
[0040] The product was characterized by NMR, and the results are as follows:
[0041] Dissolve the product in C 6 D. 6 Medium (about 0.4 mL)...
Embodiment 3
[0042] Embodiment three: Ni[P(OR 1 ) 3 ][(R 2 NCHCHNR 2 )C]X 2 (R 1 = ethyl, R 2 =2,6-diisopropylphenyl, X=bromine) synthesis
[0043] The unsaturated nitrogen heterocyclic carbene (R 2 NCHCHNR 2 )C (0.3883g, 1.0 mmol) was added into a tetrahydrofuran solution of bis(triethylphosphite)nickel(II) bromide (0.5500 g, 1.0 mmol), reacted at room temperature for 1 hour, and removed the solvent in vacuo, Wash the residue with n-hexane, extract the residue with toluene, transfer the clear liquid and remove the solvent toluene to obtain a red solid with a yield of 73%.
[0044] Product is carried out elemental analysis, and the result is as shown in table 3:
[0045] Table 3 Elemental analysis results
[0046] C:(%) H:(%) N:(%) theoretical value 51.19 6.77 3.62 actual value 51.31 6.80 3.57
[0047] The product was characterized by NMR, and the results are as follows:
[0048] Dissolve the product in C 6 D. 6 Medium (about 0.4 mL), seal the...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com