Primer probe set, kit and detection method for multiple detection of human parainfluenza virus subtypes based on fluorescence RMA method
A human parainfluenza virus, multiple detection technology, applied in biochemical equipment and methods, DNA/RNA fragments, recombinant DNA technology, etc., can solve the problems of complex operation procedures, inability to detect parainfluenza virus, and many false negatives
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
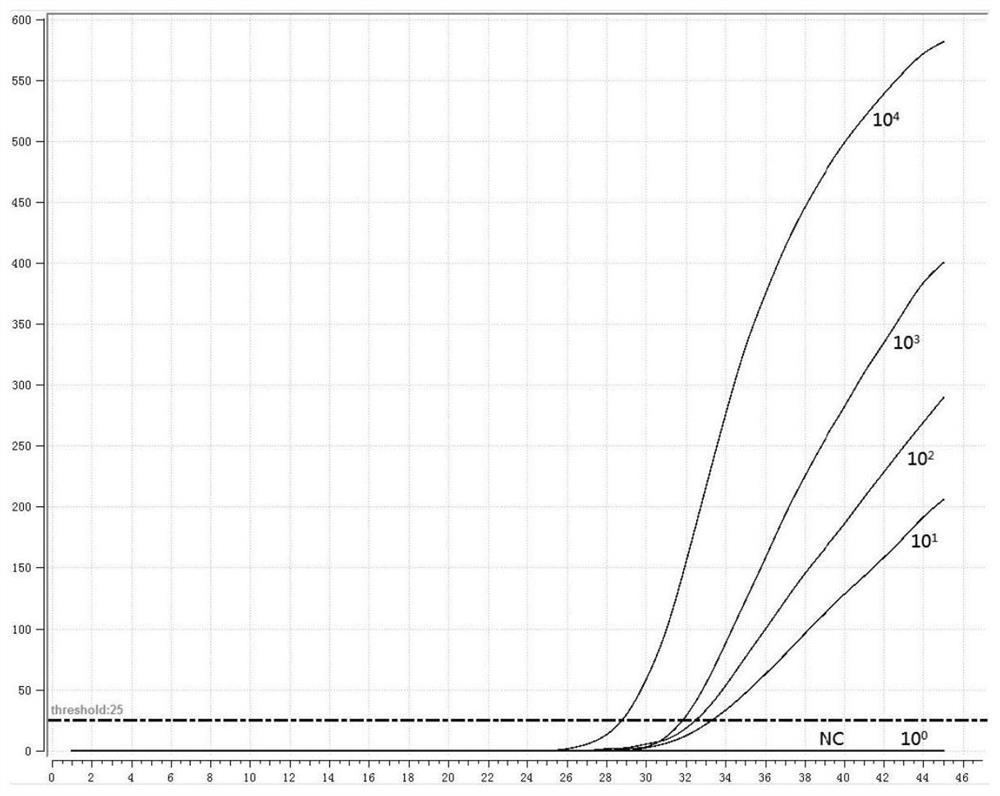
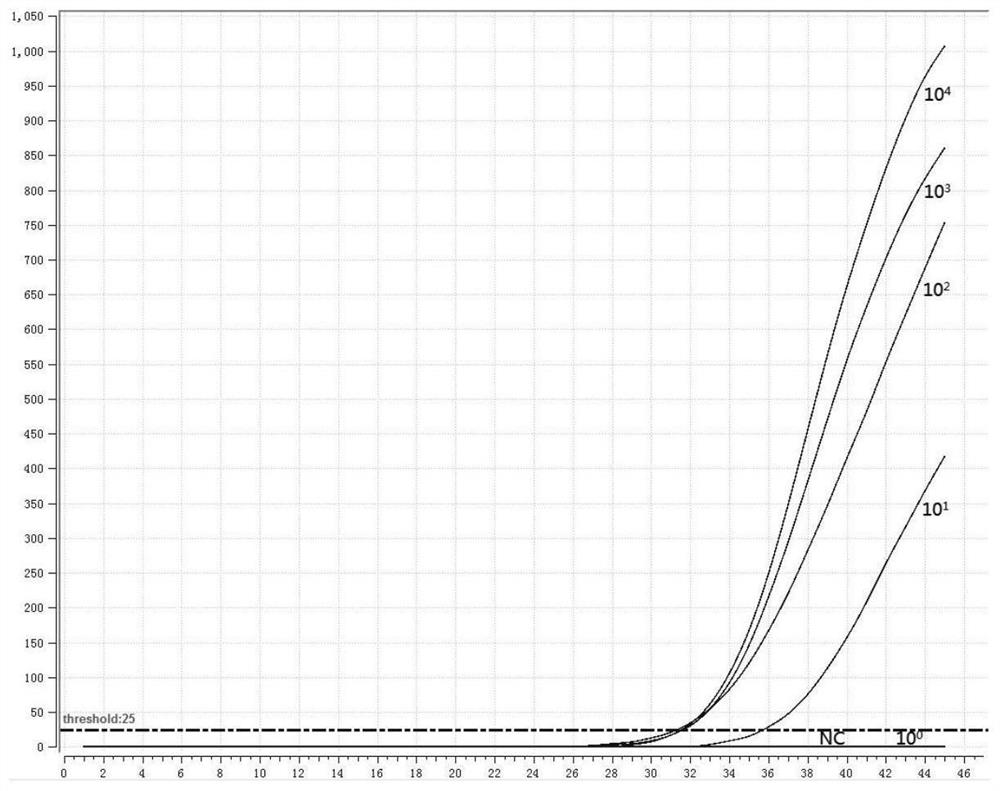
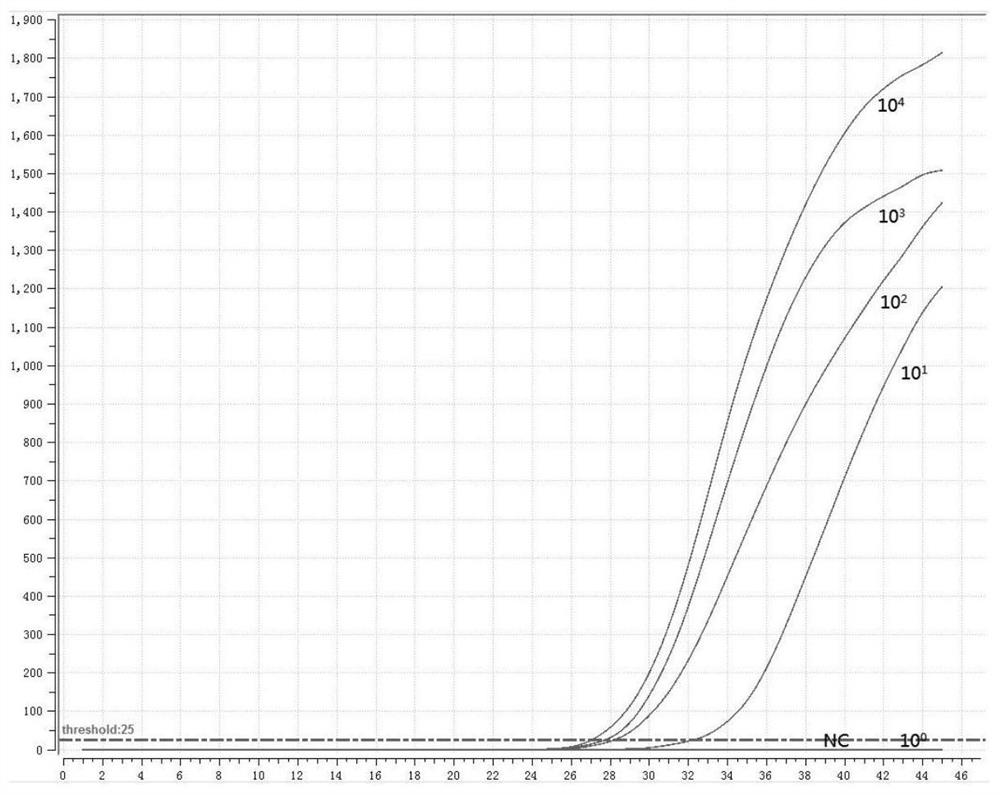
[0051] 1. Preparation of positive standard plasmid
[0052] According to the instructions of the viral DNA / RNA extraction kit, extract the RNA of the three subtypes of human parainfluenza virus, and perform reverse transcription according to the instructions of the reverse transcription kit to obtain cDNA; use the cDNA as a template to perform PCR amplification on the HN gene, The PCR amplified product was electrophoresed on 1% agarose gel, recovered by tapping the gel, cloned and connected to the pMD18-T vector, transformed into Escherichia coli competent cells, screened by blue and white spots, picked white colonies, and carried out colony PCR verification. Send the positive recombinant bacteria to the company for sequencing, culture the correctly sequenced recombinant bacteria overnight, extract plasmid DNA, and obtain positive plasmids (P-1, P-2, P-3).
[0053] 2. Design of fluorescent RMA primers and probes
[0054] Fluorescent RMA primers and probes were designed for th...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com