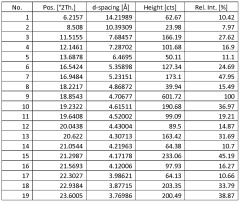
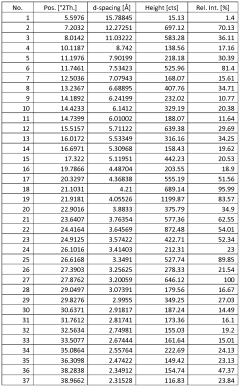
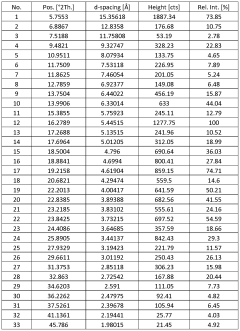
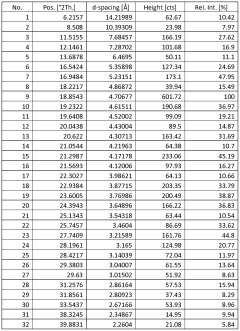
Characterize Sodium Nitrate Crystalline Structure by X-Ray Analysis
MAR 3, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sodium Nitrate XRD Background and Research Objectives
Sodium nitrate (NaNO₃) represents a fundamental ionic compound that has garnered significant attention in crystallographic research due to its unique structural properties and phase transition behaviors. This compound exhibits a complex crystalline structure that undergoes temperature-dependent polymorphic transformations, making it an ideal candidate for advanced X-ray diffraction analysis. The compound's ability to exist in multiple crystalline phases under different environmental conditions presents both opportunities and challenges for comprehensive structural characterization.
X-ray diffraction has emerged as the gold standard for crystalline structure determination, offering unparalleled precision in identifying atomic arrangements, lattice parameters, and symmetry operations within crystalline materials. The technique's non-destructive nature and high resolution make it particularly suitable for investigating sodium nitrate's intricate structural features, including its rhombohedral crystal system and space group characteristics.
Historical investigations into sodium nitrate's crystalline structure date back to early crystallographic studies in the mid-20th century, where researchers first identified its room-temperature phase as belonging to the R3c space group. However, recent advances in X-ray diffraction instrumentation and computational analysis methods have opened new avenues for more detailed structural investigations, revealing previously undetected structural nuances and phase transition mechanisms.
The primary research objective centers on achieving comprehensive characterization of sodium nitrate's crystalline structure through systematic X-ray analysis, encompassing both static structural determination and dynamic phase behavior investigation. This involves precise measurement of lattice parameters, atomic coordinates, thermal displacement parameters, and electron density distributions within the crystal lattice.
Secondary objectives include establishing correlations between structural parameters and macroscopic properties, investigating temperature-induced phase transitions, and developing standardized protocols for sodium nitrate structural analysis. These goals aim to enhance understanding of structure-property relationships and provide foundational data for applications in materials science, pharmaceutical crystallography, and industrial process optimization.
The research framework emphasizes integration of high-resolution powder diffraction, single-crystal analysis, and variable-temperature studies to construct a comprehensive structural profile. This multi-faceted approach ensures robust characterization while addressing the compound's polymorphic complexity and environmental sensitivity, ultimately contributing to the broader field of ionic crystal structure determination.
X-ray diffraction has emerged as the gold standard for crystalline structure determination, offering unparalleled precision in identifying atomic arrangements, lattice parameters, and symmetry operations within crystalline materials. The technique's non-destructive nature and high resolution make it particularly suitable for investigating sodium nitrate's intricate structural features, including its rhombohedral crystal system and space group characteristics.
Historical investigations into sodium nitrate's crystalline structure date back to early crystallographic studies in the mid-20th century, where researchers first identified its room-temperature phase as belonging to the R3c space group. However, recent advances in X-ray diffraction instrumentation and computational analysis methods have opened new avenues for more detailed structural investigations, revealing previously undetected structural nuances and phase transition mechanisms.
The primary research objective centers on achieving comprehensive characterization of sodium nitrate's crystalline structure through systematic X-ray analysis, encompassing both static structural determination and dynamic phase behavior investigation. This involves precise measurement of lattice parameters, atomic coordinates, thermal displacement parameters, and electron density distributions within the crystal lattice.
Secondary objectives include establishing correlations between structural parameters and macroscopic properties, investigating temperature-induced phase transitions, and developing standardized protocols for sodium nitrate structural analysis. These goals aim to enhance understanding of structure-property relationships and provide foundational data for applications in materials science, pharmaceutical crystallography, and industrial process optimization.
The research framework emphasizes integration of high-resolution powder diffraction, single-crystal analysis, and variable-temperature studies to construct a comprehensive structural profile. This multi-faceted approach ensures robust characterization while addressing the compound's polymorphic complexity and environmental sensitivity, ultimately contributing to the broader field of ionic crystal structure determination.
Market Demand for Crystalline Structure Analysis Services
The global market for crystalline structure analysis services has experienced substantial growth driven by expanding applications across pharmaceutical, materials science, chemical, and academic research sectors. X-ray diffraction analysis, particularly for characterizing materials like sodium nitrate, represents a critical component of this market as industries increasingly require precise structural information for product development and quality control.
Pharmaceutical companies constitute the largest demand segment for crystalline structure analysis services. Drug development processes require comprehensive understanding of active pharmaceutical ingredient polymorphs, salt forms, and co-crystals to ensure optimal bioavailability and stability. Sodium nitrate, commonly used in pharmaceutical formulations and as a precursor in drug synthesis, necessitates detailed structural characterization to meet regulatory requirements and optimize manufacturing processes.
The materials science sector demonstrates growing demand for X-ray crystallographic services, particularly in developing advanced ceramics, catalysts, and electronic materials. Research institutions and industrial laboratories require precise lattice parameter determination, phase identification, and structural refinement services for materials containing nitrate compounds. This demand is amplified by increasing focus on sustainable materials and green chemistry applications.
Chemical manufacturing industries rely heavily on crystalline structure analysis for process optimization and product quality assurance. Sodium nitrate's widespread use in fertilizers, explosives, and glass manufacturing creates consistent demand for structural characterization services to ensure product specifications and regulatory compliance. Quality control laboratories require routine analysis to verify crystal purity and identify potential polymorphic variations.
Academic and government research institutions represent a stable demand base for specialized crystallographic services. Universities conducting fundamental research on inorganic compounds, crystallography studies, and materials characterization require access to advanced X-ray analysis capabilities. Government laboratories involved in forensic analysis, environmental monitoring, and defense applications also contribute to market demand.
The market exhibits regional variations with North America and Europe maintaining strong demand due to established pharmaceutical and chemical industries. Asia-Pacific regions show rapid growth driven by expanding manufacturing capabilities and increasing research investments. Emerging markets demonstrate growing awareness of the importance of structural characterization in product development and quality control processes.
Service providers face increasing demand for rapid turnaround times, comprehensive data analysis, and specialized expertise in handling challenging samples. The trend toward outsourcing analytical services rather than maintaining in-house capabilities has strengthened the commercial market for crystalline structure analysis services.
Pharmaceutical companies constitute the largest demand segment for crystalline structure analysis services. Drug development processes require comprehensive understanding of active pharmaceutical ingredient polymorphs, salt forms, and co-crystals to ensure optimal bioavailability and stability. Sodium nitrate, commonly used in pharmaceutical formulations and as a precursor in drug synthesis, necessitates detailed structural characterization to meet regulatory requirements and optimize manufacturing processes.
The materials science sector demonstrates growing demand for X-ray crystallographic services, particularly in developing advanced ceramics, catalysts, and electronic materials. Research institutions and industrial laboratories require precise lattice parameter determination, phase identification, and structural refinement services for materials containing nitrate compounds. This demand is amplified by increasing focus on sustainable materials and green chemistry applications.
Chemical manufacturing industries rely heavily on crystalline structure analysis for process optimization and product quality assurance. Sodium nitrate's widespread use in fertilizers, explosives, and glass manufacturing creates consistent demand for structural characterization services to ensure product specifications and regulatory compliance. Quality control laboratories require routine analysis to verify crystal purity and identify potential polymorphic variations.
Academic and government research institutions represent a stable demand base for specialized crystallographic services. Universities conducting fundamental research on inorganic compounds, crystallography studies, and materials characterization require access to advanced X-ray analysis capabilities. Government laboratories involved in forensic analysis, environmental monitoring, and defense applications also contribute to market demand.
The market exhibits regional variations with North America and Europe maintaining strong demand due to established pharmaceutical and chemical industries. Asia-Pacific regions show rapid growth driven by expanding manufacturing capabilities and increasing research investments. Emerging markets demonstrate growing awareness of the importance of structural characterization in product development and quality control processes.
Service providers face increasing demand for rapid turnaround times, comprehensive data analysis, and specialized expertise in handling challenging samples. The trend toward outsourcing analytical services rather than maintaining in-house capabilities has strengthened the commercial market for crystalline structure analysis services.
Current XRD Technology Status and Crystal Analysis Challenges
X-ray diffraction technology has evolved significantly since its inception in the early 20th century, establishing itself as the gold standard for crystalline structure characterization. Modern XRD systems employ sophisticated detector arrays, high-intensity X-ray sources, and advanced computational algorithms to achieve unprecedented resolution and accuracy in crystal analysis. Contemporary instruments can detect crystalline phases at concentrations as low as 1-2% and provide structural information with atomic-level precision.
Current XRD instrumentation encompasses various configurations including powder diffractometers, single-crystal diffractometers, and specialized high-resolution systems. Powder XRD remains the most widely used technique for routine phase identification and quantitative analysis, while single-crystal XRD provides detailed three-dimensional structural information. Advanced synchrotron-based XRD facilities offer exceptional beam quality and intensity, enabling time-resolved studies and analysis of challenging samples.
Despite technological advances, several fundamental challenges persist in XRD-based crystal analysis. Peak overlap in complex multi-phase systems often complicates accurate phase identification and quantitative analysis. This issue becomes particularly pronounced when analyzing samples containing phases with similar lattice parameters or when dealing with solid solutions where continuous structural variations occur.
Sample preparation remains a critical bottleneck affecting analysis quality. Preferred orientation effects can significantly distort intensity distributions, leading to inaccurate structural refinements. Additionally, crystallite size effects, including peak broadening due to finite crystallite dimensions and microstrain, complicate precise lattice parameter determination and structural analysis.
For sodium nitrate specifically, several analytical challenges emerge. The hygroscopic nature of sodium nitrate requires careful sample handling to prevent moisture absorption, which can alter the crystalline structure during measurement. Temperature-dependent polymorphic transitions in sodium nitrate add complexity, as the material can undergo structural changes during extended exposure to X-ray radiation or ambient conditions.
Quantitative phase analysis in sodium nitrate-containing mixtures presents additional difficulties due to potential chemical interactions with other components. The relatively high atomic numbers of constituent elements can lead to significant absorption effects, requiring careful consideration of sample geometry and thickness optimization.
Modern computational approaches, including Rietveld refinement and advanced indexing algorithms, have partially addressed these challenges by enabling more sophisticated data analysis. However, the fundamental limitations of conventional laboratory X-ray sources in terms of resolution and intensity continue to constrain the precision achievable in routine crystallographic analysis of complex systems.
Current XRD instrumentation encompasses various configurations including powder diffractometers, single-crystal diffractometers, and specialized high-resolution systems. Powder XRD remains the most widely used technique for routine phase identification and quantitative analysis, while single-crystal XRD provides detailed three-dimensional structural information. Advanced synchrotron-based XRD facilities offer exceptional beam quality and intensity, enabling time-resolved studies and analysis of challenging samples.
Despite technological advances, several fundamental challenges persist in XRD-based crystal analysis. Peak overlap in complex multi-phase systems often complicates accurate phase identification and quantitative analysis. This issue becomes particularly pronounced when analyzing samples containing phases with similar lattice parameters or when dealing with solid solutions where continuous structural variations occur.
Sample preparation remains a critical bottleneck affecting analysis quality. Preferred orientation effects can significantly distort intensity distributions, leading to inaccurate structural refinements. Additionally, crystallite size effects, including peak broadening due to finite crystallite dimensions and microstrain, complicate precise lattice parameter determination and structural analysis.
For sodium nitrate specifically, several analytical challenges emerge. The hygroscopic nature of sodium nitrate requires careful sample handling to prevent moisture absorption, which can alter the crystalline structure during measurement. Temperature-dependent polymorphic transitions in sodium nitrate add complexity, as the material can undergo structural changes during extended exposure to X-ray radiation or ambient conditions.
Quantitative phase analysis in sodium nitrate-containing mixtures presents additional difficulties due to potential chemical interactions with other components. The relatively high atomic numbers of constituent elements can lead to significant absorption effects, requiring careful consideration of sample geometry and thickness optimization.
Modern computational approaches, including Rietveld refinement and advanced indexing algorithms, have partially addressed these challenges by enabling more sophisticated data analysis. However, the fundamental limitations of conventional laboratory X-ray sources in terms of resolution and intensity continue to constrain the precision achievable in routine crystallographic analysis of complex systems.
Existing XRD Solutions for Nitrate Crystal Characterization
01 Crystal structure and morphology of sodium nitrate
Sodium nitrate exhibits specific crystalline structures and morphologies that can be characterized and analyzed. The crystal structure includes trigonal or rhombohedral systems with defined lattice parameters. Understanding the crystalline structure is essential for various applications including material science and chemical processing. The morphology and crystal habit can be influenced by crystallization conditions and may affect the physical properties of the material.- Crystal structure and morphology of sodium nitrate: Sodium nitrate exhibits specific crystalline structures and morphologies that can be characterized and analyzed. The crystal structure typically involves rhombohedral or trigonal systems, with specific lattice parameters and unit cell dimensions. Understanding the crystalline structure is essential for various applications including material science and chemical processing. The morphology and crystal habit can be influenced by crystallization conditions such as temperature, concentration, and presence of additives.
- Production and crystallization methods for sodium nitrate: Various methods exist for producing and crystallizing sodium nitrate with controlled crystal properties. These methods include evaporative crystallization, cooling crystallization, and reactive crystallization processes. The crystallization conditions such as supersaturation levels, cooling rates, and agitation can significantly affect the resulting crystal size distribution and purity. Industrial production methods often involve multi-stage crystallization processes to achieve desired crystal characteristics and product quality.
- Modification and control of sodium nitrate crystal properties: The crystal properties of sodium nitrate can be modified and controlled through various techniques including the use of additives, seed crystals, and controlled crystallization environments. Crystal habit modifiers and growth inhibitors can be employed to influence crystal shape, size, and surface characteristics. These modifications are important for optimizing handling properties, dissolution rates, and application-specific requirements. Temperature cycling and controlled nucleation can also be used to achieve desired crystal characteristics.
- Applications of sodium nitrate crystals in chemical processes: Sodium nitrate crystals find applications in various chemical processes including oxidation reactions, heat storage systems, and as raw materials for other chemical syntheses. The crystalline form affects reactivity, dissolution behavior, and processing characteristics. In thermal energy storage applications, the crystal structure and phase transitions are particularly important. The material can also serve as an oxidizing agent in various industrial processes where crystal properties influence reaction kinetics and efficiency.
- Analysis and characterization techniques for sodium nitrate crystals: Various analytical techniques are employed to characterize sodium nitrate crystalline structures including X-ray diffraction, microscopy, thermal analysis, and spectroscopic methods. These techniques provide information about crystal phase, purity, particle size distribution, and structural defects. Characterization is essential for quality control in production processes and for understanding structure-property relationships. Advanced techniques can reveal detailed information about crystal lattice parameters, polymorphic forms, and surface properties.
02 Production and crystallization methods for sodium nitrate
Various methods exist for producing and crystallizing sodium nitrate with controlled crystal structures. These methods include evaporation, cooling crystallization, and controlled precipitation techniques. The crystallization process parameters such as temperature, concentration, and cooling rate significantly influence the final crystal structure and purity. Industrial production methods focus on obtaining high-purity crystalline sodium nitrate with desired particle size distribution.Expand Specific Solutions03 Phase transitions and thermal properties of sodium nitrate crystals
Sodium nitrate undergoes phase transitions at specific temperatures, affecting its crystalline structure. The material exhibits polymorphism with different crystal phases stable at different temperature ranges. Thermal analysis reveals characteristic melting points and phase transformation temperatures. These thermal properties are important for applications in heat storage and thermal management systems.Expand Specific Solutions04 Composite materials and mixtures containing sodium nitrate crystals
Sodium nitrate crystals can be incorporated into composite materials and mixtures to achieve specific functional properties. These composites may include combinations with other salts, polymers, or inorganic materials. The crystalline structure of sodium nitrate in such composites affects the overall material properties including thermal conductivity, stability, and reactivity. Applications include thermal energy storage materials and specialized chemical formulations.Expand Specific Solutions05 Applications and modifications of sodium nitrate crystalline materials
Crystalline sodium nitrate finds applications in various fields including agriculture, explosives, and chemical synthesis. Modifications to the crystal structure through doping, coating, or surface treatment can enhance specific properties. The crystalline form affects dissolution rate, reactivity, and handling characteristics. Recent developments focus on optimizing crystal structure for improved performance in energy storage and catalytic applications.Expand Specific Solutions
Key Players in XRD Equipment and Crystal Analysis Industry
The X-ray crystallographic analysis of sodium nitrate represents a mature analytical technique within the broader structural characterization market, which has reached a stable growth phase with established methodologies and standardized protocols. The market demonstrates steady demand driven by pharmaceutical quality control, materials research, and chemical manufacturing applications. Technology maturity is evidenced by the presence of established instrumentation leaders like Bruker AXS and Toshiba Corp., alongside major pharmaceutical companies including Takeda Pharmaceutical, Dr. Reddy's Laboratories, Genentech, and Bayer AG who utilize these techniques for drug development and quality assurance. Research institutions such as University of Tokyo, Columbia University, and Japan Synchrotron Radiation Research Institute continue advancing methodological refinements, while specialized companies like Merck Patent GmbH and various Chinese pharmaceutical manufacturers including Jiangsu Hengrui and Hansoh Pharmaceutical integrate these analytical capabilities into their development pipelines, indicating widespread adoption across global markets.
Bruker AXS, Inc.
Technical Solution: Bruker AXS specializes in advanced X-ray analytical instrumentation for crystallographic analysis, offering comprehensive solutions for sodium nitrate structure characterization. Their D8 ADVANCE series provides high-resolution powder diffraction capabilities with LYNXEYE XE-T detector technology, enabling precise unit cell parameter determination and phase identification of sodium nitrate polymorphs. The company's APEX DUO single crystal diffractometer systems deliver sub-angstrom resolution for detailed atomic position mapping and thermal parameter analysis. Their integrated software suite including TOPAS and SHELX facilitates automated structure refinement and Rietveld analysis, supporting comprehensive crystallographic studies of ionic compounds like sodium nitrate under various temperature and pressure conditions.
Strengths: Industry-leading detector technology and comprehensive software integration for automated analysis. Weaknesses: High equipment costs and requires specialized technical expertise for operation.
Toshiba Corp.
Technical Solution: Toshiba develops advanced X-ray imaging and analysis systems through their Electronic Devices & Storage division, offering solutions applicable to crystallographic studies including sodium nitrate structure determination. Their X-ray detector technology incorporates CMOS-based flat panel detectors with high spatial resolution and low noise characteristics, suitable for powder diffraction applications. The company's expertise in semiconductor manufacturing enables production of high-performance X-ray sources and detection systems with enhanced sensitivity for weak diffraction signals. Toshiba's integrated approach combines hardware development with signal processing algorithms optimized for crystallographic data collection, supporting automated phase identification and quantitative analysis of crystalline materials like sodium nitrate in industrial quality control applications.
Strengths: Advanced detector technology and strong manufacturing capabilities for reliable instrumentation. Weaknesses: Limited specialization in crystallographic applications compared to dedicated analytical instrument manufacturers.
Core XRD Innovations for Sodium Nitrate Structure Analysis
Salt crystals
PatentWO2013192556A2
Innovation
- Comprehensive characterization of salt crystals using specific X-ray powder diffraction patterns with detailed 2-theta angle values and d-spacing measurements for precise structural identification.
- Systematic definition of needle-like morphology salt crystals in non-solvate form with standardized copper anode X-ray diffraction conditions using dual wavelengths (1.5406A and 1.5444A).
- Integration of multiple crystallographic parameters including 2-theta angles, d-spacing values, and morphological characteristics to create a comprehensive crystal identification framework.
Method of conducting an x-ray diffraction-based crystallography analysis
PatentWO2015114613A1
Innovation
- A method involving a pre-experiment and a main experiment with different rotational speeds and exposure times to collect diffraction images, where intensities from the pre-experiment are used to replace saturated pixels in the main experiment, allowing for continuous data collection without interruptions and improving data quality.
Safety Regulations for Nitrate Compound Analysis
The analysis of sodium nitrate crystalline structures through X-ray diffraction techniques requires strict adherence to comprehensive safety protocols due to the inherent hazards associated with nitrate compounds. These regulations encompass multiple layers of protection, from personnel safety to environmental considerations, ensuring that research activities are conducted within acceptable risk parameters.
Personnel protection forms the cornerstone of nitrate compound analysis safety protocols. All laboratory personnel must undergo specialized training in nitrate handling procedures, including recognition of potential hazards such as oxidizing properties, thermal sensitivity, and potential for explosive decomposition under certain conditions. Mandatory personal protective equipment includes chemical-resistant gloves, safety goggles with side shields, and flame-resistant laboratory coats. Respiratory protection may be required when handling powdered samples to prevent inhalation of particulates.
Laboratory infrastructure must comply with specific design standards for nitrate compound research. Ventilation systems require enhanced capacity with dedicated exhaust mechanisms to prevent accumulation of potentially hazardous vapors. Work surfaces must be constructed from non-reactive materials, and electrical equipment should be intrinsically safe or properly grounded to eliminate ignition sources. Fire suppression systems must be compatible with nitrate compounds, avoiding water-based systems that could exacerbate certain chemical reactions.
Sample handling protocols mandate strict quantity limitations and storage requirements. Sodium nitrate samples must be stored in appropriate containers away from incompatible materials such as organic compounds, reducing agents, and combustible materials. Temperature control is critical, with storage areas maintained below specified thresholds to prevent thermal decomposition. Sample preparation areas require segregation from other analytical activities to minimize cross-contamination risks.
X-ray equipment operation introduces additional safety considerations specific to nitrate analysis. Beam exposure protocols must account for potential sample degradation or unexpected reactions under X-ray irradiation. Emergency shutdown procedures should be readily accessible, and continuous monitoring systems must be in place to detect any anomalous behavior during analysis. Regular calibration and maintenance of X-ray equipment ensures consistent performance while minimizing safety risks.
Waste management protocols for nitrate compound analysis require specialized disposal procedures. Contaminated materials cannot be disposed of through standard laboratory waste streams and must be collected, labeled, and processed according to hazardous waste regulations. Decontamination procedures for equipment and work surfaces must use appropriate neutralizing agents that do not create additional hazards when combined with nitrate residues.
Personnel protection forms the cornerstone of nitrate compound analysis safety protocols. All laboratory personnel must undergo specialized training in nitrate handling procedures, including recognition of potential hazards such as oxidizing properties, thermal sensitivity, and potential for explosive decomposition under certain conditions. Mandatory personal protective equipment includes chemical-resistant gloves, safety goggles with side shields, and flame-resistant laboratory coats. Respiratory protection may be required when handling powdered samples to prevent inhalation of particulates.
Laboratory infrastructure must comply with specific design standards for nitrate compound research. Ventilation systems require enhanced capacity with dedicated exhaust mechanisms to prevent accumulation of potentially hazardous vapors. Work surfaces must be constructed from non-reactive materials, and electrical equipment should be intrinsically safe or properly grounded to eliminate ignition sources. Fire suppression systems must be compatible with nitrate compounds, avoiding water-based systems that could exacerbate certain chemical reactions.
Sample handling protocols mandate strict quantity limitations and storage requirements. Sodium nitrate samples must be stored in appropriate containers away from incompatible materials such as organic compounds, reducing agents, and combustible materials. Temperature control is critical, with storage areas maintained below specified thresholds to prevent thermal decomposition. Sample preparation areas require segregation from other analytical activities to minimize cross-contamination risks.
X-ray equipment operation introduces additional safety considerations specific to nitrate analysis. Beam exposure protocols must account for potential sample degradation or unexpected reactions under X-ray irradiation. Emergency shutdown procedures should be readily accessible, and continuous monitoring systems must be in place to detect any anomalous behavior during analysis. Regular calibration and maintenance of X-ray equipment ensures consistent performance while minimizing safety risks.
Waste management protocols for nitrate compound analysis require specialized disposal procedures. Contaminated materials cannot be disposed of through standard laboratory waste streams and must be collected, labeled, and processed according to hazardous waste regulations. Decontamination procedures for equipment and work surfaces must use appropriate neutralizing agents that do not create additional hazards when combined with nitrate residues.
Quality Standards for Crystallographic Data Validation
The establishment of rigorous quality standards for crystallographic data validation represents a critical foundation for reliable sodium nitrate structural characterization through X-ray analysis. These standards encompass multiple dimensions of data quality assessment, ensuring that crystallographic measurements meet internationally recognized criteria for accuracy, precision, and reproducibility.
Data completeness serves as a fundamental quality metric, typically requiring collection of at least 95% of theoretically observable reflections within the specified resolution range. For sodium nitrate crystals, this completeness threshold ensures adequate sampling of reciprocal space to accurately determine atomic positions and thermal parameters. Resolution limits must extend sufficiently to capture fine structural details, with high-quality datasets typically achieving resolutions better than 0.8 Angstroms for small molecule structures.
Statistical indicators provide quantitative measures of data quality and structural refinement reliability. The R-factor, representing the agreement between observed and calculated structure factors, should typically remain below 5% for high-quality sodium nitrate structures. The goodness-of-fit parameter must approach unity, indicating appropriate weighting schemes and realistic error estimates. Residual electron density peaks should not exceed 0.5 electrons per cubic Angstrom in the final difference maps.
Geometric validation standards ensure chemical reasonableness of the refined structure. Bond lengths and angles must fall within established ranges for sodium-oxygen and nitrogen-oxygen interactions, with deviations not exceeding three standard deviations from expected values. Thermal parameter validation requires physically meaningful displacement ellipsoids, with no non-positive definite parameters or unrealistic anisotropic displacement values.
Systematic error detection protocols identify potential issues affecting data quality. These include assessment of absorption corrections, extinction effects, and twinning phenomena that commonly affect ionic crystals like sodium nitrate. Scale factor stability across different resolution shells indicates proper data processing and integration procedures.
Documentation standards mandate comprehensive reporting of experimental conditions, data collection parameters, and refinement procedures. This includes crystal mounting conditions, temperature control, radiation wavelength, detector specifications, and software versions used throughout the analysis pipeline, ensuring reproducibility and enabling proper peer review of crystallographic results.
Data completeness serves as a fundamental quality metric, typically requiring collection of at least 95% of theoretically observable reflections within the specified resolution range. For sodium nitrate crystals, this completeness threshold ensures adequate sampling of reciprocal space to accurately determine atomic positions and thermal parameters. Resolution limits must extend sufficiently to capture fine structural details, with high-quality datasets typically achieving resolutions better than 0.8 Angstroms for small molecule structures.
Statistical indicators provide quantitative measures of data quality and structural refinement reliability. The R-factor, representing the agreement between observed and calculated structure factors, should typically remain below 5% for high-quality sodium nitrate structures. The goodness-of-fit parameter must approach unity, indicating appropriate weighting schemes and realistic error estimates. Residual electron density peaks should not exceed 0.5 electrons per cubic Angstrom in the final difference maps.
Geometric validation standards ensure chemical reasonableness of the refined structure. Bond lengths and angles must fall within established ranges for sodium-oxygen and nitrogen-oxygen interactions, with deviations not exceeding three standard deviations from expected values. Thermal parameter validation requires physically meaningful displacement ellipsoids, with no non-positive definite parameters or unrealistic anisotropic displacement values.
Systematic error detection protocols identify potential issues affecting data quality. These include assessment of absorption corrections, extinction effects, and twinning phenomena that commonly affect ionic crystals like sodium nitrate. Scale factor stability across different resolution shells indicates proper data processing and integration procedures.
Documentation standards mandate comprehensive reporting of experimental conditions, data collection parameters, and refinement procedures. This includes crystal mounting conditions, temperature control, radiation wavelength, detector specifications, and software versions used throughout the analysis pipeline, ensuring reproducibility and enabling proper peer review of crystallographic results.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!