Cryopreservation vs Preservation in Ethanol: Practical Guide
FEB 12, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cryopreservation and Ethanol Preservation: Background and Objectives
Biological sample preservation represents a critical challenge across multiple scientific disciplines, from biodiversity conservation to biomedical research. The choice between cryopreservation and ethanol-based preservation methods has profound implications for sample integrity, research outcomes, and operational feasibility. Both techniques have evolved significantly over the past decades, yet their application remains context-dependent and often requires careful consideration of multiple technical and practical factors.
Cryopreservation, the process of preserving biological materials at ultra-low temperatures, has emerged as the gold standard for maintaining cellular viability and molecular integrity. This technique relies on controlled freezing protocols that minimize ice crystal formation and cellular damage, typically utilizing liquid nitrogen at -196°C or mechanical freezers at -80°C. The fundamental principle involves reducing metabolic activity to near-zero levels, effectively suspending biological processes while preserving structural and functional characteristics.
Ethanol preservation, conversely, represents a chemical fixation approach that has been utilized for over a century in taxonomy, ecology, and molecular biology. This method involves immersing specimens in ethanol solutions, typically at concentrations between 70% and 100%, which denatures proteins and dehydrates tissues, thereby preventing microbial degradation. While ethanol preservation sacrifices cellular viability, it offers distinct advantages in terms of accessibility, cost-effectiveness, and field applicability.
The primary objective of this technical investigation is to provide a comprehensive comparative framework for selecting between these preservation methodologies. This includes evaluating their respective capabilities in maintaining DNA integrity, protein structure, morphological features, and metabolomic profiles. Additionally, the analysis aims to establish practical guidelines considering factors such as sample type, intended downstream applications, resource availability, and long-term storage requirements.
Understanding the evolutionary trajectory of both technologies reveals important insights into their current capabilities and limitations. Cryopreservation has benefited from advances in cryoprotectant formulations and controlled-rate freezing equipment, while ethanol preservation has seen refinements in concentration optimization and storage protocols. These developments have expanded the applicability of both methods across diverse biological materials, from microbial cultures to complex tissue samples.
Cryopreservation, the process of preserving biological materials at ultra-low temperatures, has emerged as the gold standard for maintaining cellular viability and molecular integrity. This technique relies on controlled freezing protocols that minimize ice crystal formation and cellular damage, typically utilizing liquid nitrogen at -196°C or mechanical freezers at -80°C. The fundamental principle involves reducing metabolic activity to near-zero levels, effectively suspending biological processes while preserving structural and functional characteristics.
Ethanol preservation, conversely, represents a chemical fixation approach that has been utilized for over a century in taxonomy, ecology, and molecular biology. This method involves immersing specimens in ethanol solutions, typically at concentrations between 70% and 100%, which denatures proteins and dehydrates tissues, thereby preventing microbial degradation. While ethanol preservation sacrifices cellular viability, it offers distinct advantages in terms of accessibility, cost-effectiveness, and field applicability.
The primary objective of this technical investigation is to provide a comprehensive comparative framework for selecting between these preservation methodologies. This includes evaluating their respective capabilities in maintaining DNA integrity, protein structure, morphological features, and metabolomic profiles. Additionally, the analysis aims to establish practical guidelines considering factors such as sample type, intended downstream applications, resource availability, and long-term storage requirements.
Understanding the evolutionary trajectory of both technologies reveals important insights into their current capabilities and limitations. Cryopreservation has benefited from advances in cryoprotectant formulations and controlled-rate freezing equipment, while ethanol preservation has seen refinements in concentration optimization and storage protocols. These developments have expanded the applicability of both methods across diverse biological materials, from microbial cultures to complex tissue samples.
Market Demand for Biological Sample Preservation Solutions
The global market for biological sample preservation solutions has experienced substantial growth driven by expanding applications across biobanking, pharmaceutical research, clinical diagnostics, and academic institutions. The increasing emphasis on precision medicine and personalized therapeutics has amplified demand for high-quality preserved biological materials, including tissues, cells, DNA, RNA, and protein samples. Both cryopreservation and ethanol-based preservation methods serve critical roles in meeting these diverse requirements, each addressing specific market segments based on sample type, storage duration, and downstream application needs.
Biobanking facilities represent a major demand driver, as they require scalable and cost-effective preservation solutions for long-term storage of diverse biological specimens. Cryopreservation dominates in scenarios demanding maximum sample viability and functional integrity, particularly for cell lines, stem cells, and reproductive materials. However, the operational complexity and infrastructure costs associated with ultra-low temperature storage create opportunities for alternative methods. Ethanol preservation has gained traction in large-scale epidemiological studies and field research where maintaining nucleic acid integrity is prioritized over cellular viability, offering logistical advantages in resource-limited settings.
The pharmaceutical and biotechnology sectors demonstrate growing demand for preservation solutions that support drug discovery pipelines and clinical trial sample management. These industries require methods that ensure sample stability during transportation and storage while maintaining molecular integrity for genomic and proteomic analyses. The choice between cryopreservation and ethanol fixation often depends on specific analytical endpoints, with ethanol-based methods increasingly favored for nucleic acid-focused applications due to simplified handling requirements and reduced cold-chain dependencies.
Emerging markets in Asia-Pacific and Latin America show accelerated adoption of biological sample preservation technologies, driven by expanding healthcare infrastructure and increasing research investments. These regions exhibit particular interest in cost-effective preservation solutions that balance sample quality with operational feasibility. The veterinary and agricultural sectors also contribute to market expansion, requiring preservation methods for genetic material, tissue samples, and pathogen surveillance programs. Environmental monitoring and biodiversity research further broaden the application landscape, where field-deployable preservation techniques are essential for specimen collection in remote locations.
Biobanking facilities represent a major demand driver, as they require scalable and cost-effective preservation solutions for long-term storage of diverse biological specimens. Cryopreservation dominates in scenarios demanding maximum sample viability and functional integrity, particularly for cell lines, stem cells, and reproductive materials. However, the operational complexity and infrastructure costs associated with ultra-low temperature storage create opportunities for alternative methods. Ethanol preservation has gained traction in large-scale epidemiological studies and field research where maintaining nucleic acid integrity is prioritized over cellular viability, offering logistical advantages in resource-limited settings.
The pharmaceutical and biotechnology sectors demonstrate growing demand for preservation solutions that support drug discovery pipelines and clinical trial sample management. These industries require methods that ensure sample stability during transportation and storage while maintaining molecular integrity for genomic and proteomic analyses. The choice between cryopreservation and ethanol fixation often depends on specific analytical endpoints, with ethanol-based methods increasingly favored for nucleic acid-focused applications due to simplified handling requirements and reduced cold-chain dependencies.
Emerging markets in Asia-Pacific and Latin America show accelerated adoption of biological sample preservation technologies, driven by expanding healthcare infrastructure and increasing research investments. These regions exhibit particular interest in cost-effective preservation solutions that balance sample quality with operational feasibility. The veterinary and agricultural sectors also contribute to market expansion, requiring preservation methods for genetic material, tissue samples, and pathogen surveillance programs. Environmental monitoring and biodiversity research further broaden the application landscape, where field-deployable preservation techniques are essential for specimen collection in remote locations.
Current Status and Challenges in Preservation Technologies
Preservation technologies for biological specimens have evolved significantly over the past century, yet both cryopreservation and ethanol-based preservation continue to face distinct technical and operational challenges. Cryopreservation, which involves storing specimens at ultra-low temperatures typically below -130°C, has become the gold standard for maintaining cellular viability and genetic integrity. However, this method requires substantial infrastructure investment, including liquid nitrogen supply systems, specialized storage facilities, and continuous monitoring equipment. The formation of ice crystals during the freezing process remains a critical challenge, as these can cause cellular damage and compromise specimen quality. Despite advances in cryoprotective agents and controlled-rate freezing protocols, achieving consistent results across different specimen types continues to be problematic.
Ethanol preservation, conversely, offers a more accessible and cost-effective alternative, particularly for DNA-based studies and morphological analysis. This method has gained widespread adoption in field research and resource-limited settings due to its simplicity and room-temperature storage capability. However, ethanol preservation faces significant limitations in maintaining RNA integrity, protein structure, and cellular viability. The degradation of certain biomolecules over extended storage periods presents ongoing concerns for long-term archival applications. Additionally, ethanol concentration optimization, specimen-to-preservative ratios, and container selection remain areas requiring standardization across different biological materials.
Both preservation methods encounter common challenges related to quality control, standardization, and long-term sustainability. The lack of universal protocols across institutions creates difficulties in specimen exchange and comparative studies. Energy consumption and environmental impact considerations are increasingly important, particularly for cryopreservation facilities requiring continuous power supply. Furthermore, the scalability of preservation operations to accommodate growing biobank collections presents logistical and financial constraints. Emerging hybrid approaches and novel preservation media are being explored to address these limitations, yet widespread implementation remains hindered by validation requirements and regulatory considerations. The geographical distribution of advanced preservation facilities also creates disparities in access to optimal preservation technologies, particularly affecting research institutions in developing regions.
Ethanol preservation, conversely, offers a more accessible and cost-effective alternative, particularly for DNA-based studies and morphological analysis. This method has gained widespread adoption in field research and resource-limited settings due to its simplicity and room-temperature storage capability. However, ethanol preservation faces significant limitations in maintaining RNA integrity, protein structure, and cellular viability. The degradation of certain biomolecules over extended storage periods presents ongoing concerns for long-term archival applications. Additionally, ethanol concentration optimization, specimen-to-preservative ratios, and container selection remain areas requiring standardization across different biological materials.
Both preservation methods encounter common challenges related to quality control, standardization, and long-term sustainability. The lack of universal protocols across institutions creates difficulties in specimen exchange and comparative studies. Energy consumption and environmental impact considerations are increasingly important, particularly for cryopreservation facilities requiring continuous power supply. Furthermore, the scalability of preservation operations to accommodate growing biobank collections presents logistical and financial constraints. Emerging hybrid approaches and novel preservation media are being explored to address these limitations, yet widespread implementation remains hindered by validation requirements and regulatory considerations. The geographical distribution of advanced preservation facilities also creates disparities in access to optimal preservation technologies, particularly affecting research institutions in developing regions.
Comparative Analysis of Current Preservation Protocols
01 Cryopreservation methods for biological samples
Various cryopreservation techniques have been developed for preserving biological samples including cells, tissues, and organs at ultra-low temperatures. These methods typically involve controlled cooling rates, cryoprotective agents, and specialized storage containers to maintain sample viability during freezing and thawing processes. The techniques aim to minimize ice crystal formation and cellular damage while ensuring long-term preservation of biological materials.- Cryopreservation methods for biological samples: Various cryopreservation techniques have been developed to preserve biological samples at ultra-low temperatures. These methods involve the use of cryoprotective agents and controlled cooling rates to prevent ice crystal formation and maintain cell viability. The techniques are applicable to different types of biological materials including cells, tissues, and reproductive materials.
- Ethanol-based preservation solutions and formulations: Ethanol-based preservation solutions provide an alternative method for storing biological specimens at ambient or refrigerated temperatures. These formulations typically contain specific concentrations of ethanol combined with other stabilizing agents to maintain sample integrity and prevent degradation. The method offers advantages in terms of storage convenience and cost-effectiveness compared to cryogenic storage.
- Equipment and devices for cryopreservation storage: Specialized equipment and storage devices have been designed to facilitate cryopreservation processes. These include cryogenic containers, automated freezing systems, and monitoring devices that ensure optimal storage conditions. The equipment features temperature control mechanisms, sample organization systems, and safety features to maintain sample quality during long-term storage.
- Combined preservation methods and hybrid approaches: Innovative preservation strategies combine multiple preservation techniques to optimize sample stability and viability. These hybrid approaches may integrate cryopreservation with chemical stabilization methods or utilize sequential preservation steps. The combined methods aim to leverage the advantages of different preservation techniques while minimizing their individual limitations.
- Preservation of specific biological materials and applications: Specialized preservation protocols have been developed for specific types of biological materials such as stem cells, gametes, embryos, microorganisms, and tissue samples. These protocols are tailored to address the unique preservation requirements of different biological materials, taking into account factors such as cell type, intended use, and storage duration. The methods ensure optimal recovery and functionality of preserved materials for various applications in research, medicine, and biotechnology.
02 Ethanol-based preservation solutions and formulations
Ethanol-based preservation solutions are utilized for storing and maintaining biological specimens, samples, and materials. These formulations typically contain specific concentrations of ethanol combined with other stabilizing agents to prevent degradation and maintain sample integrity. The preservation solutions are designed to provide effective storage at various temperatures while maintaining the structural and molecular characteristics of the preserved materials.Expand Specific Solutions03 Specialized equipment and devices for cryopreservation
Specialized equipment and apparatus have been designed for cryopreservation processes, including storage containers, freezing devices, and temperature control systems. These devices feature specific structural designs to facilitate controlled freezing, efficient storage, and safe handling of cryopreserved materials. The equipment often incorporates features for monitoring temperature, managing liquid nitrogen, and organizing multiple samples.Expand Specific Solutions04 Combined preservation methods using cryopreservation and chemical stabilization
Hybrid preservation approaches combine cryopreservation techniques with chemical stabilization methods, including ethanol-based solutions. These combined methods aim to enhance preservation efficiency by leveraging both low-temperature storage and chemical fixation. The techniques are particularly useful for specific types of biological materials that require multi-step preservation protocols to maintain their functional and structural properties.Expand Specific Solutions05 Cryopreservation media and cryoprotective agent compositions
Specialized cryopreservation media and cryoprotective agent formulations have been developed to improve cell and tissue survival during freezing and thawing. These compositions include various combinations of cryoprotectants, nutrients, and stabilizing compounds designed to protect cellular structures from freeze-thaw damage. The formulations are optimized for different cell types and preservation requirements to maximize post-thaw viability and functionality.Expand Specific Solutions
Major Players in Biopreservation Industry
The cryopreservation versus ethanol preservation landscape represents a mature yet evolving technical domain, with market growth driven by expanding biopharmaceutical and biobanking applications. The competitive arena spans specialized cryogenic equipment manufacturers like Mayekawa MFG and Shanghai Pudong Freeze Dryer Equipment, pharmaceutical consumables providers including SiO2 Medical Products and Reflect Scientific, and biotechnology firms such as Sanaria and GenoTech Corp. Technology maturity varies significantly across segments, with established players like Grifols SA and Asymptote Ltd. offering advanced cryochain solutions, while chemical suppliers including Celanese International and Indena SpA provide preservation media and ethanol-based alternatives. Academic institutions like University of Southern California and Zhejiang University contribute foundational research, indicating ongoing innovation. The market demonstrates consolidation around integrated solution providers like CryoCrate LLC, combining hardware, consumables, and protocols for optimized sample preservation across pharmaceutical, clinical, and research applications.
Sanaria, Inc.
Technical Solution: Sanaria Inc. has developed specialized cryopreservation protocols primarily for vaccine and biological product preservation, particularly focusing on malaria vaccine components and sporozoites. Their approach combines optimized cryoprotectant formulations with precise cooling rate control to maintain biological activity of sensitive vaccine materials. The technology employs programmable freezers that execute multi-step cooling profiles, transitioning samples through critical temperature zones at controlled rates to minimize cellular stress and ice crystal damage. Sanaria's methods incorporate glycerol and other cryoprotective agents in carefully calibrated concentrations, balanced to provide protection while minimizing toxicity. Their preservation protocols have been validated for maintaining immunogenicity and infectivity of biological materials over extended storage periods, making them particularly relevant for vaccine distribution and biomedical research applications.
Strengths: Proven efficacy for complex biological products including live vaccines, maintains immunological properties and biological activity, validated protocols with regulatory acceptance for clinical applications. Weaknesses: Requires sophisticated equipment and precise protocol execution, higher complexity compared to simple ethanol preservation, limited applicability outside specialized biological products, significant infrastructure requirements for field deployment.
Reflect Scientific, Inc.
Technical Solution: Reflect Scientific develops ultra-low temperature refrigeration systems specifically designed for cryogenic preservation applications. Their technology utilizes mechanical cascade refrigeration systems that can achieve temperatures down to -86°C without requiring liquid nitrogen, providing an alternative to traditional cryopreservation methods. The systems feature advanced insulation technology and energy-efficient compressor designs that reduce operational costs while maintaining stable temperature control. Their equipment is widely used in biobanking, pharmaceutical research, and clinical sample storage where maintaining sample integrity is critical. The company's solutions bridge the gap between standard freezer storage and full cryogenic preservation, offering flexibility for different preservation requirements and sample types.
Strengths: Lower operational costs compared to liquid nitrogen systems, reduced dependency on consumable cryogens, stable temperature control with minimal fluctuation. Weaknesses: Temperature range limited compared to full cryopreservation (-86°C vs -196°C), may not be suitable for all cell types requiring deeper cryogenic temperatures, higher initial equipment investment than ethanol-based preservation.
Key Technical Innovations in Cryoprotectant and Fixative Solutions
Cryopreservation solution and cryopreservation method
PatentInactiveUS20210235687A1
Innovation
- A cryopreservation solution containing polyvinyl alcohol with a saponification degree of 84 mol % or lower, preferably 76 mol % or lower, is used to facilitate easy recovery of cells or tissues during thawing by reducing adhesion to the deposition surface.
Methods for cryopreservation
PatentWO2017072499A1
Innovation
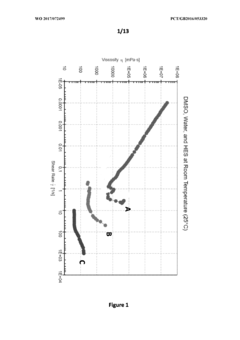
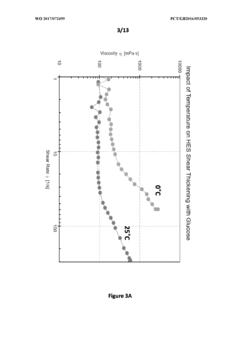
- The use of a non-Newtonian fluid as a cryopreservation medium, which modulates viscosity through shear thinning and shear thickening stresses, allowing for slower cooling rates and reduced cryoprotectant toxicity, thereby preventing ice crystal formation and enhancing sample preservation.
Storage Infrastructure and Cold Chain Requirements
The infrastructure requirements for cryopreservation and ethanol preservation differ substantially in complexity, cost, and operational demands. Cryopreservation necessitates sophisticated cold chain systems capable of maintaining ultra-low temperatures, typically ranging from -80°C for mechanical freezers to -196°C for liquid nitrogen storage. These systems require specialized facilities with adequate electrical capacity, backup power generators, and temperature monitoring equipment with alarm systems to prevent catastrophic sample loss during power failures or equipment malfunctions.
Liquid nitrogen storage facilities demand proper ventilation systems to prevent asphyxiation risks in enclosed spaces, along with appropriate safety protocols and personal protective equipment for handling cryogenic materials. The infrastructure must accommodate both storage dewars and working containers, with sufficient space for sample organization and retrieval operations. Additionally, automated filling systems or regular manual refilling schedules are essential to maintain nitrogen levels, requiring dedicated supply chains and vendor relationships.
In contrast, ethanol preservation requires significantly simpler infrastructure, typically operating at room temperature or requiring only standard refrigeration at 4°C to -20°C. Storage facilities can utilize conventional laboratory freezers or climate-controlled rooms without specialized cryogenic equipment. The primary infrastructure considerations involve adequate ventilation for volatile ethanol vapors, fire safety systems compliant with flammable liquid storage regulations, and appropriate containment measures to prevent spills.
Transportation logistics present distinct challenges for each method. Cryopreserved samples require dry shippers or specialized cryogenic transport containers that maintain ultra-low temperatures during transit, often limiting shipping options and increasing costs substantially. Ethanol-preserved samples offer greater flexibility, allowing standard courier services with minimal temperature control requirements, thereby reducing logistical complexity and expanding accessibility to remote locations.
The scalability of storage infrastructure varies considerably between methods. Cryopreservation facilities face space constraints due to the physical dimensions of dewars and safety clearances, while ethanol preservation allows more compact storage configurations using standard laboratory shelving systems. Long-term operational costs must account for continuous liquid nitrogen consumption, electricity for mechanical freezers, and maintenance contracts for specialized equipment in cryopreservation systems, whereas ethanol preservation primarily incurs costs for solvent replenishment and basic refrigeration utilities.
Liquid nitrogen storage facilities demand proper ventilation systems to prevent asphyxiation risks in enclosed spaces, along with appropriate safety protocols and personal protective equipment for handling cryogenic materials. The infrastructure must accommodate both storage dewars and working containers, with sufficient space for sample organization and retrieval operations. Additionally, automated filling systems or regular manual refilling schedules are essential to maintain nitrogen levels, requiring dedicated supply chains and vendor relationships.
In contrast, ethanol preservation requires significantly simpler infrastructure, typically operating at room temperature or requiring only standard refrigeration at 4°C to -20°C. Storage facilities can utilize conventional laboratory freezers or climate-controlled rooms without specialized cryogenic equipment. The primary infrastructure considerations involve adequate ventilation for volatile ethanol vapors, fire safety systems compliant with flammable liquid storage regulations, and appropriate containment measures to prevent spills.
Transportation logistics present distinct challenges for each method. Cryopreserved samples require dry shippers or specialized cryogenic transport containers that maintain ultra-low temperatures during transit, often limiting shipping options and increasing costs substantially. Ethanol-preserved samples offer greater flexibility, allowing standard courier services with minimal temperature control requirements, thereby reducing logistical complexity and expanding accessibility to remote locations.
The scalability of storage infrastructure varies considerably between methods. Cryopreservation facilities face space constraints due to the physical dimensions of dewars and safety clearances, while ethanol preservation allows more compact storage configurations using standard laboratory shelving systems. Long-term operational costs must account for continuous liquid nitrogen consumption, electricity for mechanical freezers, and maintenance contracts for specialized equipment in cryopreservation systems, whereas ethanol preservation primarily incurs costs for solvent replenishment and basic refrigeration utilities.
Cost-Benefit Analysis of Preservation Method Selection
When selecting between cryopreservation and ethanol preservation methods, organizations must conduct comprehensive cost-benefit analyses that extend beyond initial investment considerations. The economic viability of each approach depends on multiple factors including operational scale, specimen types, storage duration requirements, and institutional infrastructure capabilities.
Cryopreservation typically demands substantial upfront capital expenditure for specialized equipment such as liquid nitrogen storage systems, programmable freezers, and backup power supplies. Operational costs include continuous liquid nitrogen replenishment, which can range from several thousand to tens of thousands of dollars annually depending on collection size. However, this method offers superior sample integrity preservation, potentially reducing costs associated with specimen degradation and repeat collections. The long-term value proposition becomes particularly favorable for high-value biological materials where sample quality directly impacts research outcomes or commercial applications.
Ethanol preservation presents a markedly different economic profile with significantly lower initial investment requirements. Standard laboratory-grade storage facilities and ethanol solutions represent minimal capital barriers to entry. Operational expenses remain relatively stable and predictable, primarily involving ethanol replacement and basic environmental controls. This approach proves especially cost-effective for large-scale collections where individual specimen value may not justify cryopreservation expenses. Educational institutions and field research programs frequently favor this method due to budget constraints and logistical simplicity.
The decision framework must incorporate hidden costs often overlooked in preliminary assessments. Cryopreservation systems require specialized technical expertise for maintenance and troubleshooting, potentially necessitating staff training investments or external service contracts. Risk mitigation costs, including equipment redundancy and disaster recovery protocols, add substantial financial burden. Conversely, ethanol preservation incurs costs related to chemical disposal compliance, ventilation system requirements, and potential specimen quality compromises that may necessitate supplementary preservation of critical samples.
Return on investment calculations should account for specimen utilization patterns and institutional research priorities. High-throughput molecular biology applications demanding pristine nucleic acid quality typically justify cryopreservation costs through enhanced experimental success rates and reduced material waste. Morphological studies or long-term archival collections may achieve adequate outcomes with ethanol preservation at fraction of the cost, redirecting resources toward collection expansion or complementary research activities.
Cryopreservation typically demands substantial upfront capital expenditure for specialized equipment such as liquid nitrogen storage systems, programmable freezers, and backup power supplies. Operational costs include continuous liquid nitrogen replenishment, which can range from several thousand to tens of thousands of dollars annually depending on collection size. However, this method offers superior sample integrity preservation, potentially reducing costs associated with specimen degradation and repeat collections. The long-term value proposition becomes particularly favorable for high-value biological materials where sample quality directly impacts research outcomes or commercial applications.
Ethanol preservation presents a markedly different economic profile with significantly lower initial investment requirements. Standard laboratory-grade storage facilities and ethanol solutions represent minimal capital barriers to entry. Operational expenses remain relatively stable and predictable, primarily involving ethanol replacement and basic environmental controls. This approach proves especially cost-effective for large-scale collections where individual specimen value may not justify cryopreservation expenses. Educational institutions and field research programs frequently favor this method due to budget constraints and logistical simplicity.
The decision framework must incorporate hidden costs often overlooked in preliminary assessments. Cryopreservation systems require specialized technical expertise for maintenance and troubleshooting, potentially necessitating staff training investments or external service contracts. Risk mitigation costs, including equipment redundancy and disaster recovery protocols, add substantial financial burden. Conversely, ethanol preservation incurs costs related to chemical disposal compliance, ventilation system requirements, and potential specimen quality compromises that may necessitate supplementary preservation of critical samples.
Return on investment calculations should account for specimen utilization patterns and institutional research priorities. High-throughput molecular biology applications demanding pristine nucleic acid quality typically justify cryopreservation costs through enhanced experimental success rates and reduced material waste. Morphological studies or long-term archival collections may achieve adequate outcomes with ethanol preservation at fraction of the cost, redirecting resources toward collection expansion or complementary research activities.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!