Evaluate Dodecyl Acid Purity using NMR Spectroscopy
MAR 19, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
NMR Spectroscopy for Dodecyl Acid Analysis Background
Nuclear Magnetic Resonance (NMR) spectroscopy has emerged as a cornerstone analytical technique in organic chemistry since its discovery in the 1940s by Felix Bloch and Edward Purcell. The technology leverages the magnetic properties of atomic nuclei to provide detailed structural information about molecules, making it invaluable for purity assessment and compositional analysis of organic compounds.
The evolution of NMR technology has been marked by significant milestones, including the development of Fourier Transform NMR in the 1960s, which dramatically improved sensitivity and resolution. Subsequent advances in superconducting magnets, pulse sequences, and digital signal processing have transformed NMR from a research curiosity into an indispensable analytical tool across pharmaceutical, chemical, and materials industries.
Dodecyl acid, also known as lauric acid, represents a critical fatty acid component in numerous industrial applications, ranging from surfactant production to pharmaceutical formulations. The increasing demand for high-purity dodecyl acid in specialized applications has necessitated more sophisticated analytical approaches beyond traditional methods such as gas chromatography or titration techniques.
The application of NMR spectroscopy to dodecyl acid purity evaluation addresses several analytical challenges inherent in fatty acid analysis. Traditional methods often struggle with distinguishing between closely related fatty acid homologs or detecting trace impurities that can significantly impact product performance. NMR spectroscopy offers unique advantages through its ability to provide comprehensive molecular fingerprinting without requiring extensive sample preparation or derivatization procedures.
The primary objective of implementing NMR-based purity assessment for dodecyl acid centers on achieving quantitative determination of main component concentration while simultaneously identifying and quantifying potential impurities. This approach aims to establish a robust, reproducible analytical protocol that can detect impurities at levels below one percent, surpassing the sensitivity limitations of conventional analytical methods.
Furthermore, the development of NMR-based purity evaluation seeks to create a comprehensive understanding of impurity profiles commonly encountered in dodecyl acid samples, including shorter and longer chain fatty acids, unsaturated analogs, and synthetic byproducts. This knowledge base will enable more informed quality control decisions and process optimization strategies in dodecyl acid production and purification workflows.
The evolution of NMR technology has been marked by significant milestones, including the development of Fourier Transform NMR in the 1960s, which dramatically improved sensitivity and resolution. Subsequent advances in superconducting magnets, pulse sequences, and digital signal processing have transformed NMR from a research curiosity into an indispensable analytical tool across pharmaceutical, chemical, and materials industries.
Dodecyl acid, also known as lauric acid, represents a critical fatty acid component in numerous industrial applications, ranging from surfactant production to pharmaceutical formulations. The increasing demand for high-purity dodecyl acid in specialized applications has necessitated more sophisticated analytical approaches beyond traditional methods such as gas chromatography or titration techniques.
The application of NMR spectroscopy to dodecyl acid purity evaluation addresses several analytical challenges inherent in fatty acid analysis. Traditional methods often struggle with distinguishing between closely related fatty acid homologs or detecting trace impurities that can significantly impact product performance. NMR spectroscopy offers unique advantages through its ability to provide comprehensive molecular fingerprinting without requiring extensive sample preparation or derivatization procedures.
The primary objective of implementing NMR-based purity assessment for dodecyl acid centers on achieving quantitative determination of main component concentration while simultaneously identifying and quantifying potential impurities. This approach aims to establish a robust, reproducible analytical protocol that can detect impurities at levels below one percent, surpassing the sensitivity limitations of conventional analytical methods.
Furthermore, the development of NMR-based purity evaluation seeks to create a comprehensive understanding of impurity profiles commonly encountered in dodecyl acid samples, including shorter and longer chain fatty acids, unsaturated analogs, and synthetic byproducts. This knowledge base will enable more informed quality control decisions and process optimization strategies in dodecyl acid production and purification workflows.
Market Demand for High-Purity Dodecyl Acid Products
The global market for high-purity dodecyl acid products has experienced substantial growth driven by increasing demand across multiple industrial sectors. The pharmaceutical industry represents one of the most significant demand drivers, where dodecyl acid serves as a critical intermediate in the synthesis of various drug compounds and pharmaceutical excipients. The stringent regulatory requirements in pharmaceutical manufacturing necessitate extremely high purity levels, typically exceeding 99.5%, making accurate purity assessment through advanced analytical techniques like NMR spectroscopy essential for market acceptance.
Personal care and cosmetics industries constitute another major market segment for high-purity dodecyl acid products. The compound functions as an emulsifier, surfactant, and stabilizing agent in premium skincare formulations, hair care products, and luxury cosmetics. Consumer preferences for high-quality personal care products with minimal impurities have intensified the demand for pharmaceutical-grade dodecyl acid, particularly in anti-aging creams, moisturizers, and specialty hair treatments.
The specialty chemicals sector demonstrates growing appetite for ultra-pure dodecyl acid in advanced material synthesis and catalytic processes. Applications include the production of high-performance polymers, specialty coatings, and advanced lubricants where even trace impurities can significantly impact final product performance. This market segment particularly values suppliers who can provide comprehensive analytical documentation, including detailed NMR spectroscopic analysis confirming purity levels.
Food and beverage industries represent an emerging market opportunity for high-purity dodecyl acid products, primarily as food-grade emulsifiers and processing aids. Regulatory compliance requirements in food applications demand exceptional purity standards and comprehensive analytical verification, creating opportunities for suppliers equipped with advanced analytical capabilities.
The market trend toward sustainable and bio-based dodecyl acid production has created additional demand for high-purity products derived from renewable feedstocks. Manufacturers focusing on green chemistry approaches while maintaining superior purity levels through rigorous analytical control are positioned to capture premium market segments increasingly concerned with environmental sustainability and product quality assurance.
Personal care and cosmetics industries constitute another major market segment for high-purity dodecyl acid products. The compound functions as an emulsifier, surfactant, and stabilizing agent in premium skincare formulations, hair care products, and luxury cosmetics. Consumer preferences for high-quality personal care products with minimal impurities have intensified the demand for pharmaceutical-grade dodecyl acid, particularly in anti-aging creams, moisturizers, and specialty hair treatments.
The specialty chemicals sector demonstrates growing appetite for ultra-pure dodecyl acid in advanced material synthesis and catalytic processes. Applications include the production of high-performance polymers, specialty coatings, and advanced lubricants where even trace impurities can significantly impact final product performance. This market segment particularly values suppliers who can provide comprehensive analytical documentation, including detailed NMR spectroscopic analysis confirming purity levels.
Food and beverage industries represent an emerging market opportunity for high-purity dodecyl acid products, primarily as food-grade emulsifiers and processing aids. Regulatory compliance requirements in food applications demand exceptional purity standards and comprehensive analytical verification, creating opportunities for suppliers equipped with advanced analytical capabilities.
The market trend toward sustainable and bio-based dodecyl acid production has created additional demand for high-purity products derived from renewable feedstocks. Manufacturers focusing on green chemistry approaches while maintaining superior purity levels through rigorous analytical control are positioned to capture premium market segments increasingly concerned with environmental sustainability and product quality assurance.
Current NMR Purity Analysis Challenges and Limitations
Nuclear Magnetic Resonance spectroscopy faces significant technical constraints when applied to dodecyl acid purity analysis, primarily stemming from the inherent limitations of conventional NMR methodologies. The quantitative accuracy of NMR measurements is fundamentally challenged by signal overlap phenomena, where structurally similar impurities exhibit chemical shifts closely resembling those of dodecyl acid protons. This spectral congestion becomes particularly problematic in the aliphatic region between 0.8-2.5 ppm, where both the target compound and common fatty acid contaminants display overlapping multiplets.
Sample preparation protocols present another critical bottleneck in achieving reliable purity assessments. Dodecyl acid's amphiphilic nature leads to aggregation behavior in solution, resulting in broadened spectral lines and reduced resolution. The selection of appropriate deuterated solvents becomes crucial, as polar solvents may induce hydrogen bonding effects that alter chemical shift patterns, while non-polar solvents may inadequately dissolve ionic impurities present in commercial samples.
Quantitative integration accuracy represents a persistent challenge in NMR-based purity determination. The relaxation time differences between dodecyl acid and various impurities can lead to systematic errors in peak area calculations. Short relaxation delays, commonly employed to reduce acquisition time, may result in incomplete T1 recovery, causing underestimation of certain components and skewing purity calculations.
Sensitivity limitations pose significant constraints when detecting trace-level impurities below 1% concentration. Standard proton NMR techniques often lack sufficient signal-to-noise ratios for reliable quantification of minor components, particularly when these impurities possess fewer observable protons compared to the main dodecyl acid structure. This sensitivity gap creates blind spots in comprehensive purity assessment protocols.
Temperature-dependent effects introduce additional complexity to dodecyl acid analysis. Fatty acid chains exhibit conformational flexibility that varies with temperature, leading to dynamic averaging of NMR signals. These thermal effects can mask or artificially enhance certain impurity signals, compromising the reproducibility of purity measurements across different analytical conditions.
Matrix effects from residual synthesis catalysts, metal contaminants, or processing aids further complicate spectral interpretation. These species may cause paramagnetic broadening, chemical shift perturbations, or introduce additional peaks that interfere with accurate integration of dodecyl acid signals, ultimately limiting the precision of purity determinations through conventional NMR approaches.
Sample preparation protocols present another critical bottleneck in achieving reliable purity assessments. Dodecyl acid's amphiphilic nature leads to aggregation behavior in solution, resulting in broadened spectral lines and reduced resolution. The selection of appropriate deuterated solvents becomes crucial, as polar solvents may induce hydrogen bonding effects that alter chemical shift patterns, while non-polar solvents may inadequately dissolve ionic impurities present in commercial samples.
Quantitative integration accuracy represents a persistent challenge in NMR-based purity determination. The relaxation time differences between dodecyl acid and various impurities can lead to systematic errors in peak area calculations. Short relaxation delays, commonly employed to reduce acquisition time, may result in incomplete T1 recovery, causing underestimation of certain components and skewing purity calculations.
Sensitivity limitations pose significant constraints when detecting trace-level impurities below 1% concentration. Standard proton NMR techniques often lack sufficient signal-to-noise ratios for reliable quantification of minor components, particularly when these impurities possess fewer observable protons compared to the main dodecyl acid structure. This sensitivity gap creates blind spots in comprehensive purity assessment protocols.
Temperature-dependent effects introduce additional complexity to dodecyl acid analysis. Fatty acid chains exhibit conformational flexibility that varies with temperature, leading to dynamic averaging of NMR signals. These thermal effects can mask or artificially enhance certain impurity signals, compromising the reproducibility of purity measurements across different analytical conditions.
Matrix effects from residual synthesis catalysts, metal contaminants, or processing aids further complicate spectral interpretation. These species may cause paramagnetic broadening, chemical shift perturbations, or introduce additional peaks that interfere with accurate integration of dodecyl acid signals, ultimately limiting the precision of purity determinations through conventional NMR approaches.
Existing NMR Methods for Dodecyl Acid Purity Assessment
01 Purification methods using distillation techniques
Various distillation methods can be employed to purify dodecyl acid, including vacuum distillation, molecular distillation, and fractional distillation. These techniques separate dodecyl acid from impurities based on differences in boiling points and volatility. The distillation process can effectively remove lower and higher molecular weight contaminants, resulting in high-purity dodecyl acid suitable for industrial applications.- Purification methods using distillation techniques: Various distillation methods can be employed to purify dodecyl acid, including vacuum distillation, molecular distillation, and fractional distillation. These techniques separate dodecyl acid from impurities based on differences in boiling points and volatility. The distillation process can effectively remove lower and higher molecular weight contaminants, resulting in high-purity dodecyl acid suitable for industrial applications.
- Crystallization and recrystallization processes: Crystallization methods can be utilized to enhance the purity of dodecyl acid by exploiting differences in solubility. The process involves dissolving crude dodecyl acid in appropriate solvents and controlling temperature to induce crystallization of pure product while impurities remain in solution. Multiple recrystallization cycles can further improve purity levels, achieving pharmaceutical or cosmetic grade specifications.
- Chemical treatment and neutralization methods: Chemical purification approaches involve treating crude dodecyl acid with various reagents to remove specific impurities. This may include acid-base neutralization, oxidation treatments, or reaction with selective agents that bind to contaminants. These methods can target specific impurity types such as unsaturated compounds, color bodies, or residual catalysts from synthesis processes.
- Chromatographic separation techniques: Advanced chromatographic methods including column chromatography and preparative liquid chromatography can achieve high-purity dodecyl acid separation. These techniques separate components based on differential interactions with stationary and mobile phases, allowing precise removal of structurally similar impurities. Chromatographic purification is particularly effective for obtaining analytical grade or research-quality dodecyl acid.
- Extraction and solvent-based purification: Liquid-liquid extraction and solvent washing techniques can selectively remove impurities from dodecyl acid based on differential solubility. The process involves choosing appropriate solvent systems that preferentially dissolve either the desired product or the impurities. Multiple extraction stages with different solvents can progressively increase purity, and subsequent solvent removal yields purified dodecyl acid.
02 Crystallization and recrystallization processes
Crystallization methods can be utilized to enhance the purity of dodecyl acid by exploiting differences in solubility. The process involves dissolving crude dodecyl acid in appropriate solvents and controlling temperature to induce crystallization of pure product while impurities remain in solution. Multiple recrystallization cycles can further improve purity levels, achieving pharmaceutical or cosmetic grade specifications.Expand Specific Solutions03 Chemical treatment and neutralization methods
Chemical purification approaches involve treating crude dodecyl acid with various reagents to remove specific impurities. This may include acid-base neutralization, oxidation treatments, or reaction with selective agents that bind to contaminants. These methods can target specific impurity types such as unsaturated compounds, color bodies, or residual catalysts from synthesis processes.Expand Specific Solutions04 Chromatographic separation techniques
Advanced chromatographic methods including column chromatography and preparative liquid chromatography can achieve high-purity dodecyl acid separation. These techniques separate components based on differential interactions with stationary and mobile phases, allowing precise removal of structurally similar impurities. Chromatographic purification is particularly effective for obtaining analytical grade or research grade dodecyl acid with purity exceeding standard requirements.Expand Specific Solutions05 Extraction and solvent-based purification
Liquid-liquid extraction and solvent washing techniques can selectively remove impurities from dodecyl acid based on differential solubility. The process involves selecting appropriate solvent systems that preferentially dissolve either the desired product or the impurities. Multiple extraction stages with different solvents can progressively improve purity, and the method can be combined with other purification techniques for optimal results.Expand Specific Solutions
Key Players in NMR Equipment and Chemical Analysis
The competitive landscape for evaluating dodecyl acid purity using NMR spectroscopy reflects a mature analytical chemistry market within the broader pharmaceutical and chemical industries. The industry is in an established phase, with NMR spectroscopy being a well-validated analytical technique for purity assessment. Market size is substantial, driven by stringent regulatory requirements for pharmaceutical intermediates and chemical products. Technology maturity is high, with major pharmaceutical companies like Pfizer, Eli Lilly, Genentech, and Roche possessing advanced analytical capabilities and established NMR protocols. Chemical manufacturers such as Osaka Organic Chemical Industry and Idemitsu Kosan have developed specialized expertise in organic compound analysis. The competitive advantage lies in method optimization, automation, and integration with quality control systems rather than fundamental technological breakthroughs, indicating a consolidated market where established players leverage their analytical infrastructure and regulatory compliance expertise.
Osaka Organic Chemical Industry Ltd.
Technical Solution: Specializes in advanced NMR spectroscopy techniques for fatty acid analysis, including dodecyl acid purity evaluation. Their methodology involves high-resolution 1H-NMR and 13C-NMR spectroscopy with specialized sample preparation protocols for accurate quantitative analysis. The company has developed proprietary integration methods and reference standards specifically for long-chain fatty acid purity assessment, enabling detection of impurities at sub-percent levels through careful peak integration and chemical shift analysis.
Strengths: Specialized expertise in organic chemical analysis with established NMR protocols. Weaknesses: Limited to traditional NMR approaches without advanced automation capabilities.
Astellas Pharma, Inc.
Technical Solution: Develops advanced NMR analytical methods for pharmaceutical raw material purity assessment, including fatty acid derivatives. Their technical approach involves quantitative 1H-NMR spectroscopy with carefully optimized acquisition parameters and integration procedures. The company maintains sophisticated analytical laboratories with high-field NMR instruments and employs standardized protocols for sample preparation, measurement, and data interpretation to ensure reliable purity determination results.
Strengths: Pharmaceutical-grade analytical capabilities with standardized protocols and high-quality instrumentation. Weaknesses: Limited specialization in non-pharmaceutical fatty acid applications and potential cost constraints for routine analysis.
Core NMR Techniques for Fatty Acid Characterization
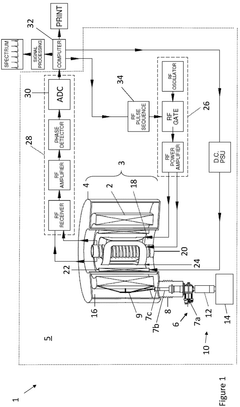
Nuclear magnetic resonance system
PatentActiveGB2597439A
Innovation
- A compact nuclear magnetic resonance system utilizing a superconducting magnetic coil cooled by a cryocooler, eliminating the need for liquid helium and achieving medium-to-high magnetic field strengths, allowing for a compact design suitable for laboratory benches.
Quality Standards and Regulations for Dodecyl Acid
The regulatory landscape for dodecyl acid quality standards encompasses multiple international and regional frameworks that directly impact NMR spectroscopy-based purity evaluation methods. The International Organization for Standardization (ISO) provides foundational guidelines through ISO 9001 quality management systems and ISO/IEC 17025 for testing and calibration laboratories, establishing the framework within which analytical methods like NMR must operate.
The United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) set specific monograph requirements for fatty acids including dodecyl acid when used in pharmaceutical applications. These compendial standards typically specify minimum purity levels of 95-99% and outline acceptable analytical methods for verification. While traditional methods like gas chromatography are commonly referenced, NMR spectroscopy is increasingly recognized as a complementary technique for structural confirmation and quantitative analysis.
The Food and Drug Administration (FDA) and European Medicines Agency (EMA) regulate dodecyl acid quality when used as pharmaceutical excipients or active ingredients. FDA's Current Good Manufacturing Practice (cGMP) regulations require validated analytical methods with demonstrated accuracy, precision, and specificity. NMR-based purity assessment must meet these validation criteria, including linearity ranges, detection limits, and robustness parameters.
Industrial applications fall under different regulatory frameworks, including REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) in Europe and TSCA (Toxic Substances Control Act) in the United States. These regulations mandate comprehensive chemical characterization data, where NMR spectroscopy provides valuable structural and purity information supporting regulatory submissions.
The American Society for Testing and Materials (ASTM) has developed specific standards for fatty acid analysis, including ASTM D5555 for fatty acid methyl esters and ASTM D6584 for biodiesel applications. While these standards primarily focus on chromatographic methods, they establish purity criteria and analytical performance requirements that NMR methods must meet or exceed.
Quality control laboratories implementing NMR-based dodecyl acid purity evaluation must comply with Good Laboratory Practice (GLP) regulations when supporting regulatory submissions. This includes maintaining detailed documentation, instrument qualification protocols, method validation studies, and analyst training records that demonstrate the reliability and reproducibility of NMR analytical results.
The United States Pharmacopeia (USP) and European Pharmacopoeia (Ph. Eur.) set specific monograph requirements for fatty acids including dodecyl acid when used in pharmaceutical applications. These compendial standards typically specify minimum purity levels of 95-99% and outline acceptable analytical methods for verification. While traditional methods like gas chromatography are commonly referenced, NMR spectroscopy is increasingly recognized as a complementary technique for structural confirmation and quantitative analysis.
The Food and Drug Administration (FDA) and European Medicines Agency (EMA) regulate dodecyl acid quality when used as pharmaceutical excipients or active ingredients. FDA's Current Good Manufacturing Practice (cGMP) regulations require validated analytical methods with demonstrated accuracy, precision, and specificity. NMR-based purity assessment must meet these validation criteria, including linearity ranges, detection limits, and robustness parameters.
Industrial applications fall under different regulatory frameworks, including REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) in Europe and TSCA (Toxic Substances Control Act) in the United States. These regulations mandate comprehensive chemical characterization data, where NMR spectroscopy provides valuable structural and purity information supporting regulatory submissions.
The American Society for Testing and Materials (ASTM) has developed specific standards for fatty acid analysis, including ASTM D5555 for fatty acid methyl esters and ASTM D6584 for biodiesel applications. While these standards primarily focus on chromatographic methods, they establish purity criteria and analytical performance requirements that NMR methods must meet or exceed.
Quality control laboratories implementing NMR-based dodecyl acid purity evaluation must comply with Good Laboratory Practice (GLP) regulations when supporting regulatory submissions. This includes maintaining detailed documentation, instrument qualification protocols, method validation studies, and analyst training records that demonstrate the reliability and reproducibility of NMR analytical results.
Cost-Benefit Analysis of NMR vs Alternative Methods
The economic evaluation of NMR spectroscopy versus alternative analytical methods for dodecyl acid purity assessment reveals significant variations in both initial investment requirements and operational expenditures. NMR systems typically demand substantial capital investment, with high-field instruments ranging from $300,000 to over $1 million, while alternative methods such as gas chromatography or high-performance liquid chromatography require considerably lower initial investments of $50,000 to $200,000.
Operational cost analysis demonstrates that NMR spectroscopy offers superior cost-effectiveness in high-throughput environments. The method eliminates sample preparation requirements, reducing reagent consumption and labor costs by approximately 40-60% compared to chromatographic techniques. Additionally, NMR analysis provides simultaneous quantification of multiple impurities and structural confirmation within a single measurement, effectively replacing multiple analytical procedures and reducing overall testing time from hours to minutes.
Alternative methods present lower barrier-to-entry costs but incur higher per-sample expenses due to consumable requirements, column replacements, and extensive sample preparation protocols. Gas chromatography methods for fatty acid analysis typically require derivatization procedures, increasing both material costs and analysis time. HPLC approaches necessitate continuous solvent consumption and regular column maintenance, contributing to recurring operational expenses.
The return on investment calculation favors NMR spectroscopy for laboratories processing more than 200 samples monthly. The break-even point typically occurs within 18-24 months for high-volume facilities, considering reduced labor requirements, eliminated consumables, and enhanced analytical throughput. Furthermore, NMR systems demonstrate exceptional longevity with minimal maintenance requirements, often operating effectively for 15-20 years with proper care.
Risk assessment indicates that NMR spectroscopy provides superior analytical reliability with minimal method development requirements across different dodecyl acid formulations. Alternative methods may require extensive validation and optimization for each sample matrix, increasing both development costs and time-to-market considerations. The non-destructive nature of NMR analysis also eliminates sample loss, providing additional economic value in research and development applications where sample quantities may be limited.
Operational cost analysis demonstrates that NMR spectroscopy offers superior cost-effectiveness in high-throughput environments. The method eliminates sample preparation requirements, reducing reagent consumption and labor costs by approximately 40-60% compared to chromatographic techniques. Additionally, NMR analysis provides simultaneous quantification of multiple impurities and structural confirmation within a single measurement, effectively replacing multiple analytical procedures and reducing overall testing time from hours to minutes.
Alternative methods present lower barrier-to-entry costs but incur higher per-sample expenses due to consumable requirements, column replacements, and extensive sample preparation protocols. Gas chromatography methods for fatty acid analysis typically require derivatization procedures, increasing both material costs and analysis time. HPLC approaches necessitate continuous solvent consumption and regular column maintenance, contributing to recurring operational expenses.
The return on investment calculation favors NMR spectroscopy for laboratories processing more than 200 samples monthly. The break-even point typically occurs within 18-24 months for high-volume facilities, considering reduced labor requirements, eliminated consumables, and enhanced analytical throughput. Furthermore, NMR systems demonstrate exceptional longevity with minimal maintenance requirements, often operating effectively for 15-20 years with proper care.
Risk assessment indicates that NMR spectroscopy provides superior analytical reliability with minimal method development requirements across different dodecyl acid formulations. Alternative methods may require extensive validation and optimization for each sample matrix, increasing both development costs and time-to-market considerations. The non-destructive nature of NMR analysis also eliminates sample loss, providing additional economic value in research and development applications where sample quantities may be limited.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!