Evaluate Sodium Nitrate CO2 Capture Potential in Combustion
MAR 3, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sodium Nitrate CO2 Capture Background and Objectives
Carbon dioxide capture and storage technologies have emerged as critical components in global climate change mitigation strategies, with industrial combustion processes representing one of the largest sources of anthropogenic CO2 emissions. Traditional carbon capture methods, including post-combustion amine-based absorption and pre-combustion gasification, face significant challenges related to energy penalties, equipment corrosion, and operational costs that can reach 15-30% of plant efficiency.
The exploration of alternative CO2 capture mechanisms has led researchers to investigate the potential of inorganic salt systems, particularly sodium nitrate, as novel capture agents in high-temperature combustion environments. Sodium nitrate presents unique thermochemical properties that may enable direct CO2 capture during combustion processes, potentially offering advantages over conventional post-combustion capture approaches.
Historical development of CO2 capture technologies began with industrial gas separation processes in the 1930s, evolving through petroleum refining applications to modern power plant implementations. The concept of using molten salt systems for CO2 capture emerged in the 1990s, driven by advances in concentrated solar power technologies and high-temperature chemical processing. Recent research has identified sodium nitrate's potential for simultaneous heat transfer and chemical absorption applications.
The primary objective of evaluating sodium nitrate CO2 capture potential centers on determining the feasibility of integrating this technology into existing combustion systems while maintaining operational efficiency. Key technical goals include quantifying CO2 absorption capacity under varying temperature and pressure conditions, establishing optimal operating parameters for maximum capture efficiency, and assessing the regeneration characteristics of sodium nitrate systems.
Secondary objectives encompass evaluating the economic viability compared to established capture technologies, determining compatibility with different fuel types and combustion configurations, and assessing environmental impacts including lifecycle carbon footprint analysis. The research aims to establish whether sodium nitrate-based systems can achieve capture rates exceeding 85% while maintaining energy penalties below 20% of total plant output.
Long-term strategic objectives involve developing scalable implementation pathways for industrial adoption, identifying potential synergies with existing thermal management systems, and establishing the foundation for next-generation integrated combustion-capture technologies that could revolutionize carbon management in power generation and industrial processes.
The exploration of alternative CO2 capture mechanisms has led researchers to investigate the potential of inorganic salt systems, particularly sodium nitrate, as novel capture agents in high-temperature combustion environments. Sodium nitrate presents unique thermochemical properties that may enable direct CO2 capture during combustion processes, potentially offering advantages over conventional post-combustion capture approaches.
Historical development of CO2 capture technologies began with industrial gas separation processes in the 1930s, evolving through petroleum refining applications to modern power plant implementations. The concept of using molten salt systems for CO2 capture emerged in the 1990s, driven by advances in concentrated solar power technologies and high-temperature chemical processing. Recent research has identified sodium nitrate's potential for simultaneous heat transfer and chemical absorption applications.
The primary objective of evaluating sodium nitrate CO2 capture potential centers on determining the feasibility of integrating this technology into existing combustion systems while maintaining operational efficiency. Key technical goals include quantifying CO2 absorption capacity under varying temperature and pressure conditions, establishing optimal operating parameters for maximum capture efficiency, and assessing the regeneration characteristics of sodium nitrate systems.
Secondary objectives encompass evaluating the economic viability compared to established capture technologies, determining compatibility with different fuel types and combustion configurations, and assessing environmental impacts including lifecycle carbon footprint analysis. The research aims to establish whether sodium nitrate-based systems can achieve capture rates exceeding 85% while maintaining energy penalties below 20% of total plant output.
Long-term strategic objectives involve developing scalable implementation pathways for industrial adoption, identifying potential synergies with existing thermal management systems, and establishing the foundation for next-generation integrated combustion-capture technologies that could revolutionize carbon management in power generation and industrial processes.
Market Demand for Industrial CO2 Capture Solutions
The global industrial CO2 capture market is experiencing unprecedented growth driven by stringent environmental regulations and corporate sustainability commitments. Major industrial sectors including power generation, cement production, steel manufacturing, and petrochemicals are actively seeking cost-effective carbon capture solutions to meet emission reduction targets. The urgency has intensified as governments worldwide implement carbon pricing mechanisms and establish net-zero emission deadlines.
Power generation facilities represent the largest market segment for CO2 capture technologies, particularly coal and natural gas-fired plants that require retrofitting solutions. The cement industry faces unique challenges due to process emissions inherent in limestone calcination, creating substantial demand for innovative capture approaches. Steel production facilities are increasingly exploring carbon capture integration as part of decarbonization strategies, while refineries and petrochemical plants seek solutions that can handle varying flue gas compositions.
Current market dynamics reveal a strong preference for post-combustion capture technologies due to their retrofit compatibility with existing infrastructure. Industrial operators prioritize solutions offering high capture efficiency, low energy penalties, and minimal operational disruption. The economic viability remains a critical factor, with industries seeking technologies that can achieve capture costs below established thresholds while maintaining process reliability.
Emerging market drivers include carbon credit monetization opportunities and the development of carbon utilization pathways. Industries are increasingly viewing captured CO2 as a potential revenue stream rather than merely a compliance cost. This shift is creating demand for capture technologies that can deliver high-purity CO2 suitable for various utilization applications including enhanced oil recovery, chemical synthesis, and permanent geological storage.
The market landscape shows growing interest in novel sorbent materials and process intensification approaches that can reduce both capital and operational expenditures. Industrial stakeholders are particularly attracted to technologies offering modular deployment options, enabling phased implementation aligned with capital investment cycles and regulatory timelines.
Regional market variations reflect different policy frameworks and industrial structures. European markets emphasize compliance-driven adoption, while North American markets focus on economic incentives and carbon credit mechanisms. Asian markets, particularly China and India, represent significant growth opportunities driven by rapid industrial expansion and evolving environmental policies.
Power generation facilities represent the largest market segment for CO2 capture technologies, particularly coal and natural gas-fired plants that require retrofitting solutions. The cement industry faces unique challenges due to process emissions inherent in limestone calcination, creating substantial demand for innovative capture approaches. Steel production facilities are increasingly exploring carbon capture integration as part of decarbonization strategies, while refineries and petrochemical plants seek solutions that can handle varying flue gas compositions.
Current market dynamics reveal a strong preference for post-combustion capture technologies due to their retrofit compatibility with existing infrastructure. Industrial operators prioritize solutions offering high capture efficiency, low energy penalties, and minimal operational disruption. The economic viability remains a critical factor, with industries seeking technologies that can achieve capture costs below established thresholds while maintaining process reliability.
Emerging market drivers include carbon credit monetization opportunities and the development of carbon utilization pathways. Industries are increasingly viewing captured CO2 as a potential revenue stream rather than merely a compliance cost. This shift is creating demand for capture technologies that can deliver high-purity CO2 suitable for various utilization applications including enhanced oil recovery, chemical synthesis, and permanent geological storage.
The market landscape shows growing interest in novel sorbent materials and process intensification approaches that can reduce both capital and operational expenditures. Industrial stakeholders are particularly attracted to technologies offering modular deployment options, enabling phased implementation aligned with capital investment cycles and regulatory timelines.
Regional market variations reflect different policy frameworks and industrial structures. European markets emphasize compliance-driven adoption, while North American markets focus on economic incentives and carbon credit mechanisms. Asian markets, particularly China and India, represent significant growth opportunities driven by rapid industrial expansion and evolving environmental policies.
Current Status of Sodium Nitrate Capture Technologies
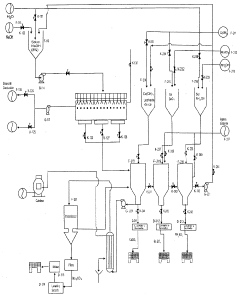
Sodium nitrate-based CO2 capture technologies are currently in the early stages of development, with limited commercial deployment compared to established amine-based systems. The technology primarily operates through thermochemical looping processes, where sodium nitrate acts as an oxygen carrier that can facilitate CO2 separation from flue gases in combustion environments.
Current research efforts focus on molten salt systems incorporating sodium nitrate as a key component for high-temperature CO2 capture applications. These systems typically operate at temperatures ranging from 500°C to 800°C, making them particularly suitable for integration with coal-fired power plants and industrial furnaces. The molten salt mixture often combines sodium nitrate with other alkali compounds to optimize capture efficiency and thermal stability.
Laboratory-scale demonstrations have shown promising results, with CO2 capture rates achieving 85-92% efficiency under controlled conditions. However, these systems face significant technical challenges including material corrosion, salt degradation at high temperatures, and complex heat integration requirements. The regeneration process requires substantial energy input, typically consuming 15-20% more energy than conventional amine-based systems.
Several pilot projects are currently underway in China, the United States, and Europe, though most remain at the 1-10 MW scale. The technology's integration with existing combustion infrastructure presents both opportunities and challenges, as retrofitting requires substantial modifications to flue gas handling systems and the addition of specialized molten salt circulation equipment.
The current technological readiness level for sodium nitrate CO2 capture systems is estimated at TRL 4-5, indicating that while basic functionality has been demonstrated, significant engineering challenges remain before commercial viability. Key limitations include the need for specialized materials to withstand corrosive molten salt environments, complex process control systems, and higher capital costs compared to established capture technologies.
Recent developments have focused on improving salt composition formulations and developing more efficient heat recovery systems to reduce the overall energy penalty. However, long-term operational data remains limited, and questions persist regarding the technology's economic competitiveness in large-scale deployment scenarios.
Current research efforts focus on molten salt systems incorporating sodium nitrate as a key component for high-temperature CO2 capture applications. These systems typically operate at temperatures ranging from 500°C to 800°C, making them particularly suitable for integration with coal-fired power plants and industrial furnaces. The molten salt mixture often combines sodium nitrate with other alkali compounds to optimize capture efficiency and thermal stability.
Laboratory-scale demonstrations have shown promising results, with CO2 capture rates achieving 85-92% efficiency under controlled conditions. However, these systems face significant technical challenges including material corrosion, salt degradation at high temperatures, and complex heat integration requirements. The regeneration process requires substantial energy input, typically consuming 15-20% more energy than conventional amine-based systems.
Several pilot projects are currently underway in China, the United States, and Europe, though most remain at the 1-10 MW scale. The technology's integration with existing combustion infrastructure presents both opportunities and challenges, as retrofitting requires substantial modifications to flue gas handling systems and the addition of specialized molten salt circulation equipment.
The current technological readiness level for sodium nitrate CO2 capture systems is estimated at TRL 4-5, indicating that while basic functionality has been demonstrated, significant engineering challenges remain before commercial viability. Key limitations include the need for specialized materials to withstand corrosive molten salt environments, complex process control systems, and higher capital costs compared to established capture technologies.
Recent developments have focused on improving salt composition formulations and developing more efficient heat recovery systems to reduce the overall energy penalty. However, long-term operational data remains limited, and questions persist regarding the technology's economic competitiveness in large-scale deployment scenarios.
Existing Sodium-Based CO2 Capture Solutions
01 Molten salt systems using sodium nitrate for CO2 capture
Molten salt systems incorporating sodium nitrate as a key component can be utilized for carbon dioxide capture applications. These systems leverage the thermal and chemical properties of sodium nitrate in high-temperature environments to facilitate CO2 absorption and separation. The molten salt medium provides advantages in terms of thermal stability and reaction kinetics for carbon capture processes.- Molten salt systems using sodium nitrate for CO2 capture: Molten salt systems incorporating sodium nitrate can be utilized for carbon dioxide capture applications. These systems leverage the thermal properties and chemical reactivity of sodium nitrate in molten state to absorb and separate CO2 from gas streams. The molten salt approach offers advantages in high-temperature industrial processes where conventional capture methods may be less effective.
- Sodium nitrate as component in chemical looping combustion for CO2 separation: Chemical looping combustion processes can employ sodium nitrate as an oxygen carrier or reactive component to facilitate carbon dioxide separation. This approach enables inherent CO2 capture during combustion by utilizing the oxidation-reduction properties of sodium-based compounds. The method provides a pathway for producing concentrated CO2 streams suitable for sequestration or utilization.
- Sodium nitrate in carbonate-based CO2 absorption systems: Carbonate-based absorption systems can incorporate sodium nitrate to enhance carbon dioxide capture efficiency. The interaction between sodium compounds and carbonate chemistry creates favorable conditions for CO2 absorption and regeneration cycles. These systems can be applied in post-combustion capture scenarios where flue gas treatment is required.
- Electrochemical CO2 capture methods utilizing sodium nitrate electrolytes: Electrochemical approaches for carbon dioxide capture can utilize sodium nitrate as an electrolyte component. These methods employ electrical potential to drive CO2 absorption and release cycles, offering potential energy efficiency advantages. The electrochemical route enables modular and scalable capture systems with controllable operation parameters.
- Integrated sodium nitrate systems for industrial CO2 capture and utilization: Integrated systems combine sodium nitrate-based capture technologies with downstream CO2 utilization processes. These approaches aim to create circular carbon economies by capturing emissions and converting them into valuable products. The integration of capture and utilization reduces overall process costs and environmental impact while providing economic incentives for implementation.
02 Sodium nitrate in carbonate-based CO2 capture processes
Sodium nitrate can be employed in carbonate-based systems for enhancing carbon dioxide capture efficiency. The compound participates in chemical reactions that promote CO2 absorption through carbonate formation and regeneration cycles. This approach utilizes the reactivity of sodium nitrate to improve the overall capture capacity and reduce energy requirements in the process.Expand Specific Solutions03 Electrochemical CO2 capture using sodium nitrate electrolytes
Electrochemical methods for carbon dioxide capture utilize sodium nitrate as an electrolyte component to facilitate CO2 conversion and separation. The electrochemical approach enables selective capture through redox reactions and ionic transport mechanisms. This technology offers potential for integration with renewable energy sources and provides controllable capture rates.Expand Specific Solutions04 Sodium nitrate as additive in solid sorbent CO2 capture
Solid sorbent materials can be enhanced with sodium nitrate additives to improve their carbon dioxide capture performance. The addition of this compound modifies the surface properties and chemical reactivity of the sorbent, leading to increased CO2 adsorption capacity and faster kinetics. The modified sorbents demonstrate improved stability and regeneration characteristics over multiple capture cycles.Expand Specific Solutions05 Hybrid systems combining sodium nitrate with other capture technologies
Integrated capture systems combine sodium nitrate-based processes with complementary technologies to achieve enhanced CO2 removal efficiency. These hybrid approaches leverage synergistic effects between different capture mechanisms, such as chemical absorption, physical adsorption, and membrane separation. The combination allows for optimization of capture performance across varying operating conditions and gas compositions.Expand Specific Solutions
Major Players in CO2 Capture Industry
The sodium nitrate CO2 capture technology represents an emerging field within the broader carbon capture landscape, currently in early-stage development with significant research momentum from academic institutions. Leading research entities including Huazhong University of Science & Technology, Michigan Technological University, University of Seville, and King Fahd University of Petroleum & Minerals are driving fundamental research, while industrial players like Air Liquide SA, Deepak Nitrite Ltd., and China National Coal Group Corp. provide manufacturing capabilities and application expertise. The technology maturity remains nascent, with most developments concentrated in laboratory-scale studies and proof-of-concept demonstrations. Market potential appears substantial given the growing carbon capture sector, though commercial viability requires further technological advancement and cost optimization to compete with established CO2 capture methods.
Air Liquide SA
Technical Solution: Air Liquide has developed advanced sodium nitrate-based CO2 capture systems that leverage the thermal decomposition properties of sodium nitrate in high-temperature combustion environments. Their technology utilizes a cyclic process where sodium nitrate acts as both an oxygen carrier and CO2 sorbent, operating at temperatures between 600-900°C. The process involves the reduction of sodium nitrate to sodium nitrite in the presence of fuel, followed by regeneration with air to release concentrated CO2 streams. This approach achieves CO2 capture efficiencies of up to 85% while maintaining combustion stability through controlled oxygen release from the nitrate decomposition.
Strengths: High-temperature stability, dual functionality as oxygen carrier and CO2 sorbent, proven industrial scalability. Weaknesses: Energy-intensive regeneration cycles, potential NOx formation, material degradation over multiple cycles.
Battelle Memorial Institute
Technical Solution: Battelle has developed an innovative sodium nitrate-enhanced combustion system that combines chemical looping combustion with CO2 capture. Their technology uses sodium nitrate as an oxygen carrier in a dual fluidized bed reactor system where fuel combustion occurs in a fuel reactor with sodium nitrate providing oxygen, while air regeneration happens in a separate air reactor. This process inherently separates CO2 from nitrogen, producing a concentrated CO2 stream without additional separation steps. The system operates at 800-950°C and achieves complete fuel conversion with CO2 purities exceeding 95%. The technology also demonstrates excellent cycling stability with minimal sodium nitrate makeup requirements.
Strengths: Inherent CO2 separation, high CO2 purity, excellent cycling stability, no additional separation equipment needed. Weaknesses: High operating temperatures, complex reactor design, potential agglomeration issues, limited fuel flexibility.
Core Patents in Nitrate CO2 Capture Systems
Method and composition for carbon capture and storage
PatentPendingUS20250339814A1
Innovation
- The use of fully carbonated amines, such as amine carbamates, as promoters to enhance the reactive absorption of CO2 into alkali metal carbonates, particularly sodium carbonate, by forming non-volatile carbamate ions that catalyze the carbonation process, even at low CO2 concentrations.
Process for the removal of acid gases from the air and from combustion gases from burners and internal combustion engines by means of absorption with sodium hydroxide solution and process for obtaining sodium carbonate in order to acquire carbon cred
PatentWO2011122925A1
Innovation
- A chemical process using a 2N sodium hydroxide solution to absorb acid gases in a horizontal absorber, converting CO2 into sodium carbonate, which can be further processed to produce sodium carbonate, thereby accrediting carbon credits through the capture and commercialization of CO2.
Carbon Emission Regulations and Policy Framework
The global regulatory landscape for carbon emissions has undergone significant transformation over the past two decades, establishing a comprehensive framework that directly impacts CO2 capture technologies including sodium nitrate-based solutions. The Paris Agreement of 2015 serves as the cornerstone of international climate policy, requiring signatory nations to implement nationally determined contributions (NDCs) that progressively reduce greenhouse gas emissions. This multilateral framework creates substantial market drivers for innovative capture technologies in combustion processes.
Regional regulatory approaches vary considerably in their implementation strategies and stringency levels. The European Union's Emissions Trading System (ETS) represents the world's largest carbon market, covering approximately 40% of the EU's greenhouse gas emissions from power generation and energy-intensive industries. Under this cap-and-trade mechanism, facilities exceeding emission allowances face significant financial penalties, creating economic incentives for deploying advanced capture technologies. The EU's Green Deal further intensifies these requirements, targeting carbon neutrality by 2050 and establishing interim reduction goals of 55% by 2030 compared to 1990 levels.
In the United States, regulatory frameworks operate through a combination of federal and state-level initiatives. The Clean Air Act provides federal authority for emission standards, while state-level programs like California's Cap-and-Trade Program and the Regional Greenhouse Gas Initiative (RGGI) in northeastern states establish mandatory emission reduction requirements for power plants and industrial facilities. These programs typically include provisions for emission offset credits, potentially benefiting novel capture technologies that demonstrate measurable CO2 reduction capabilities.
China's national ETS, launched in 2021, covers the power sector initially and plans expansion to additional industries including petrochemicals, chemicals, building materials, steel, non-ferrous metals, paper, and aviation. This system affects over 4 billion tons of CO2 annually, representing the world's largest carbon trading market by coverage. The regulatory framework emphasizes technology upgrades and efficiency improvements, creating opportunities for innovative capture solutions in combustion applications.
Emerging policy trends indicate increasing focus on technology-specific incentives and performance standards. Carbon capture, utilization, and storage (CCUS) technologies receive dedicated support through tax credits, research funding, and regulatory fast-tracking procedures in multiple jurisdictions. The U.S. 45Q tax credit provides up to $50 per ton of CO2 captured and permanently stored, while similar incentive structures exist in Canada, Norway, and the United Kingdom.
Industrial emission standards continue tightening across major economies, with specific attention to combustion processes in power generation, cement production, steel manufacturing, and chemical processing. These sectors represent primary application areas for sodium nitrate CO2 capture technologies, as regulatory compliance increasingly requires demonstrable emission reduction capabilities rather than mere efficiency improvements.
Regional regulatory approaches vary considerably in their implementation strategies and stringency levels. The European Union's Emissions Trading System (ETS) represents the world's largest carbon market, covering approximately 40% of the EU's greenhouse gas emissions from power generation and energy-intensive industries. Under this cap-and-trade mechanism, facilities exceeding emission allowances face significant financial penalties, creating economic incentives for deploying advanced capture technologies. The EU's Green Deal further intensifies these requirements, targeting carbon neutrality by 2050 and establishing interim reduction goals of 55% by 2030 compared to 1990 levels.
In the United States, regulatory frameworks operate through a combination of federal and state-level initiatives. The Clean Air Act provides federal authority for emission standards, while state-level programs like California's Cap-and-Trade Program and the Regional Greenhouse Gas Initiative (RGGI) in northeastern states establish mandatory emission reduction requirements for power plants and industrial facilities. These programs typically include provisions for emission offset credits, potentially benefiting novel capture technologies that demonstrate measurable CO2 reduction capabilities.
China's national ETS, launched in 2021, covers the power sector initially and plans expansion to additional industries including petrochemicals, chemicals, building materials, steel, non-ferrous metals, paper, and aviation. This system affects over 4 billion tons of CO2 annually, representing the world's largest carbon trading market by coverage. The regulatory framework emphasizes technology upgrades and efficiency improvements, creating opportunities for innovative capture solutions in combustion applications.
Emerging policy trends indicate increasing focus on technology-specific incentives and performance standards. Carbon capture, utilization, and storage (CCUS) technologies receive dedicated support through tax credits, research funding, and regulatory fast-tracking procedures in multiple jurisdictions. The U.S. 45Q tax credit provides up to $50 per ton of CO2 captured and permanently stored, while similar incentive structures exist in Canada, Norway, and the United Kingdom.
Industrial emission standards continue tightening across major economies, with specific attention to combustion processes in power generation, cement production, steel manufacturing, and chemical processing. These sectors represent primary application areas for sodium nitrate CO2 capture technologies, as regulatory compliance increasingly requires demonstrable emission reduction capabilities rather than mere efficiency improvements.
Environmental Impact Assessment of Nitrate Capture
The environmental impact assessment of sodium nitrate-based CO2 capture systems requires comprehensive evaluation across multiple ecological dimensions. Primary concerns center on the potential release of nitrogen compounds into atmospheric and aquatic systems during capture operations. Nitrate compounds, while naturally occurring, can contribute to eutrophication when concentrated in water bodies, potentially disrupting aquatic ecosystems and affecting biodiversity.
Atmospheric emissions represent another critical consideration, particularly regarding NOx formation during high-temperature combustion processes. The thermal decomposition of sodium nitrate at elevated temperatures may generate nitrogen oxides, which contribute to air quality degradation and ozone formation. These secondary pollutants pose risks to human health and environmental quality, potentially offsetting some benefits of CO2 capture.
Soil contamination risks emerge from potential system leakage or improper waste handling. Elevated sodium concentrations can alter soil chemistry, affecting plant growth and microbial communities. The accumulation of nitrate salts may lead to soil salinization, reducing agricultural productivity in affected areas and requiring long-term remediation efforts.
Water resource impacts demand careful monitoring, as sodium nitrate exhibits high solubility and mobility in aquatic environments. Groundwater contamination represents a persistent concern, particularly in regions with vulnerable aquifer systems. The compound's stability and persistence in water systems necessitate robust containment and treatment protocols.
Life cycle environmental impacts extend beyond operational phases to include raw material extraction, processing, and end-of-life disposal. Sodium nitrate production involves energy-intensive processes that generate their own carbon footprint, potentially reducing net CO2 capture benefits. Mining activities for sodium nitrate precursors can cause habitat disruption and landscape alteration.
Mitigation strategies must address these multifaceted environmental challenges through advanced containment systems, real-time monitoring technologies, and comprehensive waste management protocols. Environmental risk assessment frameworks should incorporate both acute and chronic exposure scenarios to ensure comprehensive protection of ecological systems and human health throughout the technology deployment lifecycle.
Atmospheric emissions represent another critical consideration, particularly regarding NOx formation during high-temperature combustion processes. The thermal decomposition of sodium nitrate at elevated temperatures may generate nitrogen oxides, which contribute to air quality degradation and ozone formation. These secondary pollutants pose risks to human health and environmental quality, potentially offsetting some benefits of CO2 capture.
Soil contamination risks emerge from potential system leakage or improper waste handling. Elevated sodium concentrations can alter soil chemistry, affecting plant growth and microbial communities. The accumulation of nitrate salts may lead to soil salinization, reducing agricultural productivity in affected areas and requiring long-term remediation efforts.
Water resource impacts demand careful monitoring, as sodium nitrate exhibits high solubility and mobility in aquatic environments. Groundwater contamination represents a persistent concern, particularly in regions with vulnerable aquifer systems. The compound's stability and persistence in water systems necessitate robust containment and treatment protocols.
Life cycle environmental impacts extend beyond operational phases to include raw material extraction, processing, and end-of-life disposal. Sodium nitrate production involves energy-intensive processes that generate their own carbon footprint, potentially reducing net CO2 capture benefits. Mining activities for sodium nitrate precursors can cause habitat disruption and landscape alteration.
Mitigation strategies must address these multifaceted environmental challenges through advanced containment systems, real-time monitoring technologies, and comprehensive waste management protocols. Environmental risk assessment frameworks should incorporate both acute and chronic exposure scenarios to ensure comprehensive protection of ecological systems and human health throughout the technology deployment lifecycle.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!