Evaluating Silicic Acid's Functionality in Nanocomposite Design
FEB 25, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Silicic Acid in Nanocomposites: Background and Objectives
Silicic acid, the hydrated form of silicon dioxide, has emerged as a critical functional component in advanced nanocomposite materials over the past two decades. Its unique molecular structure, characterized by silicon-oxygen bonds and hydroxyl groups, enables exceptional interfacial interactions with various polymer matrices and inorganic fillers. The evolution of silicic acid applications in nanocomposites traces back to early biomimetic research inspired by diatom frustules, where nature demonstrated silicon's remarkable ability to create hierarchical nanostructures with outstanding mechanical properties.
The development trajectory of silicic acid-based nanocomposites has been driven by increasing demands for materials that combine lightweight characteristics with superior mechanical strength, thermal stability, and functional versatility. Traditional composite materials often suffer from poor interfacial adhesion between organic and inorganic phases, leading to compromised performance. Silicic acid addresses this fundamental challenge through its amphiphilic nature, acting as a molecular bridge that enhances compatibility between dissimilar materials.
Current research objectives focus on systematically evaluating how silicic acid influences nanocomposite design parameters, including dispersion uniformity, interfacial bonding strength, and resultant material properties. Understanding the relationship between silicic acid concentration, polymerization state, and final composite performance remains a critical knowledge gap. The condensation behavior of silicic acid during composite formation significantly affects network formation and cross-linking density, directly impacting mechanical and thermal characteristics.
The primary technical goal involves establishing quantitative correlations between silicic acid's chemical state and nanocomposite functionality. This includes investigating optimal processing conditions, pH ranges, and silicic acid-to-matrix ratios that maximize performance benefits. Additionally, exploring synergistic effects when silicic acid is combined with other nanofillers represents a promising avenue for developing multifunctional composites with tailored properties for specific applications in aerospace, automotive, electronics, and biomedical fields.
The development trajectory of silicic acid-based nanocomposites has been driven by increasing demands for materials that combine lightweight characteristics with superior mechanical strength, thermal stability, and functional versatility. Traditional composite materials often suffer from poor interfacial adhesion between organic and inorganic phases, leading to compromised performance. Silicic acid addresses this fundamental challenge through its amphiphilic nature, acting as a molecular bridge that enhances compatibility between dissimilar materials.
Current research objectives focus on systematically evaluating how silicic acid influences nanocomposite design parameters, including dispersion uniformity, interfacial bonding strength, and resultant material properties. Understanding the relationship between silicic acid concentration, polymerization state, and final composite performance remains a critical knowledge gap. The condensation behavior of silicic acid during composite formation significantly affects network formation and cross-linking density, directly impacting mechanical and thermal characteristics.
The primary technical goal involves establishing quantitative correlations between silicic acid's chemical state and nanocomposite functionality. This includes investigating optimal processing conditions, pH ranges, and silicic acid-to-matrix ratios that maximize performance benefits. Additionally, exploring synergistic effects when silicic acid is combined with other nanofillers represents a promising avenue for developing multifunctional composites with tailored properties for specific applications in aerospace, automotive, electronics, and biomedical fields.
Market Demand for Silicic Acid-Based Nanocomposites
The global market for silicic acid-based nanocomposites is experiencing robust expansion driven by diverse industrial applications and evolving performance requirements. These materials have gained significant traction in sectors including coatings, adhesives, electronics, biomedical devices, and advanced packaging solutions. The demand is particularly pronounced in applications requiring enhanced mechanical strength, thermal stability, barrier properties, and biocompatibility, where silicic acid serves as a critical functional component in nanocomposite formulations.
In the coatings and surface treatment industry, silicic acid-based nanocomposites are increasingly sought after for their ability to provide superior scratch resistance, anti-corrosion properties, and self-cleaning functionalities. Automotive and architectural coating manufacturers are actively integrating these materials to meet stringent environmental regulations while delivering enhanced durability. Similarly, the electronics sector demonstrates growing interest in these nanocomposites for encapsulation materials, dielectric layers, and thermal management solutions, driven by miniaturization trends and the need for improved heat dissipation in high-performance devices.
The biomedical field represents another significant growth area, where silicic acid-based nanocomposites are valued for their biocompatibility and controlled degradation characteristics. Applications in drug delivery systems, tissue engineering scaffolds, and dental materials are expanding as regulatory frameworks become more accommodating of nanomaterial innovations. The pharmaceutical industry particularly recognizes the potential of these materials in developing targeted therapeutic systems with improved efficacy and reduced side effects.
Packaging industries are increasingly adopting silicic acid-enhanced nanocomposites to address sustainability concerns and functional performance demands. The materials offer improved barrier properties against moisture and gases, extending product shelf life while potentially reducing packaging thickness and material consumption. This aligns with circular economy initiatives and consumer preferences for environmentally responsible products.
Geographically, demand patterns show concentration in industrialized regions with strong manufacturing bases, particularly in Asia-Pacific, North America, and Europe. Emerging economies are demonstrating accelerated adoption rates as local industries upgrade their material technologies to compete in global markets. The market trajectory suggests sustained growth potential, contingent upon continued technological advancement, cost optimization, and successful demonstration of long-term performance benefits across application domains.
In the coatings and surface treatment industry, silicic acid-based nanocomposites are increasingly sought after for their ability to provide superior scratch resistance, anti-corrosion properties, and self-cleaning functionalities. Automotive and architectural coating manufacturers are actively integrating these materials to meet stringent environmental regulations while delivering enhanced durability. Similarly, the electronics sector demonstrates growing interest in these nanocomposites for encapsulation materials, dielectric layers, and thermal management solutions, driven by miniaturization trends and the need for improved heat dissipation in high-performance devices.
The biomedical field represents another significant growth area, where silicic acid-based nanocomposites are valued for their biocompatibility and controlled degradation characteristics. Applications in drug delivery systems, tissue engineering scaffolds, and dental materials are expanding as regulatory frameworks become more accommodating of nanomaterial innovations. The pharmaceutical industry particularly recognizes the potential of these materials in developing targeted therapeutic systems with improved efficacy and reduced side effects.
Packaging industries are increasingly adopting silicic acid-enhanced nanocomposites to address sustainability concerns and functional performance demands. The materials offer improved barrier properties against moisture and gases, extending product shelf life while potentially reducing packaging thickness and material consumption. This aligns with circular economy initiatives and consumer preferences for environmentally responsible products.
Geographically, demand patterns show concentration in industrialized regions with strong manufacturing bases, particularly in Asia-Pacific, North America, and Europe. Emerging economies are demonstrating accelerated adoption rates as local industries upgrade their material technologies to compete in global markets. The market trajectory suggests sustained growth potential, contingent upon continued technological advancement, cost optimization, and successful demonstration of long-term performance benefits across application domains.
Current Status and Challenges in Silicic Acid Functionalization
Silicic acid functionalization has emerged as a critical area in nanocomposite design, yet the field faces significant technical and practical challenges that constrain its widespread implementation. Current research demonstrates that while silicic acid offers unique advantages in terms of biocompatibility and structural versatility, achieving controlled functionalization remains problematic. The primary challenge stems from the inherent instability of silicic acid in aqueous solutions, where it readily undergoes condensation reactions leading to uncontrolled polymerization. This spontaneous oligomerization severely limits the window for effective surface modification and functional group attachment.
The geographical distribution of advanced research in this domain shows concentration in North America, Europe, and East Asia, with leading institutions focusing on stabilization strategies and controlled condensation mechanisms. However, a notable gap exists between laboratory-scale achievements and industrial-scale production capabilities. Current methodologies predominantly rely on pH manipulation, organic additives, and template-assisted approaches to temporarily stabilize silicic acid species, yet these methods often introduce contamination risks or require complex purification steps that compromise economic viability.
A major technical bottleneck involves the precise control of functionalization degree and spatial distribution of functional groups on silicic acid-derived structures. Existing analytical techniques struggle to provide real-time monitoring of the functionalization process, making reproducibility a persistent concern. The challenge intensifies when attempting to incorporate multiple functional groups simultaneously, as competitive reaction kinetics and steric hindrance effects create unpredictable outcomes. Furthermore, the transition from monomeric or low-oligomeric silicic acid to functionalized nanocomposite components often results in loss of desired properties due to premature condensation or incomplete functionalization.
Environmental and regulatory constraints add another layer of complexity, as many traditional functionalization agents involve hazardous solvents or generate problematic waste streams. The industry increasingly demands green chemistry approaches, yet sustainable alternatives frequently exhibit lower reactivity or selectivity. Additionally, scaling challenges persist regarding batch-to-batch consistency and cost-effectiveness, particularly when targeting specialized applications requiring precise functional group densities and distributions.
The geographical distribution of advanced research in this domain shows concentration in North America, Europe, and East Asia, with leading institutions focusing on stabilization strategies and controlled condensation mechanisms. However, a notable gap exists between laboratory-scale achievements and industrial-scale production capabilities. Current methodologies predominantly rely on pH manipulation, organic additives, and template-assisted approaches to temporarily stabilize silicic acid species, yet these methods often introduce contamination risks or require complex purification steps that compromise economic viability.
A major technical bottleneck involves the precise control of functionalization degree and spatial distribution of functional groups on silicic acid-derived structures. Existing analytical techniques struggle to provide real-time monitoring of the functionalization process, making reproducibility a persistent concern. The challenge intensifies when attempting to incorporate multiple functional groups simultaneously, as competitive reaction kinetics and steric hindrance effects create unpredictable outcomes. Furthermore, the transition from monomeric or low-oligomeric silicic acid to functionalized nanocomposite components often results in loss of desired properties due to premature condensation or incomplete functionalization.
Environmental and regulatory constraints add another layer of complexity, as many traditional functionalization agents involve hazardous solvents or generate problematic waste streams. The industry increasingly demands green chemistry approaches, yet sustainable alternatives frequently exhibit lower reactivity or selectivity. Additionally, scaling challenges persist regarding batch-to-batch consistency and cost-effectiveness, particularly when targeting specialized applications requiring precise functional group densities and distributions.
Existing Silicic Acid Functionalization Approaches
01 Silicic acid as a reinforcing filler in rubber and polymer compositions
Silicic acid, particularly precipitated silica and fumed silica, serves as an effective reinforcing filler in rubber compounds and polymer compositions. It enhances mechanical properties such as tensile strength, tear resistance, and abrasion resistance. The functionality of silicic acid in these applications depends on its surface area, particle size distribution, and surface chemistry. Modified silicic acids with specific functional groups can improve compatibility with polymer matrices and enhance dispersion characteristics.- Silicic acid as a reinforcing filler in rubber and elastomer compositions: Silicic acid, particularly precipitated silica, functions as a reinforcing filler in rubber compounds and elastomer compositions. It enhances mechanical properties such as tensile strength, tear resistance, and abrasion resistance. The silicic acid particles interact with polymer chains to improve the overall performance of rubber products including tires, seals, and industrial rubber goods.
- Silicic acid as an adsorbent and carrier material: Silicic acid exhibits excellent adsorption properties due to its high surface area and porous structure. It functions as a carrier material for active ingredients, catalysts, and pharmaceutical compounds. The material can selectively adsorb various substances from liquids and gases, making it useful in purification processes, chromatography, and controlled release applications.
- Silicic acid in coating and surface treatment applications: Silicic acid serves as a functional component in coating formulations and surface treatments. It provides anti-blocking properties, improves surface texture, and enhances adhesion characteristics. The material can modify surface properties such as hydrophobicity, smoothness, and optical characteristics when applied to various substrates including paper, plastics, and metals.
- Silicic acid as a rheology modifier and thickening agent: Silicic acid functions as a rheology control agent in liquid formulations. It modifies viscosity, prevents settling of suspended particles, and improves stability of dispersions and suspensions. The thixotropic properties of certain silicic acid types allow for controlled flow behavior in paints, inks, adhesives, and cosmetic formulations.
- Silicic acid in detergent and cleaning compositions: Silicic acid serves multiple functions in detergent formulations including anti-redeposition properties, water softening, and as a builder component. It prevents soil particles from redepositing onto cleaned surfaces and helps maintain alkalinity in washing solutions. The material also provides anti-caking properties in powder detergent formulations.
02 Surface modification and functionalization of silicic acid
The surface of silicic acid can be chemically modified to introduce various functional groups, enhancing its reactivity and compatibility with different materials. Surface treatment methods include silanization, coating with organic compounds, and grafting of functional molecules. These modifications alter the hydrophilic-hydrophobic balance and improve the interaction between silicic acid and organic phases. Functionalized silicic acid exhibits improved dispersibility in polymers and enhanced interfacial adhesion.Expand Specific Solutions03 Silicic acid in catalyst support and adsorption applications
Silicic acid materials serve as effective catalyst supports due to their high surface area, thermal stability, and tunable pore structure. The hydroxyl groups on the silicic acid surface provide anchoring sites for active metal species. In adsorption applications, silicic acid functions as a selective adsorbent for various substances including moisture, gases, and organic compounds. The adsorption capacity can be enhanced through controlled synthesis methods and surface modifications.Expand Specific Solutions04 Preparation methods and structural control of silicic acid
Various synthesis methods are employed to produce silicic acid with controlled properties, including precipitation from silicate solutions, sol-gel processes, and hydrothermal treatments. The preparation conditions such as pH, temperature, concentration, and aging time significantly influence the resulting particle morphology, porosity, and surface characteristics. Controlled synthesis enables the production of silicic acid with specific surface areas, pore volumes, and particle size distributions tailored for particular applications.Expand Specific Solutions05 Silicic acid in coating, adhesive and sealant formulations
Silicic acid functions as a rheology modifier, anti-settling agent, and reinforcing component in coatings, adhesives, and sealants. It provides thixotropic properties, prevents pigment settling, and improves mechanical strength of cured films. The incorporation of silicic acid enhances scratch resistance, weatherability, and durability of coating systems. In adhesive formulations, it contributes to improved bonding strength and thermal stability.Expand Specific Solutions
Key Players in Silicic Acid Nanocomposite Industry
The silicic acid nanocomposite field represents an evolving technology landscape characterized by diverse stakeholder participation and moderate market maturity. The competitive arena spans academic institutions, chemical manufacturers, and specialized materials companies, indicating a transition from fundamental research to commercial application. Market growth is driven by demand in battery technologies, coatings, and advanced materials sectors. Technology maturity varies significantly across applications, with established players like Wacker Chemie AG, Evonik Operations GmbH, and DuPont de Nemours demonstrating advanced commercialization capabilities in silica-based materials. Meanwhile, innovative entrants such as Sicona Battery Technologies and Melior Innovations focus on next-generation applications. Leading research institutions including Rice University, Central South University, and Agency for Science, Technology & Research contribute foundational knowledge, while industrial giants like Lockheed Martin explore aerospace applications, collectively positioning this technology at a critical inflection point between research validation and widespread market adoption.
Agency for Science, Technology & Research
Technical Solution: A*STAR conducts fundamental and applied research on silicic acid functionality in nanocomposite design, focusing on understanding the molecular mechanisms of silica network formation and interfacial interactions in hybrid materials. Their research programs investigate how silicic acid speciation, pH conditions, and condensation kinetics influence the structural evolution of silica phases within nanocomposites. The agency's work includes developing bio-inspired approaches that mimic natural silicification processes, where silicic acid polymerization is templated by organic molecules to create hierarchically structured nanocomposites with controlled porosity and mechanical properties. A*STAR researchers explore applications ranging from drug delivery systems utilizing mesoporous silica nanocomposites to advanced membrane materials where silicic acid-derived networks provide selective transport properties. Their multidisciplinary approach combines materials chemistry, computational modeling, and advanced characterization to optimize silicic acid functionality for next-generation nanocomposite applications.
Strengths: Strong fundamental research capabilities with access to advanced characterization facilities; collaborative network enabling translation from basic science to applications. Weaknesses: As a research institution, limited direct commercialization capabilities; longer timelines for technology maturation compared to industrial players.
Sicona Battery Technologies Pty Ltd.
Technical Solution: Sicona Battery Technologies specializes in developing silicon-based composite materials for lithium-ion battery anodes, where silicic acid derivatives play a crucial role in nanocomposite design. Their proprietary technology involves using silicic acid as a precursor to create controlled silica coatings on silicon nanoparticles, which serves as both a structural stabilizer and electrochemical buffer layer. The silicic acid-derived silica matrix helps accommodate the significant volume expansion of silicon during lithiation cycles while maintaining electrical conductivity pathways. This approach enables the creation of high-capacity anode materials with improved cycling stability, where the silicic acid functionality provides mechanical reinforcement and prevents particle agglomeration during battery operation.
Strengths: Specialized expertise in silicon-carbon nanocomposites with proven commercial applications in energy storage; effective volume expansion management through silica matrix engineering. Weaknesses: Limited application scope focused primarily on battery technology; scalability challenges in silicic acid processing for industrial production.
Core Technologies in Silicic Acid Nanocomposite Design
Cell-Based Composite Materials with Programmed Structures and Functions
PatentActiveUS20180299357A1
Innovation
- A method involving the use of silicic acid to transform biological materials, including cells, into inorganic cell/silica composites that retain biological activity and can be stabilized for long periods, allowing for the creation of biocomposites with programmed structures and functions by incubating cells in a silicic acid solution, followed by calcination to produce silica replicas.
Method of coating a particle
PatentInactiveUS20100276310A1
Innovation
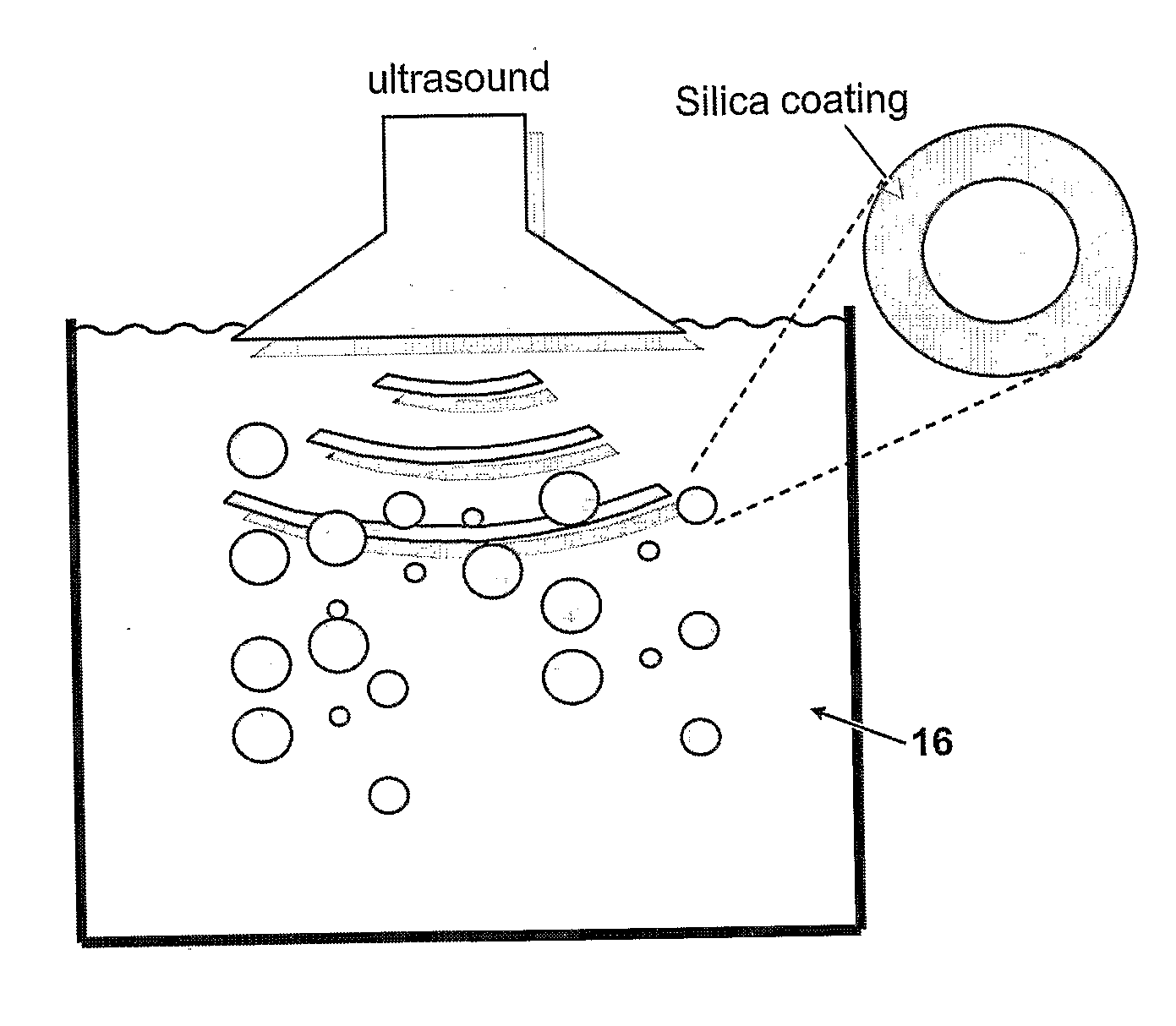
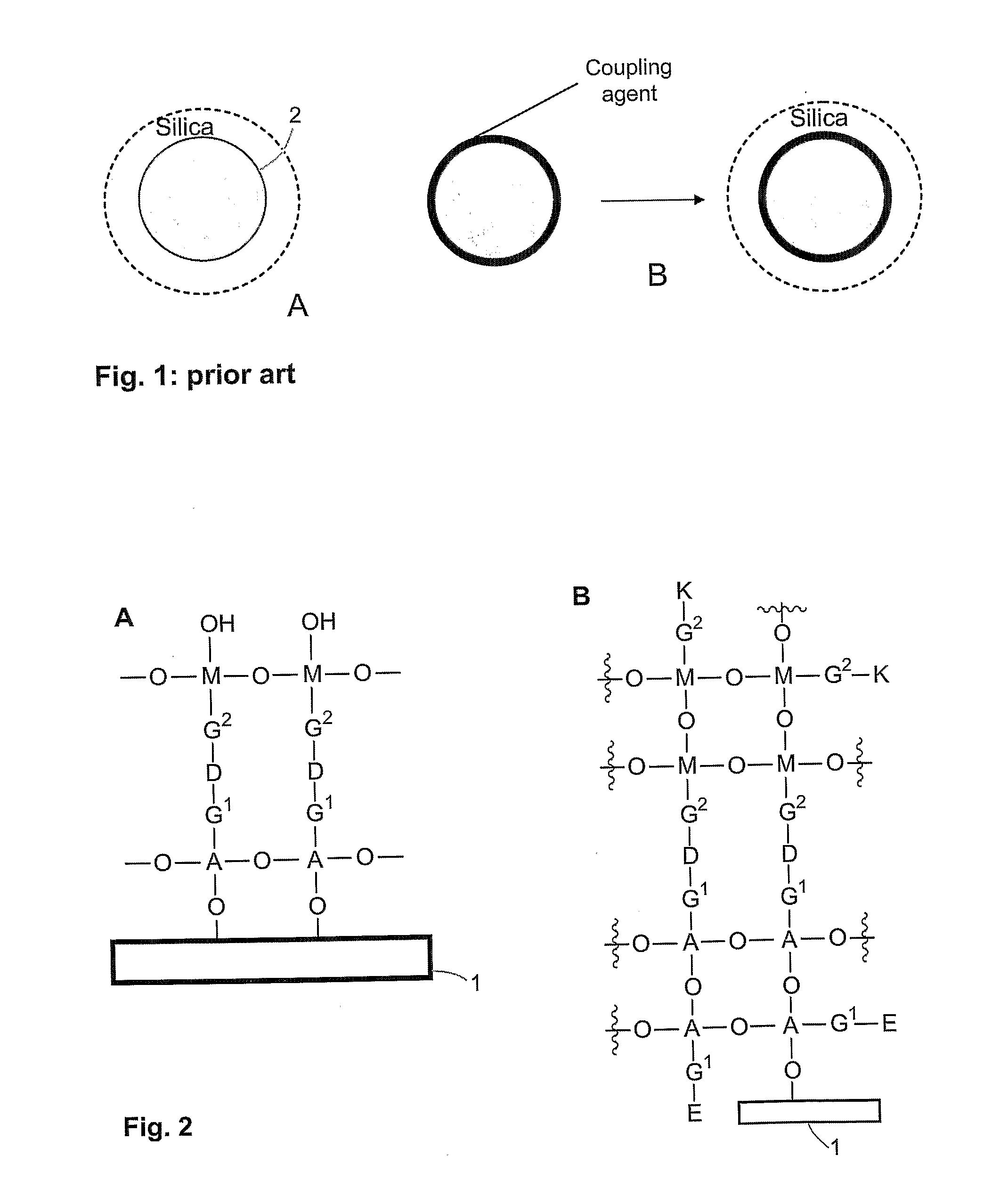
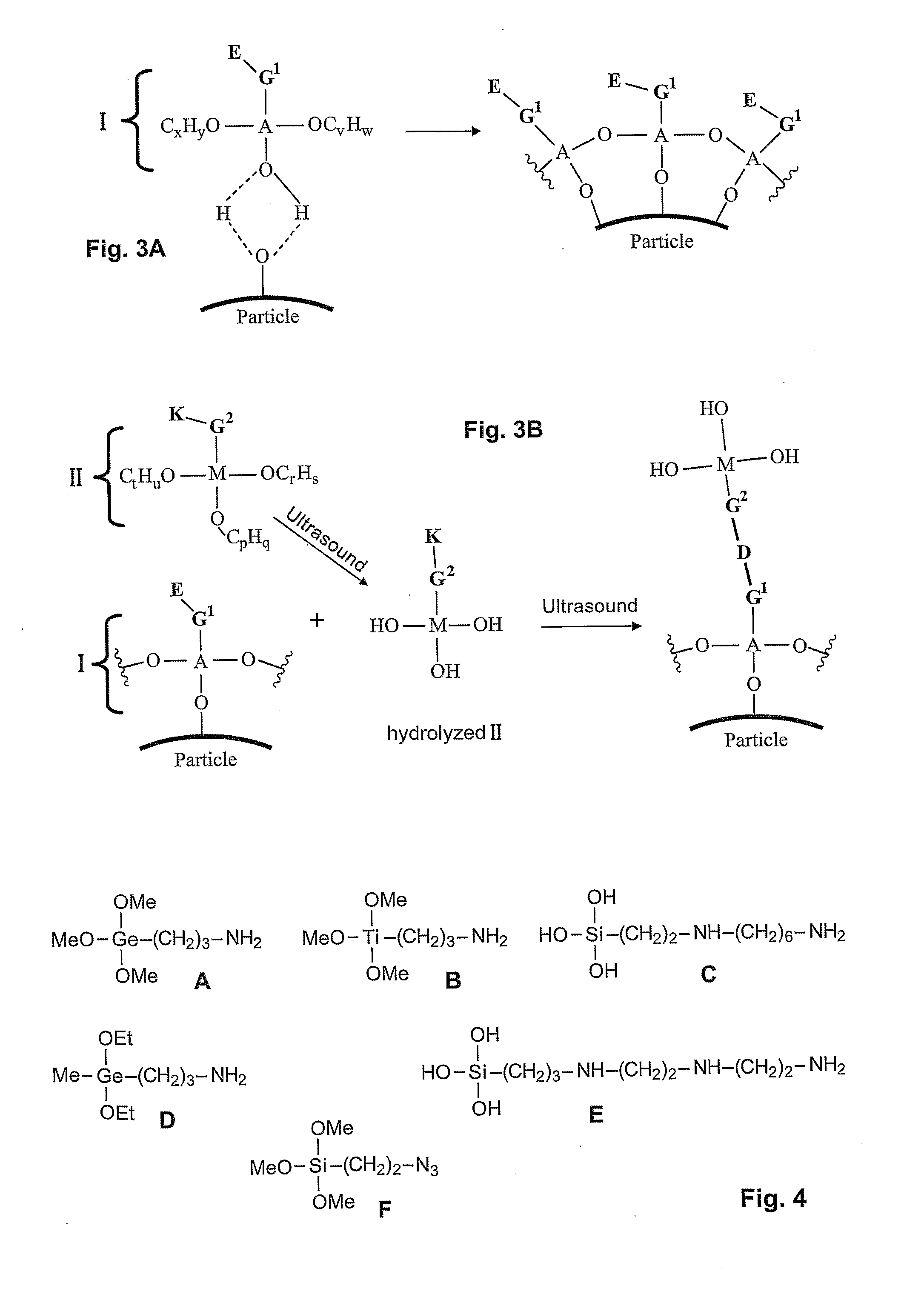
- A method involving the use of molecules with specific functional groups that undergo coupling reactions and subsequent exposure to ultrasound to form a cross-linked coating, allowing for improved dispersability and stability of particles in both aqueous and organic media.
Environmental Impact of Silicic Acid Nanocomposite Production
The production of silicic acid-based nanocomposites presents a complex environmental profile that requires comprehensive assessment across multiple lifecycle stages. Manufacturing processes typically involve sol-gel synthesis, hydrothermal treatment, or precipitation methods, each carrying distinct environmental footprints. Sol-gel routes, while offering precise control over nanostructure formation, often require organic solvents and energy-intensive drying procedures that generate volatile organic compound emissions and consume substantial thermal energy. Conversely, aqueous-based precipitation methods demonstrate reduced solvent dependency but may produce larger volumes of wastewater requiring treatment before discharge.
Energy consumption constitutes a primary environmental concern throughout nanocomposite production. High-temperature calcination steps, commonly employed to enhance material crystallinity and remove organic templates, demand significant energy inputs ranging from 400 to 800 degrees Celsius. This thermal processing contributes substantially to the carbon footprint, particularly when powered by fossil fuel-based electricity grids. Recent lifecycle assessments indicate that energy-related emissions account for approximately 60-70 percent of total greenhouse gas outputs in conventional silicic acid nanocomposite manufacturing.
Raw material extraction and processing also impose environmental burdens. Silica precursors derived from mining operations involve land disruption, water usage, and transportation emissions. However, emerging approaches utilizing agricultural waste streams such as rice husk ash or diatomaceous earth as silica sources demonstrate promising potential for reducing primary resource extraction impacts while simultaneously addressing waste management challenges.
Waste generation patterns vary significantly depending on synthesis routes. Chemical precipitation methods typically produce alkaline or acidic effluents containing residual reactants and byproducts requiring neutralization and proper disposal. Nanomaterial release during production, handling, and equipment cleaning poses potential ecological risks that remain inadequately characterized, necessitating implementation of containment protocols and filtration systems.
End-of-life considerations for silicic acid nanocomposites remain underdeveloped in current environmental assessments. While silica-based materials generally exhibit low toxicity profiles, composite formulations incorporating polymer matrices or functional additives complicate recyclability and biodegradation pathways. Establishing circular economy frameworks through material recovery strategies and designing for disassembly represents a critical frontier for minimizing long-term environmental impacts associated with these advanced materials.
Energy consumption constitutes a primary environmental concern throughout nanocomposite production. High-temperature calcination steps, commonly employed to enhance material crystallinity and remove organic templates, demand significant energy inputs ranging from 400 to 800 degrees Celsius. This thermal processing contributes substantially to the carbon footprint, particularly when powered by fossil fuel-based electricity grids. Recent lifecycle assessments indicate that energy-related emissions account for approximately 60-70 percent of total greenhouse gas outputs in conventional silicic acid nanocomposite manufacturing.
Raw material extraction and processing also impose environmental burdens. Silica precursors derived from mining operations involve land disruption, water usage, and transportation emissions. However, emerging approaches utilizing agricultural waste streams such as rice husk ash or diatomaceous earth as silica sources demonstrate promising potential for reducing primary resource extraction impacts while simultaneously addressing waste management challenges.
Waste generation patterns vary significantly depending on synthesis routes. Chemical precipitation methods typically produce alkaline or acidic effluents containing residual reactants and byproducts requiring neutralization and proper disposal. Nanomaterial release during production, handling, and equipment cleaning poses potential ecological risks that remain inadequately characterized, necessitating implementation of containment protocols and filtration systems.
End-of-life considerations for silicic acid nanocomposites remain underdeveloped in current environmental assessments. While silica-based materials generally exhibit low toxicity profiles, composite formulations incorporating polymer matrices or functional additives complicate recyclability and biodegradation pathways. Establishing circular economy frameworks through material recovery strategies and designing for disassembly represents a critical frontier for minimizing long-term environmental impacts associated with these advanced materials.
Performance Evaluation Methods for Silicic Acid Nanocomposites
Comprehensive performance evaluation of silicic acid nanocomposites requires a multi-dimensional analytical framework that encompasses mechanical, thermal, chemical, and functional properties. Standardized testing protocols must be established to ensure reproducibility and comparability across different research groups and industrial applications. The evaluation methodology should address both intrinsic material properties and application-specific performance metrics, providing quantitative data that correlates silicic acid content and distribution with composite behavior.
Mechanical characterization forms the foundation of performance assessment, employing techniques such as tensile testing, flexural analysis, and dynamic mechanical analysis (DMA) to quantify strength, modulus, and viscoelastic properties. Nanoindentation provides localized mechanical information at the nanoscale, revealing interfacial adhesion quality between silicic acid particles and the polymer matrix. Impact resistance testing evaluates energy absorption capabilities, which is particularly relevant for protective coating applications.
Thermal analysis methods including differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), and thermal conductivity measurements assess thermal stability, degradation behavior, and heat transfer properties. These parameters are critical for applications involving temperature fluctuations or thermal management requirements. The glass transition temperature and decomposition onset temperature serve as key indicators of thermal performance enhancement achieved through silicic acid incorporation.
Microstructural characterization utilizing scanning electron microscopy (SEM), transmission electron microscopy (TEM), and atomic force microscopy (AFM) provides visual confirmation of particle dispersion, size distribution, and interfacial morphology. X-ray diffraction (XRD) and Fourier-transform infrared spectroscopy (FTIR) reveal crystallographic structure and chemical bonding characteristics, elucidating interaction mechanisms between silicic acid and matrix materials.
Functional property evaluation must address application-specific requirements such as barrier properties for packaging applications, optical transparency for coatings, electrical insulation for electronic applications, and biocompatibility for biomedical uses. Water vapor transmission rate (WVTR) and oxygen transmission rate (OTR) measurements quantify barrier performance, while UV-Vis spectroscopy assesses optical properties. Long-term durability testing under accelerated aging conditions provides insights into service life prediction and degradation mechanisms.
Mechanical characterization forms the foundation of performance assessment, employing techniques such as tensile testing, flexural analysis, and dynamic mechanical analysis (DMA) to quantify strength, modulus, and viscoelastic properties. Nanoindentation provides localized mechanical information at the nanoscale, revealing interfacial adhesion quality between silicic acid particles and the polymer matrix. Impact resistance testing evaluates energy absorption capabilities, which is particularly relevant for protective coating applications.
Thermal analysis methods including differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), and thermal conductivity measurements assess thermal stability, degradation behavior, and heat transfer properties. These parameters are critical for applications involving temperature fluctuations or thermal management requirements. The glass transition temperature and decomposition onset temperature serve as key indicators of thermal performance enhancement achieved through silicic acid incorporation.
Microstructural characterization utilizing scanning electron microscopy (SEM), transmission electron microscopy (TEM), and atomic force microscopy (AFM) provides visual confirmation of particle dispersion, size distribution, and interfacial morphology. X-ray diffraction (XRD) and Fourier-transform infrared spectroscopy (FTIR) reveal crystallographic structure and chemical bonding characteristics, elucidating interaction mechanisms between silicic acid and matrix materials.
Functional property evaluation must address application-specific requirements such as barrier properties for packaging applications, optical transparency for coatings, electrical insulation for electronic applications, and biocompatibility for biomedical uses. Water vapor transmission rate (WVTR) and oxygen transmission rate (OTR) measurements quantify barrier performance, while UV-Vis spectroscopy assesses optical properties. Long-term durability testing under accelerated aging conditions provides insights into service life prediction and degradation mechanisms.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!