Exploring Polymer Electrolyte Membranes for Hydrogen Fuel Cell Efficiency
OCT 24, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
PEM Fuel Cell Technology Background and Objectives
Polymer Electrolyte Membrane (PEM) fuel cells have emerged as a promising clean energy technology since their development in the early 1960s by General Electric. The evolution of this technology has been marked by significant improvements in membrane materials, catalysts, and system integration, transforming PEM fuel cells from laboratory curiosities to commercially viable energy solutions.
The fundamental principle of PEM fuel cells involves the electrochemical conversion of hydrogen and oxygen into electricity, with water as the only byproduct. This clean energy generation process positions PEM fuel cells as a critical technology in the global transition away from fossil fuels, particularly in transportation and stationary power applications.
Historical development of PEM technology has progressed through several distinct phases. The initial discovery phase (1960s-1970s) focused on basic principles and proof-of-concept. The optimization phase (1980s-1990s) saw improvements in membrane durability and power density. The commercialization phase (2000s-2010s) brought cost reductions and manufacturing scale-up. Currently, we are in the mass adoption phase, characterized by integration into commercial products and infrastructure development.
The primary technical objective in PEM fuel cell development is enhancing energy conversion efficiency while reducing system costs. Current state-of-the-art PEM fuel cells achieve approximately 60% electrical efficiency, but theoretical limits suggest potential for up to 83% efficiency. Closing this gap represents a significant opportunity for technological advancement and commercial viability improvement.
Key technical goals include developing membranes with higher proton conductivity, reduced gas crossover, enhanced mechanical stability, and longer operational lifetimes under variable conditions. Additionally, reducing platinum catalyst loading or finding alternative catalysts remains critical for cost reduction, as platinum currently represents approximately 40% of stack costs.
The industry aims to achieve durability targets of 5,000 operating hours for light-duty vehicles and 30,000 hours for stationary applications, while simultaneously reducing system costs to under $30/kW for automotive applications by 2025. These ambitious targets necessitate fundamental innovations in membrane materials and manufacturing processes.
Global research efforts are increasingly focused on next-generation membranes incorporating novel polymers, composite structures, and nanomaterials. These advanced materials promise to overcome current limitations in temperature range, humidity dependence, and chemical stability that restrict broader adoption of PEM fuel cell technology.
The convergence of materials science breakthroughs, manufacturing innovations, and growing market demand positions PEM fuel cell technology at an inflection point, with significant potential for accelerated development and deployment across multiple sectors in the coming decade.
The fundamental principle of PEM fuel cells involves the electrochemical conversion of hydrogen and oxygen into electricity, with water as the only byproduct. This clean energy generation process positions PEM fuel cells as a critical technology in the global transition away from fossil fuels, particularly in transportation and stationary power applications.
Historical development of PEM technology has progressed through several distinct phases. The initial discovery phase (1960s-1970s) focused on basic principles and proof-of-concept. The optimization phase (1980s-1990s) saw improvements in membrane durability and power density. The commercialization phase (2000s-2010s) brought cost reductions and manufacturing scale-up. Currently, we are in the mass adoption phase, characterized by integration into commercial products and infrastructure development.
The primary technical objective in PEM fuel cell development is enhancing energy conversion efficiency while reducing system costs. Current state-of-the-art PEM fuel cells achieve approximately 60% electrical efficiency, but theoretical limits suggest potential for up to 83% efficiency. Closing this gap represents a significant opportunity for technological advancement and commercial viability improvement.
Key technical goals include developing membranes with higher proton conductivity, reduced gas crossover, enhanced mechanical stability, and longer operational lifetimes under variable conditions. Additionally, reducing platinum catalyst loading or finding alternative catalysts remains critical for cost reduction, as platinum currently represents approximately 40% of stack costs.
The industry aims to achieve durability targets of 5,000 operating hours for light-duty vehicles and 30,000 hours for stationary applications, while simultaneously reducing system costs to under $30/kW for automotive applications by 2025. These ambitious targets necessitate fundamental innovations in membrane materials and manufacturing processes.
Global research efforts are increasingly focused on next-generation membranes incorporating novel polymers, composite structures, and nanomaterials. These advanced materials promise to overcome current limitations in temperature range, humidity dependence, and chemical stability that restrict broader adoption of PEM fuel cell technology.
The convergence of materials science breakthroughs, manufacturing innovations, and growing market demand positions PEM fuel cell technology at an inflection point, with significant potential for accelerated development and deployment across multiple sectors in the coming decade.
Hydrogen Fuel Cell Market Analysis and Demand
The global hydrogen fuel cell market has experienced significant growth in recent years, driven by increasing environmental concerns and the push for clean energy alternatives. As of 2023, the market was valued at approximately 5.6 billion USD, with projections indicating a compound annual growth rate (CAGR) of 21.4% through 2030. This remarkable growth trajectory reflects the expanding applications of hydrogen fuel cells across various sectors, particularly in transportation, stationary power generation, and portable electronics.
Transportation represents the largest application segment, accounting for over 60% of the total market share. Major automotive manufacturers including Toyota, Hyundai, and Honda have commercially launched fuel cell electric vehicles (FCEVs), while heavy-duty applications such as buses and trucks are gaining traction due to their operational advantages over battery electric alternatives for long-range transportation.
Regional analysis reveals that Asia Pacific currently dominates the market, led by Japan, South Korea, and China, which collectively hold approximately 45% of the global market share. These countries have implemented aggressive hydrogen strategies and substantial government subsidies to accelerate adoption. North America and Europe follow closely, with significant investments in hydrogen infrastructure and research initiatives.
Consumer demand for hydrogen fuel cells is primarily driven by their environmental benefits, including zero-emission operation and water as the only byproduct. Additionally, the quick refueling time (3-5 minutes) compared to the extended charging periods required for battery electric vehicles represents a significant advantage for time-sensitive applications.
Industry surveys indicate that efficiency improvements remain a critical factor influencing market adoption. Current polymer electrolyte membrane fuel cells (PEMFCs) typically achieve 40-60% efficiency, but potential users consistently express demand for higher efficiency systems. Market research shows that a 10% improvement in fuel cell efficiency could expand the addressable market by approximately 30%, particularly in price-sensitive segments.
Supply chain challenges present significant market constraints, with platinum catalyst costs representing 30-40% of stack manufacturing expenses. This has created strong market demand for advanced membrane technologies that can maintain performance while reducing precious metal content. Manufacturers capable of delivering high-performance polymer electrolyte membranes with enhanced durability and reduced platinum loading are positioned to capture premium market segments.
The COVID-19 pandemic temporarily disrupted market growth in 2020, but recovery has been robust, with hydrogen increasingly featured in national energy strategies and economic stimulus packages worldwide. This policy support, combined with technological advancements in polymer electrolyte membranes, is expected to accelerate market penetration across multiple sectors in the coming decade.
Transportation represents the largest application segment, accounting for over 60% of the total market share. Major automotive manufacturers including Toyota, Hyundai, and Honda have commercially launched fuel cell electric vehicles (FCEVs), while heavy-duty applications such as buses and trucks are gaining traction due to their operational advantages over battery electric alternatives for long-range transportation.
Regional analysis reveals that Asia Pacific currently dominates the market, led by Japan, South Korea, and China, which collectively hold approximately 45% of the global market share. These countries have implemented aggressive hydrogen strategies and substantial government subsidies to accelerate adoption. North America and Europe follow closely, with significant investments in hydrogen infrastructure and research initiatives.
Consumer demand for hydrogen fuel cells is primarily driven by their environmental benefits, including zero-emission operation and water as the only byproduct. Additionally, the quick refueling time (3-5 minutes) compared to the extended charging periods required for battery electric vehicles represents a significant advantage for time-sensitive applications.
Industry surveys indicate that efficiency improvements remain a critical factor influencing market adoption. Current polymer electrolyte membrane fuel cells (PEMFCs) typically achieve 40-60% efficiency, but potential users consistently express demand for higher efficiency systems. Market research shows that a 10% improvement in fuel cell efficiency could expand the addressable market by approximately 30%, particularly in price-sensitive segments.
Supply chain challenges present significant market constraints, with platinum catalyst costs representing 30-40% of stack manufacturing expenses. This has created strong market demand for advanced membrane technologies that can maintain performance while reducing precious metal content. Manufacturers capable of delivering high-performance polymer electrolyte membranes with enhanced durability and reduced platinum loading are positioned to capture premium market segments.
The COVID-19 pandemic temporarily disrupted market growth in 2020, but recovery has been robust, with hydrogen increasingly featured in national energy strategies and economic stimulus packages worldwide. This policy support, combined with technological advancements in polymer electrolyte membranes, is expected to accelerate market penetration across multiple sectors in the coming decade.
Current PEM Technology Challenges and Limitations
Despite significant advancements in polymer electrolyte membrane (PEM) technology for hydrogen fuel cells, several critical challenges continue to impede widespread commercial adoption and optimal efficiency. Current PEM materials, predominantly perfluorosulfonic acid (PFSA) polymers like Nafion, face substantial durability issues under real-world operating conditions. These membranes experience chemical degradation through radical attack, particularly from hydroxyl and peroxyl radicals formed during fuel cell operation, leading to membrane thinning and eventual failure.
Mechanical stability represents another significant limitation, as PEMs undergo dimensional changes during hydration/dehydration cycles, causing mechanical stress that results in pinhole formation and catastrophic gas crossover. This problem becomes particularly acute during freeze-thaw cycles and high-temperature operations, severely limiting the operational temperature range of current systems.
Cost factors remain prohibitive for mass-market applications. The high price of PFSA membranes, primarily due to complex fluorination processes and expensive raw materials, contributes significantly to the overall cost structure of fuel cell systems. Current manufacturing techniques also lack scalability for high-volume production without compromising quality consistency.
Water management presents a persistent engineering challenge. PEMs require precise hydration levels to maintain proton conductivity, yet excess water causes flooding that blocks reactant transport. Conversely, insufficient hydration increases ohmic resistance and accelerates degradation. This delicate balance is difficult to maintain across varying operational conditions and power demands.
Temperature limitations further constrain fuel cell performance. Most commercial PEMs operate optimally between 60-80°C, with significant conductivity losses above 90°C. This narrow temperature window restricts heat rejection capabilities and requires complex cooling systems, while also making catalyst poisoning by impurities more problematic due to slower reaction kinetics at lower temperatures.
Ion conductivity at low relative humidity remains suboptimal in current membranes. As humidity decreases, proton transport efficiency drops dramatically, necessitating complex humidification systems that add weight, volume, and parasitic energy losses to the overall system.
Cross-contamination issues persist, with fuel crossover (hydrogen diffusion through the membrane) reducing fuel utilization efficiency and voltage output. This problem becomes more pronounced at higher temperatures and pressures, creating a technical barrier to high-power density applications.
Manufacturing consistency and quality control continue to challenge producers, with batch-to-batch variations affecting performance predictability and system integration. These limitations collectively highlight the need for innovative materials and manufacturing approaches to advance PEM technology toward greater commercial viability.
Mechanical stability represents another significant limitation, as PEMs undergo dimensional changes during hydration/dehydration cycles, causing mechanical stress that results in pinhole formation and catastrophic gas crossover. This problem becomes particularly acute during freeze-thaw cycles and high-temperature operations, severely limiting the operational temperature range of current systems.
Cost factors remain prohibitive for mass-market applications. The high price of PFSA membranes, primarily due to complex fluorination processes and expensive raw materials, contributes significantly to the overall cost structure of fuel cell systems. Current manufacturing techniques also lack scalability for high-volume production without compromising quality consistency.
Water management presents a persistent engineering challenge. PEMs require precise hydration levels to maintain proton conductivity, yet excess water causes flooding that blocks reactant transport. Conversely, insufficient hydration increases ohmic resistance and accelerates degradation. This delicate balance is difficult to maintain across varying operational conditions and power demands.
Temperature limitations further constrain fuel cell performance. Most commercial PEMs operate optimally between 60-80°C, with significant conductivity losses above 90°C. This narrow temperature window restricts heat rejection capabilities and requires complex cooling systems, while also making catalyst poisoning by impurities more problematic due to slower reaction kinetics at lower temperatures.
Ion conductivity at low relative humidity remains suboptimal in current membranes. As humidity decreases, proton transport efficiency drops dramatically, necessitating complex humidification systems that add weight, volume, and parasitic energy losses to the overall system.
Cross-contamination issues persist, with fuel crossover (hydrogen diffusion through the membrane) reducing fuel utilization efficiency and voltage output. This problem becomes more pronounced at higher temperatures and pressures, creating a technical barrier to high-power density applications.
Manufacturing consistency and quality control continue to challenge producers, with batch-to-batch variations affecting performance predictability and system integration. These limitations collectively highlight the need for innovative materials and manufacturing approaches to advance PEM technology toward greater commercial viability.
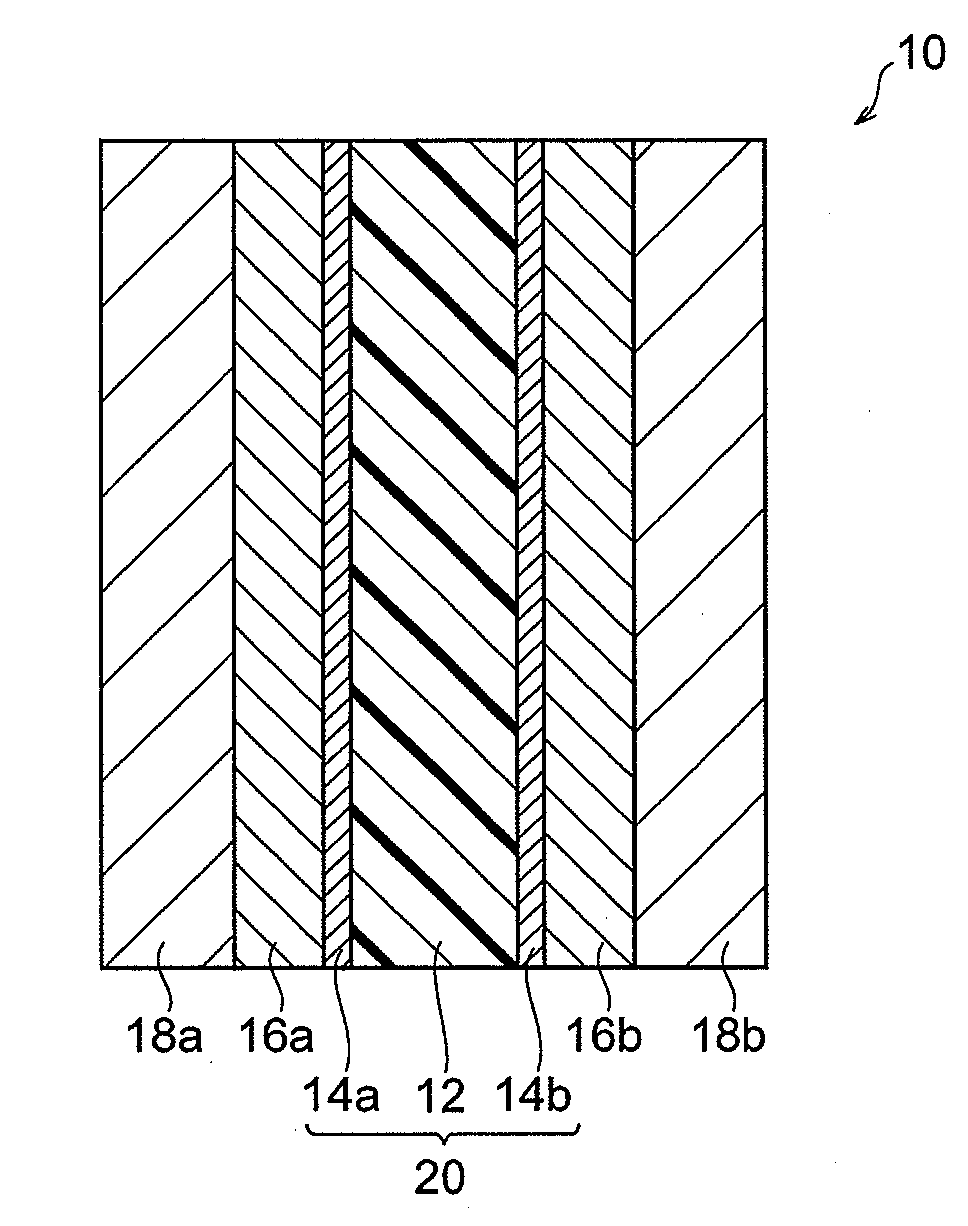
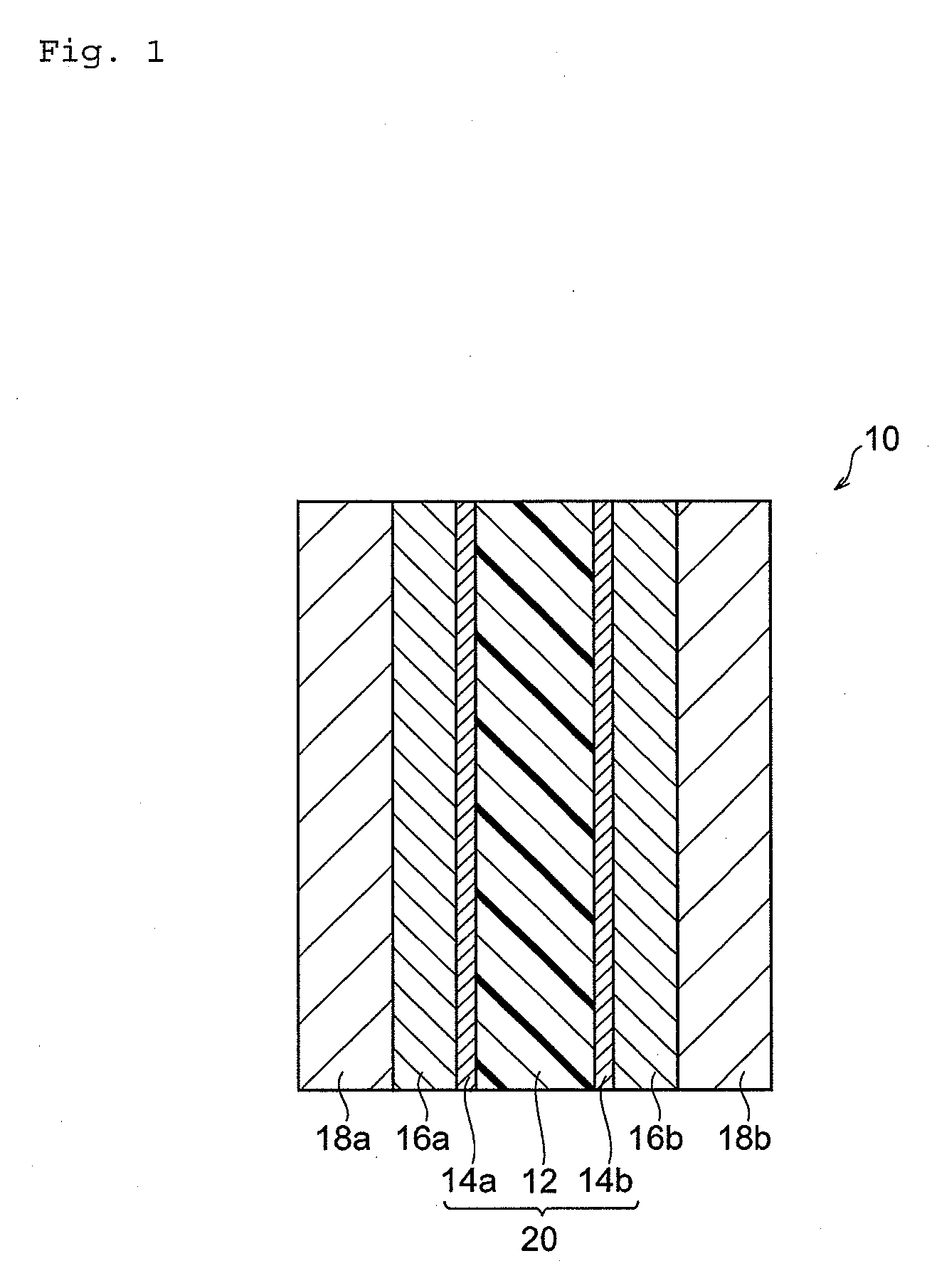
State-of-the-Art PEM Materials and Designs
01 Polymer composition for improved membrane efficiency
Specific polymer compositions can significantly enhance the efficiency of electrolyte membranes. These compositions typically include fluorinated polymers, sulfonated polymers, or composite materials that offer improved proton conductivity while maintaining mechanical stability. The incorporation of specific functional groups and optimized molecular structures allows for better ion transport across the membrane, resulting in higher overall efficiency for fuel cell applications.- Fluoropolymer-based membranes for enhanced conductivity: Fluoropolymer-based electrolyte membranes, particularly those using perfluorosulfonic acid polymers, demonstrate superior proton conductivity and chemical stability in fuel cell applications. These membranes feature optimized sulfonic acid group distribution and controlled polymer architecture to enhance ion transport while maintaining mechanical durability. Modifications with reinforcing structures or composite formations further improve their performance under varying operating conditions, resulting in higher overall efficiency and longer operational lifetimes.
- Composite membranes with inorganic fillers: Incorporating inorganic fillers such as metal oxides, silica, or zeolites into polymer matrices creates composite membranes with enhanced water retention and thermal stability. These fillers create additional pathways for proton transport while reducing membrane swelling and improving dimensional stability at higher temperatures. The synergistic interaction between the polymer matrix and inorganic components results in membranes that maintain high conductivity even under low humidity conditions, addressing a key limitation of conventional polymer electrolyte membranes.
- Cross-linked polymer architectures for stability: Cross-linking techniques applied to polymer electrolyte membranes significantly improve their mechanical strength and dimensional stability under fluctuating humidity and temperature conditions. These architectures utilize chemical or radiation-induced cross-linking to create interconnected polymer networks that resist degradation while maintaining adequate ion channels for efficient proton transport. The optimized cross-linking density balances the competing requirements of mechanical robustness and ionic conductivity, resulting in membranes with extended operational lifetimes and consistent performance.
- Surface modification techniques: Surface modification of polymer electrolyte membranes through plasma treatment, grafting, or layer-by-layer assembly enhances interfacial properties and reduces resistance at the membrane-electrode interface. These techniques create hydrophilic surface domains that facilitate water management and ion transport while potentially reducing catalyst poisoning. Modified membrane surfaces demonstrate improved electrode adhesion and reduced contact resistance, contributing to higher overall fuel cell efficiency and power density without compromising the bulk properties of the membrane.
- Hydrocarbon-based alternative membranes: Hydrocarbon-based polymer electrolyte membranes represent a cost-effective alternative to fluoropolymer membranes while offering comparable or superior performance in specific applications. These membranes utilize sulfonated aromatic polymers such as polyether sulfones, polyimides, or polybenzimidazoles that can be precisely engineered for high ion exchange capacity. Their advantages include better high-temperature stability, reduced gas permeability, and more environmentally friendly production processes, though challenges remain in achieving the chemical durability of fluorinated counterparts under aggressive operating conditions.
02 Membrane fabrication techniques for enhanced performance
Advanced fabrication methods can significantly improve polymer electrolyte membrane efficiency. These techniques include controlled phase separation, electrospinning, solution casting with specific solvent systems, and various surface modification approaches. By optimizing the manufacturing process, membranes with more uniform structure, controlled porosity, and better interfacial properties can be produced, leading to enhanced proton conductivity and reduced resistance.Expand Specific Solutions03 Composite membranes with inorganic fillers
Incorporating inorganic fillers into polymer electrolyte membranes can significantly improve their efficiency. Materials such as silica, titanium dioxide, zirconium phosphate, and various metal oxides create composite membranes with enhanced water retention, mechanical strength, and thermal stability. These fillers can create additional proton conduction pathways and improve the membrane's performance under various operating conditions, particularly at higher temperatures and lower humidity.Expand Specific Solutions04 Cross-linking strategies for stability and conductivity
Cross-linking techniques can be employed to improve both the stability and conductivity of polymer electrolyte membranes. By creating covalent bonds between polymer chains, these methods enhance mechanical strength, reduce swelling, and improve dimensional stability while maintaining or enhancing proton conductivity. Various cross-linking agents and radiation-induced methods can be used to achieve an optimal balance between mechanical properties and electrochemical performance.Expand Specific Solutions05 Additives for improved water management and conductivity
Specific additives can be incorporated into polymer electrolyte membranes to enhance water management and conductivity. These include hygroscopic compounds, ionic liquids, heteropolyacids, and various proton-conducting materials that help maintain appropriate hydration levels and create additional proton transport pathways. Improved water retention and distribution within the membrane structure leads to better proton conductivity across a wider range of operating conditions, particularly at elevated temperatures.Expand Specific Solutions
Leading Companies in PEM Fuel Cell Development
The polymer electrolyte membrane (PEM) fuel cell market is currently in a growth phase, with increasing adoption driven by the global push for clean energy solutions. Market size is projected to expand significantly as hydrogen technologies gain traction in transportation, stationary power, and portable applications. Leading companies like Toyota Motor Corp., LG Chem, and W.L. Gore & Associates have established strong technological positions through extensive R&D investments. Technical maturity varies across membrane types, with perfluorosulfonic acid membranes being most commercially advanced. Research institutions like Nanyang Technological University and companies including DuPont, Toray Industries, and Samsung SDI are advancing next-generation membranes focusing on higher temperature operation, reduced humidity dependence, and improved durability—critical factors for broader commercial adoption in competitive energy markets.
LG Chem Ltd.
Technical Solution: LG Chem has developed advanced polymer electrolyte membranes based on their proprietary sulfonated poly(arylene ether sulfone) chemistry. Their membrane technology focuses on high-temperature operation (up to 120°C) while maintaining dimensional stability through cross-linked polymer architectures. LG Chem's membranes incorporate nanocomposite additives, specifically functionalized titanium dioxide particles (3-5 nm diameter) that create additional water retention sites, enabling operation at lower relative humidity (down to 30% RH) while maintaining acceptable proton conductivity (>50 mS/cm). Their manufacturing process employs solution casting techniques that enable precise control of membrane thickness and uniformity at scale. LG Chem has developed specialized surface treatment processes that enhance the membrane-electrode interface, reducing contact resistance in the assembled fuel cell. Their membranes demonstrate excellent oxidative stability through the incorporation of radical scavenging moieties directly into the polymer backbone, addressing a key degradation mechanism in fuel cell operation. Recent advancements include membranes with gradient sulfonation levels through the thickness, optimizing the balance between mechanical properties and proton conductivity.
Strengths: Excellent high-temperature performance; reduced dependency on external humidification; competitive manufacturing costs; strong integration with LG's broader energy storage portfolio. Weaknesses: Less field validation data compared to PFSA membranes; potential challenges in sub-zero startup conditions; mechanical properties may degrade after extended cycling; higher water uptake can create challenges in water management.
Toyota Motor Corp.
Technical Solution: Toyota has developed proprietary polymer electrolyte membranes optimized for their fuel cell electric vehicles (FCEVs), most notably implemented in the Mirai. Their membrane technology focuses on three key innovations: 1) A 3D nanostructured membrane architecture that creates distinct hydrophilic proton conduction channels while maintaining mechanical integrity through hydrophobic domains, allowing operation across wider humidity ranges; 2) Integration of cerium-based antioxidant compounds that selectively decompose hydrogen peroxide formed during operation, extending membrane lifetime by approximately 60% compared to conventional membranes; 3) A gradient-layer design that optimizes water management by varying the equivalent weight of the ionomer through the membrane thickness. Toyota's membranes demonstrate exceptional durability, maintaining over 90% of initial performance after 5,000 hours of accelerated stress testing under automotive duty cycles. Their manufacturing process incorporates automated quality control systems that ensure consistent membrane properties across production batches, critical for commercial-scale FCEV deployment.
Strengths: Optimized for automotive duty cycles; excellent cold-start capability; integrated with Toyota's comprehensive fuel cell system design; proven real-world durability in commercial vehicles. Weaknesses: Proprietary technology with limited availability outside Toyota's supply chain; potentially higher cost due to specialized manufacturing processes; performance characteristics specifically tailored to Toyota's system architecture may limit broader applicability.
Critical Patents and Research in PEM Technology
Polymer electrolyte, and polymer electrolyte membrane, membrane-electrode assembly and fuel cell that are using the polymer electrolyte
PatentInactiveUS20090117438A1
Innovation
- A polymer electrolyte with an ion-exchange capacity of 1.7 meq/g or more and reduced viscosity of 160 mL/g or more at 40°C, incorporating a block copolymer with a segment having an ion-exchange group and another segment with no ion-exchange group, which forms a membrane with low resistance in both surface and thickness directions, allowing for stable operation in low humidity conditions.
Solid polymer electrolyte membrane, membrane electrode assembly and solid polymer fuel cell
PatentWO2020175675A1
Innovation
- A solid polymer electrolyte membrane with a hydrogen gas permeability coefficient of 2.4 x 10^-9 cm^3/cm^2/s/cmHg or less at 80°C and 10% relative humidity, and a thickness between 7 to 20 μm, incorporating a perfluoropolymer with specific units such as perfluorovinyl ether and perfluoroallyl ether units, and an ion exchange capacity of 1.4 to 2.5 meq/g dry resin, is used to reduce hydrogen crossover and enhance power generation.
Environmental Impact and Sustainability Assessment
The environmental impact of polymer electrolyte membrane (PEM) fuel cells represents a critical consideration in their development and deployment. When compared to conventional energy technologies, PEM fuel cells offer significant environmental advantages, primarily through their zero-emission operation at point of use, producing only water and heat as byproducts. This characteristic positions them as a potentially transformative technology for reducing greenhouse gas emissions, particularly in transportation and stationary power applications.
However, a comprehensive sustainability assessment must consider the entire lifecycle of PEM fuel cell systems. The manufacturing of polymer electrolyte membranes involves energy-intensive processes and specialized materials, including fluoropolymers like Nafion, which carry their own environmental footprint. The extraction and processing of platinum and other noble metals used as catalysts present additional environmental challenges, including habitat disruption, water usage, and potential contamination at mining sites.
Water management represents another important environmental consideration. While fuel cells produce clean water, they also require purified water for operation in many configurations. In water-scarce regions, this dependency could create sustainability challenges that must be addressed through system optimization and water recycling technologies.
The hydrogen production pathway significantly influences the overall environmental profile of PEM fuel cell systems. Green hydrogen produced via electrolysis powered by renewable energy offers the most environmentally beneficial approach, while hydrogen derived from natural gas reforming without carbon capture presents less favorable environmental outcomes despite still offering improvements over conventional combustion technologies.
End-of-life considerations for polymer electrolyte membranes present both challenges and opportunities. Current recycling technologies for fluorinated polymers and precious metal recovery are advancing but remain energy-intensive. Developing more efficient recycling processes and designing membranes with recyclability in mind represents an important frontier for improving sustainability.
Regulatory frameworks worldwide are increasingly incorporating lifecycle assessment requirements for emerging energy technologies. PEM fuel cell developers must proactively address these considerations to ensure market acceptance and regulatory compliance. Several leading manufacturers have already implemented sustainability initiatives focused on reducing manufacturing impacts and improving end-of-life recovery of valuable materials.
The transition to more sustainable membrane materials represents a promising research direction, with bio-based and non-fluorinated alternatives showing potential to reduce environmental impacts while maintaining performance requirements. These developments align with broader sustainability goals and may help address concerns about persistent fluorinated compounds in the environment.
However, a comprehensive sustainability assessment must consider the entire lifecycle of PEM fuel cell systems. The manufacturing of polymer electrolyte membranes involves energy-intensive processes and specialized materials, including fluoropolymers like Nafion, which carry their own environmental footprint. The extraction and processing of platinum and other noble metals used as catalysts present additional environmental challenges, including habitat disruption, water usage, and potential contamination at mining sites.
Water management represents another important environmental consideration. While fuel cells produce clean water, they also require purified water for operation in many configurations. In water-scarce regions, this dependency could create sustainability challenges that must be addressed through system optimization and water recycling technologies.
The hydrogen production pathway significantly influences the overall environmental profile of PEM fuel cell systems. Green hydrogen produced via electrolysis powered by renewable energy offers the most environmentally beneficial approach, while hydrogen derived from natural gas reforming without carbon capture presents less favorable environmental outcomes despite still offering improvements over conventional combustion technologies.
End-of-life considerations for polymer electrolyte membranes present both challenges and opportunities. Current recycling technologies for fluorinated polymers and precious metal recovery are advancing but remain energy-intensive. Developing more efficient recycling processes and designing membranes with recyclability in mind represents an important frontier for improving sustainability.
Regulatory frameworks worldwide are increasingly incorporating lifecycle assessment requirements for emerging energy technologies. PEM fuel cell developers must proactively address these considerations to ensure market acceptance and regulatory compliance. Several leading manufacturers have already implemented sustainability initiatives focused on reducing manufacturing impacts and improving end-of-life recovery of valuable materials.
The transition to more sustainable membrane materials represents a promising research direction, with bio-based and non-fluorinated alternatives showing potential to reduce environmental impacts while maintaining performance requirements. These developments align with broader sustainability goals and may help address concerns about persistent fluorinated compounds in the environment.
Cost Analysis and Commercial Viability
The economic viability of polymer electrolyte membrane (PEM) fuel cells remains a critical barrier to widespread adoption despite their technical advantages. Current production costs for automotive PEM fuel cell systems range between $50-60/kW, significantly higher than the U.S. Department of Energy's target of $30/kW for competitive market entry. This cost differential primarily stems from expensive membrane materials, particularly the industry-standard Nafion, which accounts for approximately 25-30% of the total stack cost.
Manufacturing scale presents another substantial challenge. Current production volumes remain relatively low, preventing economies of scale that could drive down unit costs. Industry analysis indicates that achieving a production volume of 500,000 units annually could potentially reduce manufacturing costs by 35-45%, bringing PEM fuel cells closer to commercial viability.
Material optimization offers promising cost reduction pathways. Research into alternative membrane materials such as sulfonated poly(ether ether ketone) (SPEEK) and polybenzimidazole (PBI) demonstrates potential for 30-40% cost reduction compared to Nafion while maintaining acceptable performance metrics. However, these alternatives typically face durability challenges that must be addressed before commercial implementation.
The platinum catalyst represents another significant cost component, contributing approximately 15-20% of total stack costs. Recent innovations in platinum-alloy catalysts and ultra-low platinum loading techniques have demonstrated potential to reduce catalyst costs by up to 60% while maintaining performance standards. These developments could substantially improve the commercial outlook for PEM fuel cells.
Market analysis reveals that initial commercial viability will likely emerge in specific sectors before broader adoption occurs. The heavy-duty transportation sector, particularly long-haul trucking and bus fleets, presents the most promising near-term commercial opportunity due to operational advantages over battery electric alternatives. The stationary power market also offers viable commercialization pathways, particularly for backup power systems where reliability justifies premium pricing.
Government incentives significantly impact the commercial landscape. Countries with strong hydrogen strategies like Japan, South Korea, and Germany have implemented subsidies reducing end-user costs by 30-50%, accelerating market adoption. Similar policy frameworks in other regions could substantially improve commercial viability timelines.
Investment trends indicate growing confidence in PEM fuel cell commercialization. Venture capital funding in this sector has increased by approximately 65% over the past three years, with particular focus on manufacturing scale-up and material innovations that address cost barriers. This investment momentum suggests improving commercial prospects despite current challenges.
Manufacturing scale presents another substantial challenge. Current production volumes remain relatively low, preventing economies of scale that could drive down unit costs. Industry analysis indicates that achieving a production volume of 500,000 units annually could potentially reduce manufacturing costs by 35-45%, bringing PEM fuel cells closer to commercial viability.
Material optimization offers promising cost reduction pathways. Research into alternative membrane materials such as sulfonated poly(ether ether ketone) (SPEEK) and polybenzimidazole (PBI) demonstrates potential for 30-40% cost reduction compared to Nafion while maintaining acceptable performance metrics. However, these alternatives typically face durability challenges that must be addressed before commercial implementation.
The platinum catalyst represents another significant cost component, contributing approximately 15-20% of total stack costs. Recent innovations in platinum-alloy catalysts and ultra-low platinum loading techniques have demonstrated potential to reduce catalyst costs by up to 60% while maintaining performance standards. These developments could substantially improve the commercial outlook for PEM fuel cells.
Market analysis reveals that initial commercial viability will likely emerge in specific sectors before broader adoption occurs. The heavy-duty transportation sector, particularly long-haul trucking and bus fleets, presents the most promising near-term commercial opportunity due to operational advantages over battery electric alternatives. The stationary power market also offers viable commercialization pathways, particularly for backup power systems where reliability justifies premium pricing.
Government incentives significantly impact the commercial landscape. Countries with strong hydrogen strategies like Japan, South Korea, and Germany have implemented subsidies reducing end-user costs by 30-50%, accelerating market adoption. Similar policy frameworks in other regions could substantially improve commercial viability timelines.
Investment trends indicate growing confidence in PEM fuel cell commercialization. Venture capital funding in this sector has increased by approximately 65% over the past three years, with particular focus on manufacturing scale-up and material innovations that address cost barriers. This investment momentum suggests improving commercial prospects despite current challenges.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!