How Prelithiation Influences SEI Stability and Morphology
SEP 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Prelithiation Technology Background and Objectives
Prelithiation technology has emerged as a critical advancement in lithium-ion battery development, evolving significantly over the past decade. Initially conceived as a solution to first-cycle capacity loss, this technology has expanded to address fundamental challenges in battery performance and longevity. The evolution of prelithiation techniques has progressed from simple mechanical methods to sophisticated electrochemical approaches, reflecting the growing understanding of interfacial chemistry in battery systems.
The solid-electrolyte interphase (SEI) formation consumes lithium ions during initial battery cycling, resulting in irreversible capacity loss of 10-20% in conventional lithium-ion batteries. This phenomenon has become increasingly problematic as manufacturers push for higher energy densities and longer cycle life. Prelithiation technology aims to compensate for this initial lithium loss by introducing additional lithium before battery assembly or during the formation process.
Recent research has revealed that prelithiation not only addresses first-cycle capacity loss but significantly influences the stability and morphology of the SEI layer. The SEI serves as a protective barrier between the electrode and electrolyte, determining critical battery parameters including coulombic efficiency, rate capability, and long-term cycling stability. Understanding how prelithiation affects SEI formation mechanisms has become essential for optimizing battery performance.
The technical objectives of prelithiation research focus on several interconnected goals. Primary among these is developing methods to create more stable and uniform SEI layers that minimize continuous lithium consumption during cycling. Researchers aim to establish correlations between prelithiation parameters (amount, timing, method) and resulting SEI properties (thickness, composition, homogeneity, mechanical stability).
Another critical objective involves understanding the molecular-level interactions between prelithiation agents and electrolyte components during SEI formation. This includes investigating how different prelithiation approaches affect the chemical composition and structural organization of the SEI layer across various electrode materials and electrolyte systems.
The technology trend indicates a shift toward prelithiation methods that can be seamlessly integrated into existing manufacturing processes without significant cost increases or safety concerns. This includes developing stable prelithiation materials with long shelf lives and exploring in-situ prelithiation techniques that can be applied during cell assembly rather than requiring additional processing steps.
As battery applications diversify beyond consumer electronics to electric vehicles and grid storage, prelithiation technology must adapt to different performance requirements and operating conditions. The ultimate goal is to develop tailored prelithiation strategies that can be optimized for specific battery chemistries and use cases, maximizing energy density while ensuring safety and longevity.
The solid-electrolyte interphase (SEI) formation consumes lithium ions during initial battery cycling, resulting in irreversible capacity loss of 10-20% in conventional lithium-ion batteries. This phenomenon has become increasingly problematic as manufacturers push for higher energy densities and longer cycle life. Prelithiation technology aims to compensate for this initial lithium loss by introducing additional lithium before battery assembly or during the formation process.
Recent research has revealed that prelithiation not only addresses first-cycle capacity loss but significantly influences the stability and morphology of the SEI layer. The SEI serves as a protective barrier between the electrode and electrolyte, determining critical battery parameters including coulombic efficiency, rate capability, and long-term cycling stability. Understanding how prelithiation affects SEI formation mechanisms has become essential for optimizing battery performance.
The technical objectives of prelithiation research focus on several interconnected goals. Primary among these is developing methods to create more stable and uniform SEI layers that minimize continuous lithium consumption during cycling. Researchers aim to establish correlations between prelithiation parameters (amount, timing, method) and resulting SEI properties (thickness, composition, homogeneity, mechanical stability).
Another critical objective involves understanding the molecular-level interactions between prelithiation agents and electrolyte components during SEI formation. This includes investigating how different prelithiation approaches affect the chemical composition and structural organization of the SEI layer across various electrode materials and electrolyte systems.
The technology trend indicates a shift toward prelithiation methods that can be seamlessly integrated into existing manufacturing processes without significant cost increases or safety concerns. This includes developing stable prelithiation materials with long shelf lives and exploring in-situ prelithiation techniques that can be applied during cell assembly rather than requiring additional processing steps.
As battery applications diversify beyond consumer electronics to electric vehicles and grid storage, prelithiation technology must adapt to different performance requirements and operating conditions. The ultimate goal is to develop tailored prelithiation strategies that can be optimized for specific battery chemistries and use cases, maximizing energy density while ensuring safety and longevity.
Market Analysis for Prelithiated Battery Materials
The global market for prelithiated battery materials is experiencing significant growth, driven by the increasing demand for high-performance lithium-ion batteries across various industries. The market size for prelithiated materials was valued at approximately $420 million in 2022 and is projected to reach $1.2 billion by 2028, representing a compound annual growth rate (CAGR) of 19.2% during the forecast period.
Electric vehicles (EVs) constitute the largest application segment for prelithiated battery materials, accounting for over 60% of the market share. This dominance is attributed to the automotive industry's aggressive shift toward electrification, with major manufacturers committing to electric-only product lines within the next decade. The consumer electronics sector represents the second-largest market segment at 25%, followed by energy storage systems at 10%.
Geographically, Asia-Pacific dominates the market with approximately 65% share, led by China, Japan, and South Korea. These countries house major battery manufacturers and have established robust supply chains for battery materials. North America and Europe follow with 20% and 12% market shares respectively, with both regions investing heavily in domestic battery production capabilities to reduce dependence on Asian suppliers.
The market dynamics are significantly influenced by the technical advantages that prelithiation offers in terms of SEI stability and morphology control. Batteries incorporating prelithiated materials demonstrate 15-20% higher initial capacity, 30% improved cycle life, and substantially reduced first-cycle irreversible capacity loss compared to conventional lithium-ion batteries. These performance improvements directly translate to market value, with prelithiated battery cells commanding a 10-15% premium over standard cells.
Key market drivers include stringent vehicle emission regulations, government subsidies for electric vehicles, and increasing consumer demand for longer-lasting electronic devices. The push for higher energy density batteries with faster charging capabilities has further accelerated market growth for advanced prelithiation technologies.
Market challenges include high production costs, scalability issues, and safety concerns related to the handling of prelithiated materials. The price premium for prelithiated materials remains a significant barrier to wider adoption, particularly in price-sensitive market segments. Additionally, the complex manufacturing processes required for effective prelithiation present challenges for mass production.
Emerging opportunities exist in the development of solid-state batteries, where prelithiation techniques can address interface stability issues that currently limit commercialization. The growing market for grid-scale energy storage also presents significant potential for prelithiated materials that can deliver improved cycle life and reduced maintenance requirements.
Electric vehicles (EVs) constitute the largest application segment for prelithiated battery materials, accounting for over 60% of the market share. This dominance is attributed to the automotive industry's aggressive shift toward electrification, with major manufacturers committing to electric-only product lines within the next decade. The consumer electronics sector represents the second-largest market segment at 25%, followed by energy storage systems at 10%.
Geographically, Asia-Pacific dominates the market with approximately 65% share, led by China, Japan, and South Korea. These countries house major battery manufacturers and have established robust supply chains for battery materials. North America and Europe follow with 20% and 12% market shares respectively, with both regions investing heavily in domestic battery production capabilities to reduce dependence on Asian suppliers.
The market dynamics are significantly influenced by the technical advantages that prelithiation offers in terms of SEI stability and morphology control. Batteries incorporating prelithiated materials demonstrate 15-20% higher initial capacity, 30% improved cycle life, and substantially reduced first-cycle irreversible capacity loss compared to conventional lithium-ion batteries. These performance improvements directly translate to market value, with prelithiated battery cells commanding a 10-15% premium over standard cells.
Key market drivers include stringent vehicle emission regulations, government subsidies for electric vehicles, and increasing consumer demand for longer-lasting electronic devices. The push for higher energy density batteries with faster charging capabilities has further accelerated market growth for advanced prelithiation technologies.
Market challenges include high production costs, scalability issues, and safety concerns related to the handling of prelithiated materials. The price premium for prelithiated materials remains a significant barrier to wider adoption, particularly in price-sensitive market segments. Additionally, the complex manufacturing processes required for effective prelithiation present challenges for mass production.
Emerging opportunities exist in the development of solid-state batteries, where prelithiation techniques can address interface stability issues that currently limit commercialization. The growing market for grid-scale energy storage also presents significant potential for prelithiated materials that can deliver improved cycle life and reduced maintenance requirements.
Current Challenges in SEI Formation and Stability
The formation of a stable and effective Solid Electrolyte Interphase (SEI) remains one of the most critical yet challenging aspects in lithium-ion battery development. Despite decades of research, several fundamental challenges persist in achieving optimal SEI formation and stability, particularly when considering prelithiation techniques. The dynamic nature of SEI formation during initial cycling creates inconsistencies in composition and morphology, leading to unpredictable battery performance and degradation patterns.
A primary challenge lies in controlling the chemical composition of the SEI layer. The interphase forms through complex reduction reactions between electrolyte components and the electrode surface, resulting in a heterogeneous structure comprising both organic and inorganic compounds. This chemical complexity makes it difficult to establish direct correlations between prelithiation conditions and resulting SEI properties, hampering systematic optimization efforts.
Morphological control presents another significant hurdle. The thickness, porosity, and uniformity of the SEI layer directly impact lithium-ion transport kinetics and overall battery performance. Current manufacturing processes lack precise control mechanisms to ensure consistent SEI morphology across the entire electrode surface, especially when prelithiation is introduced as an additional variable. This inconsistency leads to localized degradation and performance variations within the same cell.
Temperature sensitivity further complicates SEI stability. The chemical composition and physical properties of the SEI layer exhibit strong temperature dependence, with accelerated degradation occurring at elevated temperatures. This thermal instability becomes particularly problematic when prelithiation alters the initial chemical environment, potentially creating more temperature-sensitive SEI components that compromise long-term cycling stability.
Electrolyte decomposition products continuously evolve during cycling, causing the SEI layer to grow progressively thicker. This growth increases internal resistance and impedes lithium-ion diffusion, ultimately leading to capacity fade. Prelithiation techniques, while beneficial for initial capacity, can sometimes accelerate this growth process by introducing additional reactive lithium species that participate in ongoing SEI formation reactions.
Mechanical stability represents another critical challenge. The SEI layer must withstand volume changes during cycling while maintaining adhesion to the electrode surface. Prelithiation can alter the mechanical properties of the initial SEI layer, potentially making it more susceptible to cracking and delamination during subsequent cycling. These mechanical failures expose fresh electrode surfaces, triggering additional electrolyte decomposition and SEI reformation in a continuous degradation cycle.
Advanced characterization limitations further hinder progress in this field. The nanoscale dimensions and air-sensitive nature of the SEI layer make in-situ and operando analysis extremely challenging. This characterization gap severely limits our understanding of how prelithiation influences real-time SEI formation and evolution processes, forcing researchers to rely heavily on post-mortem analyses that may not capture the dynamic aspects of SEI development.
A primary challenge lies in controlling the chemical composition of the SEI layer. The interphase forms through complex reduction reactions between electrolyte components and the electrode surface, resulting in a heterogeneous structure comprising both organic and inorganic compounds. This chemical complexity makes it difficult to establish direct correlations between prelithiation conditions and resulting SEI properties, hampering systematic optimization efforts.
Morphological control presents another significant hurdle. The thickness, porosity, and uniformity of the SEI layer directly impact lithium-ion transport kinetics and overall battery performance. Current manufacturing processes lack precise control mechanisms to ensure consistent SEI morphology across the entire electrode surface, especially when prelithiation is introduced as an additional variable. This inconsistency leads to localized degradation and performance variations within the same cell.
Temperature sensitivity further complicates SEI stability. The chemical composition and physical properties of the SEI layer exhibit strong temperature dependence, with accelerated degradation occurring at elevated temperatures. This thermal instability becomes particularly problematic when prelithiation alters the initial chemical environment, potentially creating more temperature-sensitive SEI components that compromise long-term cycling stability.
Electrolyte decomposition products continuously evolve during cycling, causing the SEI layer to grow progressively thicker. This growth increases internal resistance and impedes lithium-ion diffusion, ultimately leading to capacity fade. Prelithiation techniques, while beneficial for initial capacity, can sometimes accelerate this growth process by introducing additional reactive lithium species that participate in ongoing SEI formation reactions.
Mechanical stability represents another critical challenge. The SEI layer must withstand volume changes during cycling while maintaining adhesion to the electrode surface. Prelithiation can alter the mechanical properties of the initial SEI layer, potentially making it more susceptible to cracking and delamination during subsequent cycling. These mechanical failures expose fresh electrode surfaces, triggering additional electrolyte decomposition and SEI reformation in a continuous degradation cycle.
Advanced characterization limitations further hinder progress in this field. The nanoscale dimensions and air-sensitive nature of the SEI layer make in-situ and operando analysis extremely challenging. This characterization gap severely limits our understanding of how prelithiation influences real-time SEI formation and evolution processes, forcing researchers to rely heavily on post-mortem analyses that may not capture the dynamic aspects of SEI development.
Current Methodologies for SEI Morphology Control
01 Prelithiation techniques for SEI formation
Various prelithiation techniques can be employed to form a stable solid electrolyte interphase (SEI) layer on electrode materials. These techniques involve introducing lithium ions into electrode materials before cell assembly, which helps to form a protective SEI layer with improved stability and controlled morphology. Prelithiation can compensate for lithium loss during initial cycling and contribute to better battery performance by establishing a robust SEI foundation.- Prelithiation techniques for SEI formation: Various prelithiation techniques can be employed to form a stable solid electrolyte interphase (SEI) layer on electrode materials. These techniques involve introducing lithium ions into electrode materials before cell assembly, which helps to form a protective SEI layer with improved stability and controlled morphology. Prelithiation can be achieved through chemical, electrochemical, or physical methods, resulting in enhanced battery performance and cycle life.
- SEI additives and composition control: The stability and morphology of the SEI layer can be enhanced through the use of specific additives and careful control of its composition. Various organic and inorganic compounds can be incorporated into the electrolyte to promote the formation of a more stable and uniform SEI layer. These additives can influence the chemical composition, thickness, and mechanical properties of the SEI, leading to improved lithium-ion transport and reduced degradation during cycling.
- Morphology control of SEI layer: The morphology of the SEI layer significantly impacts battery performance and stability. Various approaches can be used to control SEI morphology, including temperature regulation during formation, electrolyte composition adjustment, and surface modification of electrode materials. A uniform and compact SEI layer with controlled thickness and porosity can enhance lithium-ion diffusion, reduce impedance, and improve the overall electrochemical performance of the battery.
- Advanced characterization of SEI stability: Advanced analytical techniques are employed to characterize the stability and morphology of the SEI layer formed during prelithiation. These techniques include scanning electron microscopy, transmission electron microscopy, X-ray photoelectron spectroscopy, and electrochemical impedance spectroscopy. Such characterization methods provide insights into the chemical composition, structural features, and degradation mechanisms of the SEI layer, enabling the development of strategies to enhance its stability and performance.
- Novel materials for enhanced SEI stability: Novel electrode and electrolyte materials are being developed to enhance the stability and optimize the morphology of the SEI layer. These materials include advanced silicon-based anodes, lithium-rich cathodes, solid-state electrolytes, and composite materials. By incorporating these novel materials, the formation of a more stable and functional SEI layer can be achieved, leading to improved battery performance, longer cycle life, and enhanced safety characteristics.
02 SEI morphology control through additives and coatings
The morphology of the SEI layer can be controlled through the use of specific additives and surface coatings. These materials can guide the formation of a more uniform and stable SEI structure with desired physical properties. Electrolyte additives can modify the composition and structure of the SEI layer, while protective coatings on electrode surfaces can serve as templates for SEI growth, resulting in improved morphological characteristics and enhanced stability during cycling.Expand Specific Solutions03 Temperature and pressure effects on SEI stability
Temperature and pressure conditions during battery formation and operation significantly impact SEI stability and morphology. Controlled temperature treatments during prelithiation can promote the formation of more crystalline and stable SEI structures. Similarly, applying specific pressure conditions during SEI formation can influence its density, uniformity, and mechanical properties. Optimizing these parameters helps to create an SEI layer with enhanced thermal and mechanical stability.Expand Specific Solutions04 Advanced characterization of SEI morphology and composition
Advanced analytical techniques are employed to characterize the morphology, composition, and stability of SEI layers formed through prelithiation. These include electron microscopy, spectroscopic methods, and in-situ monitoring approaches that provide insights into SEI formation mechanisms and structural evolution. Understanding the relationship between SEI morphology and battery performance enables the development of more effective prelithiation strategies and improved SEI stability.Expand Specific Solutions05 Novel materials for enhanced SEI stability
Novel electrode and electrolyte materials are being developed to enhance SEI stability and optimize its morphology. These include engineered silicon-based anodes, composite cathode materials, and specialized electrolyte formulations designed to form more stable SEI layers. The incorporation of nanostructured materials and functional polymers can also contribute to improved SEI characteristics by providing better mechanical support and chemical stability during battery cycling.Expand Specific Solutions
Leading Companies and Research Institutions in Prelithiation
The prelithiation technology market is currently in a growth phase, with increasing demand driven by the need for improved battery performance in electric vehicles and energy storage systems. The global market size for advanced battery technologies is projected to reach $150 billion by 2025, with prelithiation solutions representing a significant segment. Technologically, the field is in mid-maturity, with companies like LG Energy Solution, BYD, and SK ON leading commercial implementation, while research-focused entities like NanoGraf and Group14 Technologies are advancing silicon-based anode innovations. A123 Systems and PolyPlus Battery are developing proprietary prelithiation methods to enhance SEI stability. Collaboration between academic institutions (University of Texas System, Jiangnan University) and industry players is accelerating technological breakthroughs, particularly in understanding how prelithiation influences long-term battery performance and safety.
BYD Co., Ltd.
Technical Solution: BYD has pioneered a comprehensive prelithiation strategy focused on morphological control of the SEI layer. Their technology employs a dual-phase prelithiation approach where silicon-carbon composite anodes undergo controlled pre-lithiation using a proprietary lithium-containing paste applied before cell assembly. This creates an engineered SEI foundation layer with optimized porosity and thickness. During initial formation cycles, BYD's electrolyte formulation containing film-forming additives builds upon this foundation, creating a multi-layered SEI structure with distinct mechanical and chemical properties. The inner layer provides flexibility to accommodate volume changes, while the outer layer offers chemical stability against electrolyte decomposition. BYD's research indicates this approach reduces first-cycle capacity loss by up to 15% while extending cycle life by preventing continuous SEI growth during operation.
Strengths: Creates mechanically robust SEI structures that accommodate volume changes; significantly reduces first-cycle capacity loss; enables higher silicon content in anodes. Weaknesses: Requires additional manufacturing steps and specialized equipment; process sensitivity to environmental conditions can affect quality consistency; potential safety concerns with handling reactive prelithiation materials.
LG Chem Ltd.
Technical Solution: LG Chem has developed advanced prelithiation techniques to enhance SEI stability in lithium-ion batteries. Their approach involves pre-storing lithium in the anode material before cell assembly, which compensates for the irreversible capacity loss during initial SEI formation. The company employs both ex-situ and in-situ prelithiation methods: ex-situ involves treating anode materials with lithium compounds before cell assembly, while in-situ uses additives that release lithium during the formation process. LG Chem's research has demonstrated that controlled prelithiation creates a more uniform and stable SEI layer, reducing impedance growth during cycling. Their proprietary SAIDATM (Surface-Activated In-situ Designed Anode Technology) process introduces stabilizing compounds that co-intercalate with lithium ions, forming a more elastic and ion-conductive SEI structure that better accommodates volume changes during cycling.
Strengths: Creates more uniform and durable SEI layers that resist cracking during cycling; compensates for first-cycle capacity loss; improves long-term cycling stability. Weaknesses: Adds manufacturing complexity and cost; requires precise control of prelithiation degree to avoid lithium plating; some prelithiation agents are sensitive to air and moisture, requiring specialized handling.
Key Patents and Research on Prelithiation-SEI Interactions
Solid electrolyte interphase (SEI) and a method for its preparation
PatentActiveIN202141055967A
Innovation
- A solid electrolyte interphase (SEI) comprising a porous polymer matrix embedded with metal precursors like lithium fluoride and lithium phosphate, coated on the current collector to prevent dendrite formation and enhance battery performance and safety, prepared through a method involving mixing metal precursors with an electrically conducting polymer and solvent, followed by solvent removal.
Battery Performance Metrics and Testing Standards
Evaluating the impact of prelithiation on SEI stability and morphology requires standardized battery performance metrics and testing protocols. The electrochemical performance of lithium-ion batteries is typically assessed through capacity retention, coulombic efficiency, and impedance measurements, which directly reflect SEI quality and stability.
Capacity retention tests provide critical insights into how prelithiation affects long-term battery performance. Standard protocols involve galvanostatic cycling at various C-rates (0.1C to 2C) for hundreds or thousands of cycles, with periodic reference performance tests to isolate degradation mechanisms. Prelithiated electrodes often demonstrate distinct capacity retention profiles compared to conventional electrodes, necessitating specialized testing parameters.
Coulombic efficiency measurements are particularly valuable for SEI studies, as they directly quantify irreversible lithium consumption. High-precision coulometry techniques with accuracy better than 0.01% have become essential for evaluating subtle differences in SEI formation and evolution between prelithiated and standard electrodes. The first-cycle efficiency improvement is a key metric for prelithiation effectiveness.
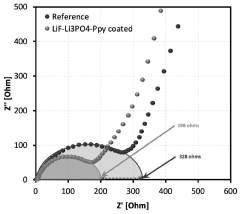
Electrochemical impedance spectroscopy (EIS) provides detailed information about SEI resistance and morphological characteristics. Standard protocols involve measurements at different states of charge and after various cycling intervals. The high-frequency region (100 Hz to 100 kHz) typically reveals SEI-related impedance features, while lower frequencies correspond to charge transfer and diffusion processes.
Differential capacity analysis (dQ/dV) has emerged as a powerful diagnostic tool for identifying voltage signatures associated with SEI formation and evolution. This technique requires precise voltage measurements (±1 mV) and controlled testing conditions to detect subtle changes in the electrochemical profile of prelithiated cells.
Post-mortem analysis standards have also evolved to include specialized protocols for SEI characterization. These include standardized sample preparation methods for electron microscopy, X-ray photoelectron spectroscopy, and time-of-flight secondary ion mass spectrometry. Maintaining consistent environmental conditions during sample transfer is critical, as SEI components are highly sensitive to air and moisture exposure.
Temperature-dependent performance metrics are increasingly important, as SEI stability is strongly influenced by thermal conditions. Accelerated aging tests at elevated temperatures (45-60°C) following standards like IEC 62660 provide valuable insights into the thermal stability advantages of prelithiated electrodes compared to conventional formulations.
Capacity retention tests provide critical insights into how prelithiation affects long-term battery performance. Standard protocols involve galvanostatic cycling at various C-rates (0.1C to 2C) for hundreds or thousands of cycles, with periodic reference performance tests to isolate degradation mechanisms. Prelithiated electrodes often demonstrate distinct capacity retention profiles compared to conventional electrodes, necessitating specialized testing parameters.
Coulombic efficiency measurements are particularly valuable for SEI studies, as they directly quantify irreversible lithium consumption. High-precision coulometry techniques with accuracy better than 0.01% have become essential for evaluating subtle differences in SEI formation and evolution between prelithiated and standard electrodes. The first-cycle efficiency improvement is a key metric for prelithiation effectiveness.
Electrochemical impedance spectroscopy (EIS) provides detailed information about SEI resistance and morphological characteristics. Standard protocols involve measurements at different states of charge and after various cycling intervals. The high-frequency region (100 Hz to 100 kHz) typically reveals SEI-related impedance features, while lower frequencies correspond to charge transfer and diffusion processes.
Differential capacity analysis (dQ/dV) has emerged as a powerful diagnostic tool for identifying voltage signatures associated with SEI formation and evolution. This technique requires precise voltage measurements (±1 mV) and controlled testing conditions to detect subtle changes in the electrochemical profile of prelithiated cells.
Post-mortem analysis standards have also evolved to include specialized protocols for SEI characterization. These include standardized sample preparation methods for electron microscopy, X-ray photoelectron spectroscopy, and time-of-flight secondary ion mass spectrometry. Maintaining consistent environmental conditions during sample transfer is critical, as SEI components are highly sensitive to air and moisture exposure.
Temperature-dependent performance metrics are increasingly important, as SEI stability is strongly influenced by thermal conditions. Accelerated aging tests at elevated temperatures (45-60°C) following standards like IEC 62660 provide valuable insights into the thermal stability advantages of prelithiated electrodes compared to conventional formulations.
Environmental Impact of Prelithiation Processes
Prelithiation processes, while beneficial for battery performance, carry significant environmental implications that warrant careful consideration. The manufacturing of prelithiation materials often involves energy-intensive processes and the use of reactive chemicals such as lithium metal, lithium-containing compounds, and various solvents. These processes contribute to carbon emissions, particularly when powered by non-renewable energy sources, exacerbating climate change concerns in the battery production ecosystem.
The extraction of lithium itself presents substantial environmental challenges. Mining operations for lithium, whether from hard rock sources or brine deposits, can lead to habitat destruction, soil degradation, and water pollution. Brine extraction methods, common in South America, consume vast quantities of water in often water-scarce regions, potentially disrupting local ecosystems and agricultural activities. These environmental costs must be weighed against the performance benefits that prelithiation provides.
Waste management represents another critical environmental dimension of prelithiation processes. The reactive nature of many prelithiation materials necessitates specialized handling and disposal protocols. Improper management can result in hazardous waste contamination, posing risks to both environmental and human health. Additionally, the production of prelithiated materials often generates byproducts that require appropriate treatment before disposal.
From a life-cycle perspective, prelithiation processes present a complex environmental trade-off. While they may extend battery life and improve performance—potentially reducing the overall environmental footprint of battery usage—the initial manufacturing impact is heightened. This creates a tension between immediate environmental costs and long-term sustainability benefits that must be carefully evaluated through comprehensive life-cycle assessment methodologies.
Recent innovations are addressing these environmental concerns through the development of greener prelithiation techniques. Water-based processing methods, ambient temperature reactions, and the use of less hazardous lithium sources represent promising approaches to reducing environmental impact. Additionally, closed-loop manufacturing systems that recapture and reuse materials are emerging as potential solutions to minimize waste generation.
Regulatory frameworks worldwide are increasingly focusing on the environmental aspects of battery production, including prelithiation processes. Manufacturers are facing growing pressure to document and reduce the environmental footprint of their operations, driving innovation toward more sustainable practices throughout the battery production chain.
The extraction of lithium itself presents substantial environmental challenges. Mining operations for lithium, whether from hard rock sources or brine deposits, can lead to habitat destruction, soil degradation, and water pollution. Brine extraction methods, common in South America, consume vast quantities of water in often water-scarce regions, potentially disrupting local ecosystems and agricultural activities. These environmental costs must be weighed against the performance benefits that prelithiation provides.
Waste management represents another critical environmental dimension of prelithiation processes. The reactive nature of many prelithiation materials necessitates specialized handling and disposal protocols. Improper management can result in hazardous waste contamination, posing risks to both environmental and human health. Additionally, the production of prelithiated materials often generates byproducts that require appropriate treatment before disposal.
From a life-cycle perspective, prelithiation processes present a complex environmental trade-off. While they may extend battery life and improve performance—potentially reducing the overall environmental footprint of battery usage—the initial manufacturing impact is heightened. This creates a tension between immediate environmental costs and long-term sustainability benefits that must be carefully evaluated through comprehensive life-cycle assessment methodologies.
Recent innovations are addressing these environmental concerns through the development of greener prelithiation techniques. Water-based processing methods, ambient temperature reactions, and the use of less hazardous lithium sources represent promising approaches to reducing environmental impact. Additionally, closed-loop manufacturing systems that recapture and reuse materials are emerging as potential solutions to minimize waste generation.
Regulatory frameworks worldwide are increasingly focusing on the environmental aspects of battery production, including prelithiation processes. Manufacturers are facing growing pressure to document and reduce the environmental footprint of their operations, driving innovation toward more sustainable practices throughout the battery production chain.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!