How Surface Functional Groups Modulate Hydroxyapatite's Interaction with Proteins
JUL 23, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydroxyapatite-Protein Interaction Background
Hydroxyapatite (HAp) is a naturally occurring calcium phosphate mineral that forms the primary inorganic component of bone and teeth. Its unique properties, including biocompatibility, osteoconductivity, and the ability to form strong bonds with surrounding tissues, have made it a subject of intense research in the fields of biomaterials and regenerative medicine. The interaction between HAp and proteins plays a crucial role in various biological processes, including bone formation, remodeling, and the integration of implants.
The surface of HAp is characterized by a complex arrangement of functional groups, primarily consisting of calcium ions, phosphate groups, and hydroxyl groups. These surface features are responsible for the material's ability to interact with proteins and other biomolecules. The nature and distribution of these functional groups can significantly influence the adsorption, conformation, and activity of proteins on the HAp surface.
Proteins, as complex macromolecules, possess a variety of functional groups and structural elements that can interact with the HAp surface. These interactions are governed by a combination of electrostatic forces, hydrogen bonding, van der Waals forces, and hydrophobic interactions. The specific arrangement and properties of the functional groups on both the HAp surface and the protein determine the strength and nature of their interaction.
Understanding the mechanisms by which surface functional groups modulate HAp-protein interactions is crucial for several reasons. Firstly, it provides insights into the fundamental processes of biomineralization and bone formation. Secondly, it enables the development of improved biomaterials for applications such as bone grafts, dental implants, and drug delivery systems. Lastly, it allows for the manipulation of protein adsorption and activity on HAp surfaces, which can be leveraged to enhance biocompatibility and promote desired biological responses.
Research in this area has focused on elucidating the role of specific functional groups in protein adsorption and the subsequent effects on protein structure and function. Studies have investigated how variations in surface charge, hydrophobicity, and the presence of specific chemical moieties influence the binding affinity and orientation of proteins on HAp surfaces. Additionally, efforts have been made to modify HAp surfaces to achieve desired protein interactions, either through chemical treatments or the incorporation of bioactive molecules.
The complexity of HAp-protein interactions presents both challenges and opportunities for researchers. While the multitude of factors involved makes it difficult to predict and control these interactions precisely, it also offers the potential for fine-tuning material properties to achieve specific biological outcomes. As research in this field progresses, it promises to yield valuable insights that can be applied to the development of advanced biomaterials and therapeutic strategies.
The surface of HAp is characterized by a complex arrangement of functional groups, primarily consisting of calcium ions, phosphate groups, and hydroxyl groups. These surface features are responsible for the material's ability to interact with proteins and other biomolecules. The nature and distribution of these functional groups can significantly influence the adsorption, conformation, and activity of proteins on the HAp surface.
Proteins, as complex macromolecules, possess a variety of functional groups and structural elements that can interact with the HAp surface. These interactions are governed by a combination of electrostatic forces, hydrogen bonding, van der Waals forces, and hydrophobic interactions. The specific arrangement and properties of the functional groups on both the HAp surface and the protein determine the strength and nature of their interaction.
Understanding the mechanisms by which surface functional groups modulate HAp-protein interactions is crucial for several reasons. Firstly, it provides insights into the fundamental processes of biomineralization and bone formation. Secondly, it enables the development of improved biomaterials for applications such as bone grafts, dental implants, and drug delivery systems. Lastly, it allows for the manipulation of protein adsorption and activity on HAp surfaces, which can be leveraged to enhance biocompatibility and promote desired biological responses.
Research in this area has focused on elucidating the role of specific functional groups in protein adsorption and the subsequent effects on protein structure and function. Studies have investigated how variations in surface charge, hydrophobicity, and the presence of specific chemical moieties influence the binding affinity and orientation of proteins on HAp surfaces. Additionally, efforts have been made to modify HAp surfaces to achieve desired protein interactions, either through chemical treatments or the incorporation of bioactive molecules.
The complexity of HAp-protein interactions presents both challenges and opportunities for researchers. While the multitude of factors involved makes it difficult to predict and control these interactions precisely, it also offers the potential for fine-tuning material properties to achieve specific biological outcomes. As research in this field progresses, it promises to yield valuable insights that can be applied to the development of advanced biomaterials and therapeutic strategies.
Market Demand Analysis
The market demand for understanding how surface functional groups modulate hydroxyapatite's interaction with proteins is driven by several key factors in the biomedical and materials science industries. This research area has significant implications for the development of advanced biomaterials, particularly in orthopedic and dental applications.
In the orthopedic implant market, which is projected to reach $64 billion by 2026, there is a growing need for improved implant materials that can better integrate with the body's natural tissues. The ability to control protein interactions with hydroxyapatite surfaces is crucial for enhancing implant biocompatibility and reducing rejection rates. This has led to increased investment in research and development focused on surface functionalization techniques.
The dental implant sector, another major market driver, is expected to grow at a CAGR of 9.7% from 2021 to 2028. Dental professionals are seeking implant materials that promote faster osseointegration and reduce healing times. Understanding protein-hydroxyapatite interactions at the molecular level can lead to the development of more effective dental implants, driving demand for this specialized knowledge.
In the field of drug delivery systems, there is a rising interest in using functionalized hydroxyapatite nanoparticles as carriers for therapeutic proteins. This application requires a deep understanding of how surface modifications affect protein binding and release kinetics. The global drug delivery market, valued at $1,430 billion in 2021, is expected to benefit significantly from advancements in this area.
The biomaterials market, which encompasses various applications of hydroxyapatite, is projected to reach $297 billion by 2028. This growth is partly fueled by the increasing demand for personalized medicine and tissue engineering solutions. Researchers and companies in this space are actively seeking ways to fine-tune material properties through surface modifications, creating a strong market pull for studies on protein-hydroxyapatite interactions.
Academic and industrial research institutions are allocating substantial resources to this field, recognizing its potential to revolutionize biomaterial design. The number of publications and patents related to surface functionalization of hydroxyapatite has shown a steady increase over the past decade, indicating a growing scientific and commercial interest.
In conclusion, the market demand for research on how surface functional groups modulate hydroxyapatite's interaction with proteins is robust and multifaceted. It is driven by the needs of the orthopedic and dental implant industries, the potential for advanced drug delivery systems, and the broader biomaterials market. As these sectors continue to grow and evolve, the demand for innovative solutions based on this fundamental research is expected to intensify, creating opportunities for both academic discoveries and commercial applications.
In the orthopedic implant market, which is projected to reach $64 billion by 2026, there is a growing need for improved implant materials that can better integrate with the body's natural tissues. The ability to control protein interactions with hydroxyapatite surfaces is crucial for enhancing implant biocompatibility and reducing rejection rates. This has led to increased investment in research and development focused on surface functionalization techniques.
The dental implant sector, another major market driver, is expected to grow at a CAGR of 9.7% from 2021 to 2028. Dental professionals are seeking implant materials that promote faster osseointegration and reduce healing times. Understanding protein-hydroxyapatite interactions at the molecular level can lead to the development of more effective dental implants, driving demand for this specialized knowledge.
In the field of drug delivery systems, there is a rising interest in using functionalized hydroxyapatite nanoparticles as carriers for therapeutic proteins. This application requires a deep understanding of how surface modifications affect protein binding and release kinetics. The global drug delivery market, valued at $1,430 billion in 2021, is expected to benefit significantly from advancements in this area.
The biomaterials market, which encompasses various applications of hydroxyapatite, is projected to reach $297 billion by 2028. This growth is partly fueled by the increasing demand for personalized medicine and tissue engineering solutions. Researchers and companies in this space are actively seeking ways to fine-tune material properties through surface modifications, creating a strong market pull for studies on protein-hydroxyapatite interactions.
Academic and industrial research institutions are allocating substantial resources to this field, recognizing its potential to revolutionize biomaterial design. The number of publications and patents related to surface functionalization of hydroxyapatite has shown a steady increase over the past decade, indicating a growing scientific and commercial interest.
In conclusion, the market demand for research on how surface functional groups modulate hydroxyapatite's interaction with proteins is robust and multifaceted. It is driven by the needs of the orthopedic and dental implant industries, the potential for advanced drug delivery systems, and the broader biomaterials market. As these sectors continue to grow and evolve, the demand for innovative solutions based on this fundamental research is expected to intensify, creating opportunities for both academic discoveries and commercial applications.
Current Challenges
The interaction between hydroxyapatite (HAp) and proteins is a complex process influenced by various factors, including surface functional groups. Despite significant advancements in understanding this interaction, several challenges persist in fully elucidating the mechanisms and optimizing the performance of HAp-based materials in biomedical applications.
One of the primary challenges is the heterogeneity of protein structures and their diverse binding affinities to HAp surfaces. Proteins exhibit a wide range of sizes, shapes, and surface chemistries, making it difficult to predict and control their interactions with HAp. This variability complicates the design of HAp-based materials for specific applications, such as drug delivery systems or bone tissue engineering scaffolds.
Another significant challenge is the dynamic nature of the HAp-protein interface. The surface functional groups on HAp can undergo changes in response to environmental factors, such as pH, temperature, and ionic strength. These changes can alter the binding properties of proteins, leading to unpredictable behavior in biological systems. Understanding and controlling these dynamic interactions remains a major hurdle in developing stable and effective HAp-based biomaterials.
The complexity of the biological environment also poses challenges in studying HAp-protein interactions. In vivo, multiple proteins compete for binding sites on HAp surfaces, and the presence of other biomolecules and ions can influence these interactions. Replicating these complex conditions in laboratory settings to obtain accurate and relevant data is challenging, limiting our ability to translate findings from in vitro studies to practical applications.
Furthermore, there is a lack of standardized methods for characterizing and quantifying HAp-protein interactions. Different research groups often employ varied techniques and experimental conditions, making it difficult to compare results across studies and draw definitive conclusions. This inconsistency hinders the development of a comprehensive understanding of how surface functional groups modulate these interactions.
The multiscale nature of HAp-protein interactions presents another challenge. While molecular-level interactions between specific functional groups and protein residues are crucial, the overall performance of HAp-based materials also depends on macro-scale properties such as surface topography and porosity. Bridging the gap between these different scales and understanding their interplay remains a significant research challenge.
Lastly, the development of computational models that accurately predict HAp-protein interactions based on surface functional groups is still in its infancy. Current models often struggle to account for the complexity and dynamic nature of these interactions, limiting their predictive power and usefulness in material design and optimization.
One of the primary challenges is the heterogeneity of protein structures and their diverse binding affinities to HAp surfaces. Proteins exhibit a wide range of sizes, shapes, and surface chemistries, making it difficult to predict and control their interactions with HAp. This variability complicates the design of HAp-based materials for specific applications, such as drug delivery systems or bone tissue engineering scaffolds.
Another significant challenge is the dynamic nature of the HAp-protein interface. The surface functional groups on HAp can undergo changes in response to environmental factors, such as pH, temperature, and ionic strength. These changes can alter the binding properties of proteins, leading to unpredictable behavior in biological systems. Understanding and controlling these dynamic interactions remains a major hurdle in developing stable and effective HAp-based biomaterials.
The complexity of the biological environment also poses challenges in studying HAp-protein interactions. In vivo, multiple proteins compete for binding sites on HAp surfaces, and the presence of other biomolecules and ions can influence these interactions. Replicating these complex conditions in laboratory settings to obtain accurate and relevant data is challenging, limiting our ability to translate findings from in vitro studies to practical applications.
Furthermore, there is a lack of standardized methods for characterizing and quantifying HAp-protein interactions. Different research groups often employ varied techniques and experimental conditions, making it difficult to compare results across studies and draw definitive conclusions. This inconsistency hinders the development of a comprehensive understanding of how surface functional groups modulate these interactions.
The multiscale nature of HAp-protein interactions presents another challenge. While molecular-level interactions between specific functional groups and protein residues are crucial, the overall performance of HAp-based materials also depends on macro-scale properties such as surface topography and porosity. Bridging the gap between these different scales and understanding their interplay remains a significant research challenge.
Lastly, the development of computational models that accurately predict HAp-protein interactions based on surface functional groups is still in its infancy. Current models often struggle to account for the complexity and dynamic nature of these interactions, limiting their predictive power and usefulness in material design and optimization.
Existing Modulation Techniques
01 Protein adsorption on hydroxyapatite surfaces
Hydroxyapatite interacts with proteins through adsorption processes on its surface. This interaction is crucial for understanding biomineralization, bone formation, and the development of biomaterials. The adsorption behavior depends on various factors such as protein structure, surface properties of hydroxyapatite, and environmental conditions.- Protein adsorption on hydroxyapatite surfaces: Hydroxyapatite surfaces interact with proteins through adsorption mechanisms. This interaction is crucial for understanding biomineralization processes and developing biocompatible materials. The adsorption behavior depends on various factors such as protein structure, surface properties of hydroxyapatite, and environmental conditions.
- Hydroxyapatite-protein composites for biomedical applications: Combining hydroxyapatite with specific proteins creates composite materials with enhanced biocompatibility and functionality. These composites find applications in bone tissue engineering, drug delivery systems, and dental materials. The interaction between hydroxyapatite and proteins in these composites influences their mechanical properties and biological performance.
- Molecular modeling of hydroxyapatite-protein interactions: Computational methods are employed to study the molecular-level interactions between hydroxyapatite and proteins. These simulations provide insights into binding mechanisms, conformational changes, and energetics of the interactions. Such models help in predicting and optimizing the behavior of hydroxyapatite-based biomaterials.
- Protein-mediated hydroxyapatite formation and growth: Certain proteins play a crucial role in the nucleation and growth of hydroxyapatite crystals. This process is fundamental in bone and tooth formation. Understanding these protein-mediated mechanisms aids in developing strategies for bone regeneration and treating mineralization disorders.
- Analytical techniques for studying hydroxyapatite-protein interactions: Various analytical methods are used to investigate the interactions between hydroxyapatite and proteins. These include spectroscopic techniques, microscopy, chromatography, and surface analysis methods. These techniques provide valuable information about binding kinetics, structural changes, and the nature of the interactions at the molecular level.
02 Hydroxyapatite-protein composites for biomedical applications
The interaction between hydroxyapatite and proteins is utilized to create composite materials for various biomedical applications. These composites can enhance bone regeneration, improve implant integration, and serve as drug delivery systems. The synergistic effects of hydroxyapatite and proteins contribute to improved biocompatibility and functionality of these materials.Expand Specific Solutions03 Molecular modeling of hydroxyapatite-protein interactions
Computational methods are employed to study the molecular interactions between hydroxyapatite and proteins. These models help predict binding affinities, identify key interaction sites, and understand the mechanisms of protein adsorption on hydroxyapatite surfaces. Such insights are valuable for designing novel biomaterials and optimizing existing ones.Expand Specific Solutions04 Influence of hydroxyapatite-protein interactions on biomineralization
The interaction between hydroxyapatite and proteins plays a crucial role in biomineralization processes, particularly in bone and tooth formation. Proteins can modulate the nucleation, growth, and morphology of hydroxyapatite crystals. Understanding these interactions is essential for developing strategies to control and enhance biomineralization in various applications.Expand Specific Solutions05 Characterization techniques for hydroxyapatite-protein interactions
Various analytical techniques are used to study the interactions between hydroxyapatite and proteins. These include spectroscopic methods, microscopy, calorimetry, and chromatography. Advanced characterization methods help elucidate the binding mechanisms, quantify adsorption parameters, and assess the structural changes in both hydroxyapatite and proteins upon interaction.Expand Specific Solutions
Key Industry Players
The field of surface functional group modulation of hydroxyapatite's protein interactions is in a nascent stage, with significant potential for growth. The market size is relatively small but expanding, driven by applications in biomaterials and drug delivery. Technological maturity is moderate, with ongoing research to optimize protein-material interactions. Key players like Bio-Rad Laboratories, Pfizer, and L'Oréal are investing in R&D, while academic institutions such as Sichuan University and Zhejiang University contribute to fundamental research. The competitive landscape is diverse, with pharmaceutical companies, materials science firms, and research institutions collaborating and competing to advance the technology.
Sichuan University
Technical Solution: Sichuan University has developed a novel approach to modulate hydroxyapatite's interaction with proteins by tailoring surface functional groups. Their research focuses on creating bioactive hydroxyapatite nanoparticles with controlled surface chemistry. They have successfully synthesized hydroxyapatite with various functional groups such as -OH, -COOH, and -NH2 on the surface[1]. These functionalized nanoparticles demonstrate enhanced protein adsorption capacity and improved biocompatibility. The university's team has also investigated the impact of surface charge and hydrophilicity on protein-hydroxyapatite interactions, providing valuable insights for biomaterial design in tissue engineering and drug delivery applications[2].
Strengths: Precise control over surface chemistry, enhanced protein adsorption, improved biocompatibility. Weaknesses: Potential scalability issues for large-scale production, need for further in vivo studies to validate long-term effects.
Zhejiang University
Technical Solution: Zhejiang University has made significant strides in understanding and manipulating hydroxyapatite-protein interactions through surface modification. Their research team has developed a series of hydroxyapatite nanoparticles with gradient surface properties, allowing for fine-tuned protein adsorption and release profiles[3]. They have employed advanced surface characterization techniques, including X-ray photoelectron spectroscopy (XPS) and atomic force microscopy (AFM), to elucidate the mechanisms of protein-surface interactions at the molecular level. Additionally, Zhejiang University researchers have explored the use of biomimetic coatings inspired by natural bone mineralization processes to enhance the bioactivity of hydroxyapatite implants[4].
Strengths: Advanced surface characterization capabilities, biomimetic approach to enhance bioactivity. Weaknesses: Complexity in reproducing gradient surface properties consistently, potential regulatory challenges for novel biomimetic coatings.
Core Innovations
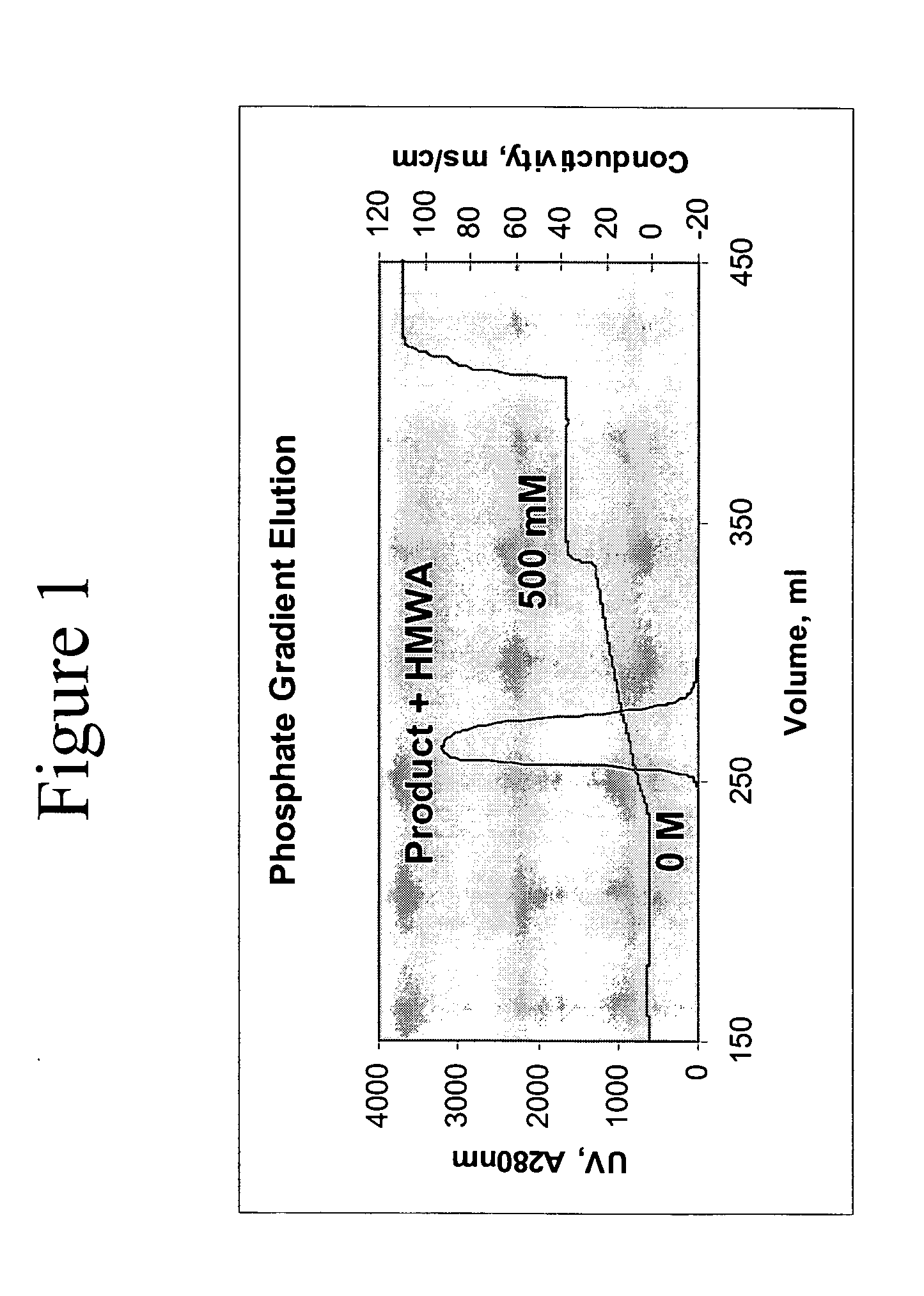
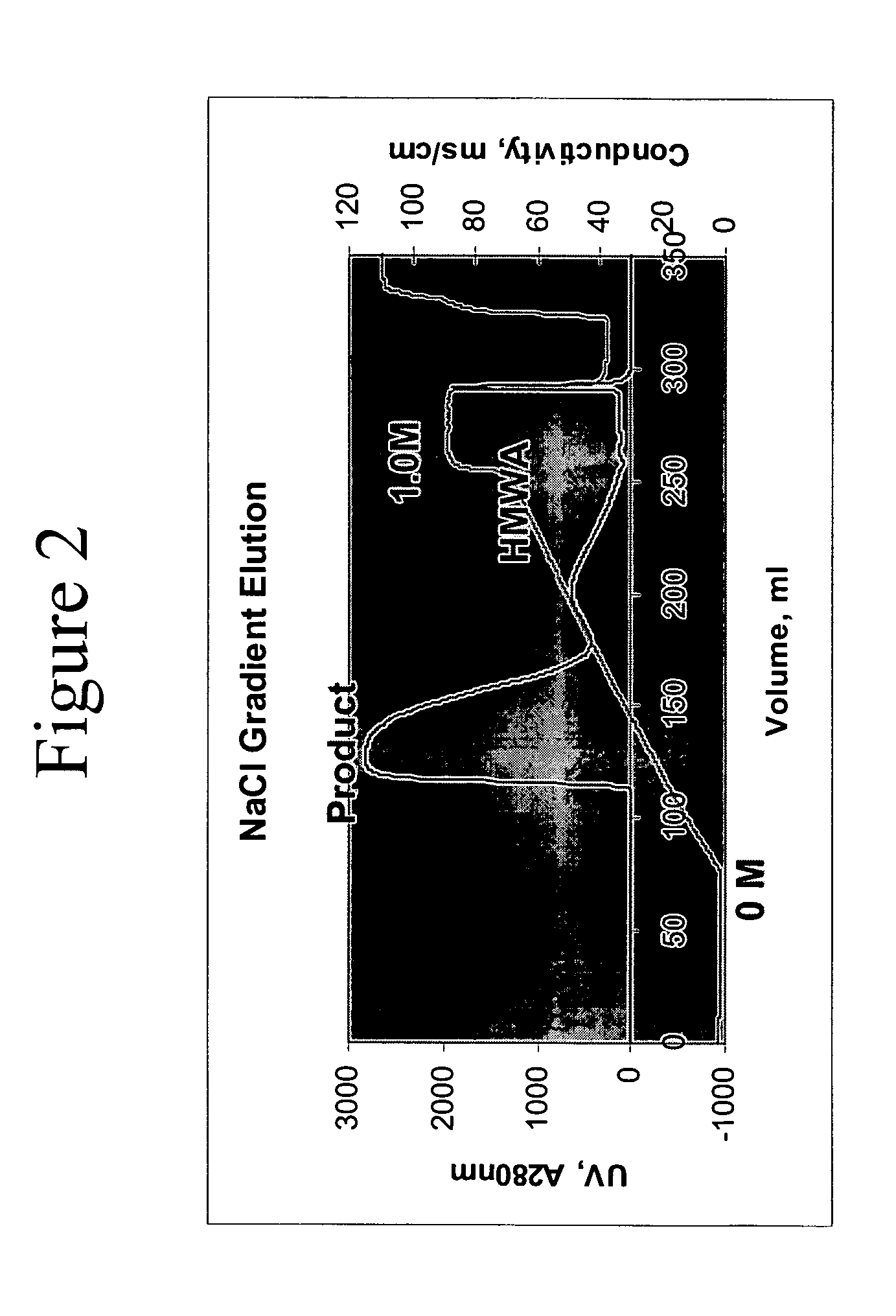
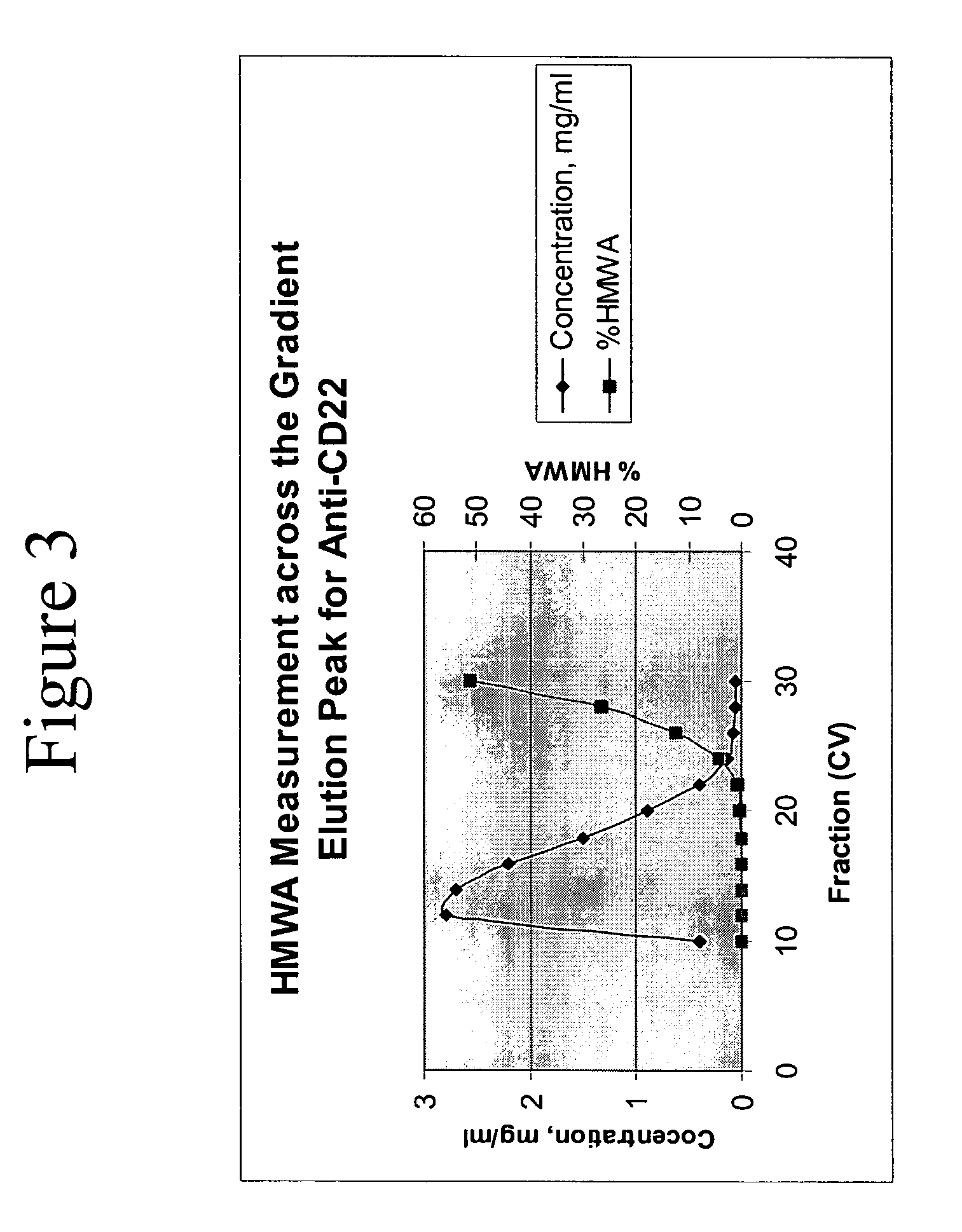
Removal of high molecular weight aggregates using hydroxyapatite chromatography
PatentActiveUS20050107594A1
Innovation
- A novel hydroxyapatite chromatography method using a buffer with 1 to 20 mM sodium phosphate and 0.2 to 2.5 M NaCl at pH 6.4 to 7.6 for elution, allowing for the selective removal of high molecular weight aggregates from antibody preparations without compromising the biological activity of the antibodies.
Biomolecule separation using apatite
PatentWO2016009221A1
Innovation
- The method employs a differential transit rate separation technique using an apatite matrix, where the biomolecule of interest and contaminant species do not adsorb to the matrix, allowing for their separation through bulk flow-through without desorption, utilizing a buffered solution throughout the process to maintain consistency.
Biocompatibility Considerations
The biocompatibility of hydroxyapatite (HA) and its interactions with proteins are crucial considerations in the development of biomaterials for medical applications. The surface functional groups of HA play a significant role in modulating these interactions, which directly impact the material's biocompatibility and performance in biological environments.
Surface chemistry is a key factor in determining how HA interacts with proteins and cells. The presence of specific functional groups on the HA surface, such as hydroxyl, phosphate, and calcium ions, can influence protein adsorption, cell adhesion, and overall biocompatibility. These interactions are governed by various forces, including electrostatic attractions, hydrogen bonding, and van der Waals forces.
The adsorption of proteins onto HA surfaces is a complex process that depends on the properties of both the protein and the HA surface. Proteins with different charges, sizes, and conformations may interact differently with HA surfaces bearing various functional groups. This selective adsorption can influence the subsequent cellular response and tissue integration.
Cell adhesion and proliferation on HA surfaces are also affected by the surface functional groups. Certain functional groups may promote the attachment and growth of specific cell types, while others may inhibit cellular interactions. This selectivity is crucial in designing HA-based materials for targeted applications, such as bone regeneration or drug delivery systems.
The biocompatibility of HA can be further enhanced by modifying its surface functional groups. Techniques such as plasma treatment, chemical functionalization, or coating with bioactive molecules can alter the surface properties of HA to improve its interactions with proteins and cells. These modifications can lead to improved osseointegration, reduced inflammatory responses, and enhanced overall biocompatibility.
Understanding the relationship between surface functional groups and protein interactions is essential for predicting and controlling the in vivo performance of HA-based materials. This knowledge can be leveraged to design biomaterials with tailored surface properties for specific medical applications, optimizing their biocompatibility and therapeutic efficacy.
Long-term biocompatibility considerations must also account for the potential degradation of HA and changes in surface functional groups over time. The dynamic nature of these interactions in physiological environments can affect the material's performance and integration with surrounding tissues. Therefore, studying the stability and evolution of surface functional groups under biological conditions is crucial for developing reliable and effective HA-based biomaterials.
Surface chemistry is a key factor in determining how HA interacts with proteins and cells. The presence of specific functional groups on the HA surface, such as hydroxyl, phosphate, and calcium ions, can influence protein adsorption, cell adhesion, and overall biocompatibility. These interactions are governed by various forces, including electrostatic attractions, hydrogen bonding, and van der Waals forces.
The adsorption of proteins onto HA surfaces is a complex process that depends on the properties of both the protein and the HA surface. Proteins with different charges, sizes, and conformations may interact differently with HA surfaces bearing various functional groups. This selective adsorption can influence the subsequent cellular response and tissue integration.
Cell adhesion and proliferation on HA surfaces are also affected by the surface functional groups. Certain functional groups may promote the attachment and growth of specific cell types, while others may inhibit cellular interactions. This selectivity is crucial in designing HA-based materials for targeted applications, such as bone regeneration or drug delivery systems.
The biocompatibility of HA can be further enhanced by modifying its surface functional groups. Techniques such as plasma treatment, chemical functionalization, or coating with bioactive molecules can alter the surface properties of HA to improve its interactions with proteins and cells. These modifications can lead to improved osseointegration, reduced inflammatory responses, and enhanced overall biocompatibility.
Understanding the relationship between surface functional groups and protein interactions is essential for predicting and controlling the in vivo performance of HA-based materials. This knowledge can be leveraged to design biomaterials with tailored surface properties for specific medical applications, optimizing their biocompatibility and therapeutic efficacy.
Long-term biocompatibility considerations must also account for the potential degradation of HA and changes in surface functional groups over time. The dynamic nature of these interactions in physiological environments can affect the material's performance and integration with surrounding tissues. Therefore, studying the stability and evolution of surface functional groups under biological conditions is crucial for developing reliable and effective HA-based biomaterials.
Regulatory Landscape
The regulatory landscape surrounding the interaction between hydroxyapatite (HAp) and proteins is complex and multifaceted, involving various governmental agencies and international organizations. This landscape is primarily shaped by the applications of HAp in biomedical fields, particularly in bone tissue engineering and drug delivery systems.
In the United States, the Food and Drug Administration (FDA) plays a crucial role in regulating HAp-based medical devices and implants. The FDA's Center for Devices and Radiological Health (CDRH) oversees the approval process for such products, ensuring their safety and efficacy. The regulatory pathway for HAp-based devices often falls under the 510(k) premarket notification process, requiring manufacturers to demonstrate substantial equivalence to a predicate device.
The European Union has implemented the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR), which came into full effect in May 2021. These regulations have significant implications for HAp-based products, imposing stricter requirements for clinical evidence and post-market surveillance. Manufacturers must comply with these regulations to obtain CE marking for their products.
In Japan, the Pharmaceuticals and Medical Devices Agency (PMDA) regulates HAp-based medical devices. The PMDA has established specific guidelines for the evaluation of orthopedic and dental implants, which often incorporate HAp coatings or composites.
International standards organizations, such as the International Organization for Standardization (ISO) and ASTM International, have developed specific standards for testing and characterizing HAp materials. These standards, including ISO 13779 and ASTM F1185, provide guidelines for assessing the physical, chemical, and biological properties of HAp coatings and bulk materials.
Regulatory bodies are increasingly focusing on the potential environmental impact of HAp production and disposal. This has led to the development of guidelines for sustainable manufacturing practices and proper disposal methods for HAp-containing products.
As research continues to uncover the complexities of HAp-protein interactions, regulatory agencies are likely to update their guidelines to address emerging concerns. This may include more stringent requirements for characterizing surface functional groups and their effects on protein adsorption, as well as assessing the long-term stability of HAp-protein complexes in physiological environments.
In the United States, the Food and Drug Administration (FDA) plays a crucial role in regulating HAp-based medical devices and implants. The FDA's Center for Devices and Radiological Health (CDRH) oversees the approval process for such products, ensuring their safety and efficacy. The regulatory pathway for HAp-based devices often falls under the 510(k) premarket notification process, requiring manufacturers to demonstrate substantial equivalence to a predicate device.
The European Union has implemented the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR), which came into full effect in May 2021. These regulations have significant implications for HAp-based products, imposing stricter requirements for clinical evidence and post-market surveillance. Manufacturers must comply with these regulations to obtain CE marking for their products.
In Japan, the Pharmaceuticals and Medical Devices Agency (PMDA) regulates HAp-based medical devices. The PMDA has established specific guidelines for the evaluation of orthopedic and dental implants, which often incorporate HAp coatings or composites.
International standards organizations, such as the International Organization for Standardization (ISO) and ASTM International, have developed specific standards for testing and characterizing HAp materials. These standards, including ISO 13779 and ASTM F1185, provide guidelines for assessing the physical, chemical, and biological properties of HAp coatings and bulk materials.
Regulatory bodies are increasingly focusing on the potential environmental impact of HAp production and disposal. This has led to the development of guidelines for sustainable manufacturing practices and proper disposal methods for HAp-containing products.
As research continues to uncover the complexities of HAp-protein interactions, regulatory agencies are likely to update their guidelines to address emerging concerns. This may include more stringent requirements for characterizing surface functional groups and their effects on protein adsorption, as well as assessing the long-term stability of HAp-protein complexes in physiological environments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!