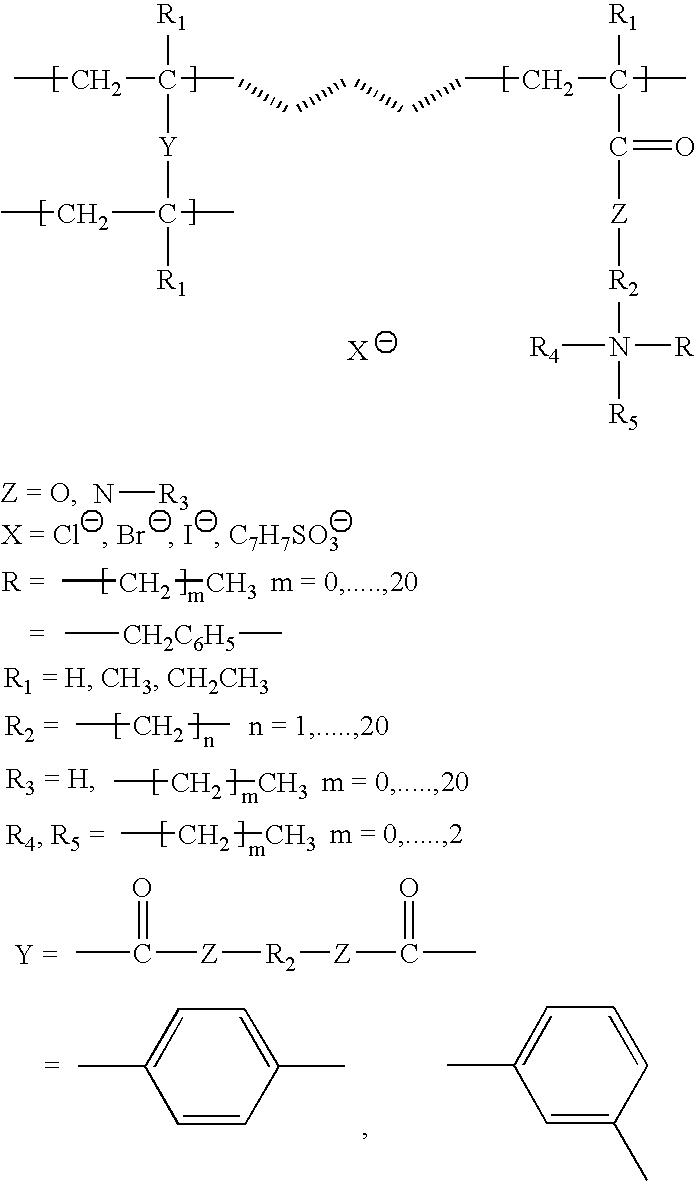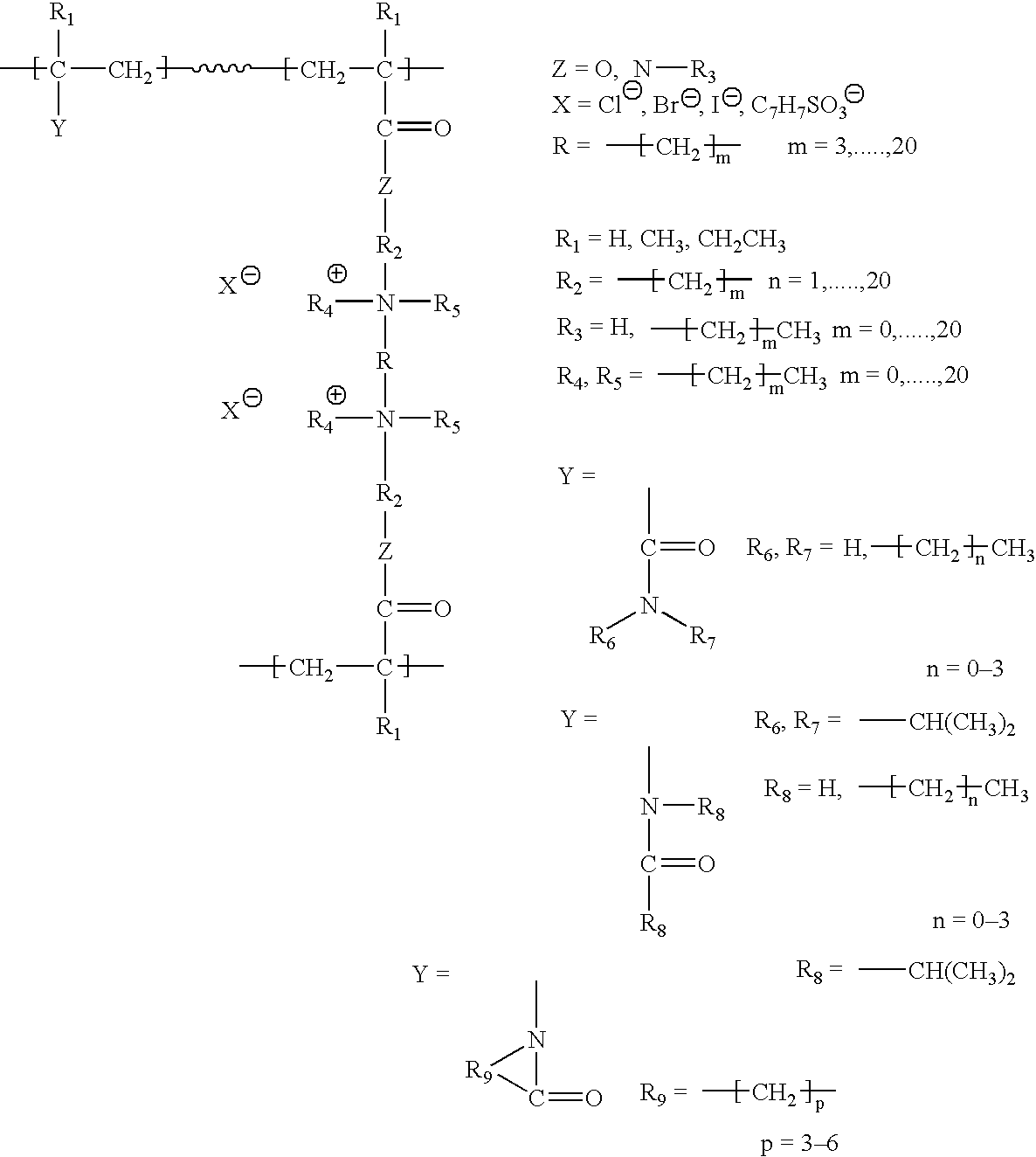How to Develop COFs for Selective Anion Exchange
APR 16, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
COF Anion Exchange Background and Objectives
Covalent Organic Frameworks (COFs) represent a revolutionary class of crystalline porous materials that have emerged as promising candidates for selective anion exchange applications. These materials are constructed through the formation of strong covalent bonds between organic building blocks, resulting in highly ordered, periodic structures with predictable pore architectures and tunable surface functionalities. The unique combination of structural stability, porosity, and chemical versatility positions COFs as next-generation materials for addressing critical challenges in environmental remediation, water treatment, and separation technologies.
The development of COFs for selective anion exchange has gained significant momentum due to the pressing need for efficient removal of harmful anions from contaminated water sources. Traditional anion exchange materials often suffer from limited selectivity, poor stability, or inadequate capacity, creating substantial gaps in current treatment technologies. COFs offer unprecedented opportunities to overcome these limitations through their designable framework structures and customizable functional groups that can be precisely engineered to target specific anionic contaminants.
The historical evolution of COF technology traces back to the early 2000s when the first stable COF structures were successfully synthesized. Initial research focused primarily on gas storage and separation applications, but the field has rapidly expanded to encompass diverse applications including catalysis, sensing, and ion exchange. The transition toward anion exchange applications represents a natural progression, leveraging the inherent advantages of COF materials while addressing specific challenges associated with anionic species removal.
Current research objectives in COF-based anion exchange systems center on achieving exceptional selectivity for target anions while maintaining high exchange capacity and long-term stability. Key technical goals include developing robust synthetic methodologies for incorporating cationic functional groups into COF frameworks, optimizing pore size and geometry for enhanced anion accessibility, and establishing structure-property relationships that enable rational design of high-performance materials.
The strategic importance of advancing COF technology for anion exchange extends beyond immediate environmental applications. These materials hold potential for revolutionizing industrial processes involving anion separation, including pharmaceutical purification, nuclear waste treatment, and resource recovery from complex mixtures. The ability to design COFs with predetermined selectivity profiles represents a paradigm shift from empirical material development toward predictive, theory-guided synthesis approaches that can accelerate innovation cycles and reduce development costs.
The development of COFs for selective anion exchange has gained significant momentum due to the pressing need for efficient removal of harmful anions from contaminated water sources. Traditional anion exchange materials often suffer from limited selectivity, poor stability, or inadequate capacity, creating substantial gaps in current treatment technologies. COFs offer unprecedented opportunities to overcome these limitations through their designable framework structures and customizable functional groups that can be precisely engineered to target specific anionic contaminants.
The historical evolution of COF technology traces back to the early 2000s when the first stable COF structures were successfully synthesized. Initial research focused primarily on gas storage and separation applications, but the field has rapidly expanded to encompass diverse applications including catalysis, sensing, and ion exchange. The transition toward anion exchange applications represents a natural progression, leveraging the inherent advantages of COF materials while addressing specific challenges associated with anionic species removal.
Current research objectives in COF-based anion exchange systems center on achieving exceptional selectivity for target anions while maintaining high exchange capacity and long-term stability. Key technical goals include developing robust synthetic methodologies for incorporating cationic functional groups into COF frameworks, optimizing pore size and geometry for enhanced anion accessibility, and establishing structure-property relationships that enable rational design of high-performance materials.
The strategic importance of advancing COF technology for anion exchange extends beyond immediate environmental applications. These materials hold potential for revolutionizing industrial processes involving anion separation, including pharmaceutical purification, nuclear waste treatment, and resource recovery from complex mixtures. The ability to design COFs with predetermined selectivity profiles represents a paradigm shift from empirical material development toward predictive, theory-guided synthesis approaches that can accelerate innovation cycles and reduce development costs.
Market Demand for Selective Anion Exchange Materials
The global market for selective anion exchange materials is experiencing robust growth driven by increasing environmental regulations and the need for advanced water treatment solutions. Industrial sectors including pharmaceuticals, electronics manufacturing, and chemical processing are generating substantial demand for materials capable of selectively removing specific anionic contaminants from process streams and wastewater.
Water treatment applications represent the largest market segment, where selective anion exchange materials are essential for removing harmful anions such as perchlorate, chromate, arsenate, and various organic pollutants. Municipal water treatment facilities and industrial wastewater treatment plants require increasingly sophisticated materials to meet stringent discharge standards and drinking water quality regulations.
The pharmaceutical and biotechnology industries present a rapidly expanding market opportunity. These sectors demand highly selective materials for purification processes, where traditional ion exchange resins often lack the specificity required for complex molecular separations. COF-based materials offer potential advantages in terms of selectivity and regeneration efficiency compared to conventional polymer-based exchangers.
Nuclear waste management and remediation applications constitute a specialized but significant market niche. The ability to selectively capture radioactive anions from contaminated water sources represents a critical need, particularly in regions dealing with legacy contamination or active nuclear operations. Current solutions often suffer from limited selectivity and disposal challenges.
Environmental remediation projects worldwide are driving demand for materials capable of addressing emerging contaminants. Per- and polyfluoroalkyl substances (PFAS) removal has become a priority concern, creating market opportunities for advanced materials that can selectively capture these persistent pollutants where conventional technologies prove inadequate.
The electronics industry requires ultrapure water for semiconductor manufacturing, creating demand for selective anion exchange materials that can achieve extremely low contaminant levels. As semiconductor manufacturing processes become more sophisticated, the purity requirements continue to increase, driving the need for more selective and efficient materials.
Market growth is further supported by increasing awareness of the limitations of existing ion exchange technologies, including poor selectivity, fouling issues, and disposal challenges. These factors are creating opportunities for innovative materials that can address current technological gaps while providing improved performance characteristics.
Water treatment applications represent the largest market segment, where selective anion exchange materials are essential for removing harmful anions such as perchlorate, chromate, arsenate, and various organic pollutants. Municipal water treatment facilities and industrial wastewater treatment plants require increasingly sophisticated materials to meet stringent discharge standards and drinking water quality regulations.
The pharmaceutical and biotechnology industries present a rapidly expanding market opportunity. These sectors demand highly selective materials for purification processes, where traditional ion exchange resins often lack the specificity required for complex molecular separations. COF-based materials offer potential advantages in terms of selectivity and regeneration efficiency compared to conventional polymer-based exchangers.
Nuclear waste management and remediation applications constitute a specialized but significant market niche. The ability to selectively capture radioactive anions from contaminated water sources represents a critical need, particularly in regions dealing with legacy contamination or active nuclear operations. Current solutions often suffer from limited selectivity and disposal challenges.
Environmental remediation projects worldwide are driving demand for materials capable of addressing emerging contaminants. Per- and polyfluoroalkyl substances (PFAS) removal has become a priority concern, creating market opportunities for advanced materials that can selectively capture these persistent pollutants where conventional technologies prove inadequate.
The electronics industry requires ultrapure water for semiconductor manufacturing, creating demand for selective anion exchange materials that can achieve extremely low contaminant levels. As semiconductor manufacturing processes become more sophisticated, the purity requirements continue to increase, driving the need for more selective and efficient materials.
Market growth is further supported by increasing awareness of the limitations of existing ion exchange technologies, including poor selectivity, fouling issues, and disposal challenges. These factors are creating opportunities for innovative materials that can address current technological gaps while providing improved performance characteristics.
Current COF Development Status and Technical Challenges
Covalent Organic Frameworks (COFs) have emerged as a promising class of crystalline porous materials with significant potential for selective anion exchange applications. The current development status reveals substantial progress in synthetic methodologies, with researchers successfully creating two-dimensional and three-dimensional COF structures through various condensation reactions including boronate ester, imine, and triazine linkages. However, the field faces considerable challenges in achieving the precise control necessary for selective anion exchange functionality.
The synthesis of COFs with tailored pore environments remains a primary technical hurdle. While conventional COF synthesis has focused on creating stable, crystalline frameworks, developing materials specifically for anion selectivity requires sophisticated control over pore size, shape, and chemical functionality. Current synthetic approaches often result in frameworks with limited tunability of the internal pore environment, making it difficult to achieve the precise electrostatic and geometric complementarity needed for selective anion recognition.
Stability under operational conditions presents another significant challenge. Many existing COFs demonstrate excellent stability under ambient conditions but suffer from degradation when exposed to aqueous environments or extreme pH conditions commonly encountered in anion exchange applications. The hydrolytic stability of common linkage chemistries, particularly imine and boronate ester bonds, limits their practical application in water-based separation processes.
Characterization and understanding of anion exchange mechanisms within COF structures remain incomplete. While researchers have successfully demonstrated anion uptake in various COF systems, the fundamental understanding of how framework topology, pore chemistry, and guest-host interactions influence selectivity is still developing. Advanced characterization techniques are needed to probe the dynamic behavior of anions within the framework and elucidate structure-property relationships.
Scalability and reproducibility issues also constrain current COF development efforts. Laboratory-scale synthesis often yields materials with inconsistent properties, and scaling up production while maintaining structural integrity and performance characteristics remains challenging. The development of robust synthetic protocols that can reliably produce COFs with consistent anion exchange properties is essential for practical applications.
Despite these challenges, recent advances in reticular chemistry and post-synthetic modification strategies offer promising pathways forward. Researchers are exploring novel linkage chemistries with enhanced stability, developing hierarchical pore structures for improved mass transport, and implementing computational design approaches to predict and optimize anion selectivity. These emerging strategies represent significant opportunities for overcoming current limitations and advancing COF technology toward practical anion exchange applications.
The synthesis of COFs with tailored pore environments remains a primary technical hurdle. While conventional COF synthesis has focused on creating stable, crystalline frameworks, developing materials specifically for anion selectivity requires sophisticated control over pore size, shape, and chemical functionality. Current synthetic approaches often result in frameworks with limited tunability of the internal pore environment, making it difficult to achieve the precise electrostatic and geometric complementarity needed for selective anion recognition.
Stability under operational conditions presents another significant challenge. Many existing COFs demonstrate excellent stability under ambient conditions but suffer from degradation when exposed to aqueous environments or extreme pH conditions commonly encountered in anion exchange applications. The hydrolytic stability of common linkage chemistries, particularly imine and boronate ester bonds, limits their practical application in water-based separation processes.
Characterization and understanding of anion exchange mechanisms within COF structures remain incomplete. While researchers have successfully demonstrated anion uptake in various COF systems, the fundamental understanding of how framework topology, pore chemistry, and guest-host interactions influence selectivity is still developing. Advanced characterization techniques are needed to probe the dynamic behavior of anions within the framework and elucidate structure-property relationships.
Scalability and reproducibility issues also constrain current COF development efforts. Laboratory-scale synthesis often yields materials with inconsistent properties, and scaling up production while maintaining structural integrity and performance characteristics remains challenging. The development of robust synthetic protocols that can reliably produce COFs with consistent anion exchange properties is essential for practical applications.
Despite these challenges, recent advances in reticular chemistry and post-synthetic modification strategies offer promising pathways forward. Researchers are exploring novel linkage chemistries with enhanced stability, developing hierarchical pore structures for improved mass transport, and implementing computational design approaches to predict and optimize anion selectivity. These emerging strategies represent significant opportunities for overcoming current limitations and advancing COF technology toward practical anion exchange applications.
Existing COF Strategies for Anion Selectivity
01 Cationic COF materials for selective anion exchange
Covalent organic frameworks (COFs) with cationic functional groups or positively charged frameworks can be designed to selectively capture and exchange anionic species. These materials utilize electrostatic interactions between the positively charged framework and target anions, enabling selective separation based on charge, size, and chemical affinity. The cationic nature of these COFs allows for efficient anion exchange in aqueous solutions and various environmental applications.- Cationic COF materials for selective anion exchange: Covalent organic frameworks (COFs) with cationic functional groups or positively charged frameworks can be designed to selectively capture and exchange anionic species. These materials utilize electrostatic interactions between the positively charged framework and target anions, enabling selective separation based on charge, size, and chemical affinity. The cationic nature of the COF structure provides binding sites for various anions including halides, oxyanions, and other negatively charged species.
- Ionic COF membranes for anion separation: COF-based membranes with ionic functionalities can be utilized for selective anion transport and separation processes. These membranes combine the ordered porous structure of COFs with ion-exchange capabilities, allowing for size-selective and charge-selective anion permeation. The membrane configuration enables continuous separation processes with high selectivity and flux for target anions while rejecting other species.
- Functionalized COFs with specific anion recognition sites: COF materials can be designed with specific functional groups or binding sites that provide selective recognition and exchange of particular anions. These functionalized frameworks incorporate chemical moieties that have preferential affinity for target anions through hydrogen bonding, coordination, or other specific interactions. The selectivity can be tuned by modifying the chemical nature and spatial arrangement of the functional groups within the COF structure.
- COF-based adsorbents for environmental anion removal: COF materials can serve as efficient adsorbents for removing harmful or unwanted anions from aqueous solutions and environmental matrices. These materials demonstrate high adsorption capacity and selectivity for pollutant anions such as chromate, arsenate, phosphate, and other contaminants. The porous structure and tunable surface chemistry of COFs enable effective capture and exchange of target anions for water treatment and environmental remediation applications.
- Composite COF materials for enhanced anion exchange performance: Composite materials combining COFs with other functional components can provide enhanced anion exchange properties. These composites may incorporate COFs with polymers, inorganic materials, or other porous frameworks to improve mechanical stability, processability, and exchange kinetics. The composite approach allows for synergistic effects that enhance selectivity, capacity, and regeneration performance compared to pure COF materials.
02 Functionalized COFs with specific binding sites for anion selectivity
COF structures can be modified with specific functional groups or binding sites that provide selective recognition and exchange of particular anions. These functionalized frameworks incorporate chemical moieties that have preferential affinity for target anions through hydrogen bonding, coordination, or other specific interactions. This approach enables highly selective anion exchange for applications in water purification, catalysis, and separation processes.Expand Specific Solutions03 Porous COF architectures optimized for anion transport and exchange kinetics
The design of COF pore structures, including pore size, shape, and connectivity, can be tailored to optimize anion diffusion and exchange rates. These architectures feature controlled porosity that facilitates rapid anion transport while maintaining selectivity. The ordered porous structure of COFs provides advantages over traditional ion exchange materials by offering uniform channels and predictable exchange behavior.Expand Specific Solutions04 Composite materials combining COFs with other ion exchange components
Hybrid materials that integrate COFs with other ion exchange materials, such as polymers, inorganic matrices, or layered materials, can enhance anion exchange performance. These composites leverage the structural advantages of COFs while incorporating complementary properties from other materials to improve mechanical stability, processability, and exchange capacity. The synergistic effects in these composites result in enhanced selectivity and efficiency for anion exchange applications.Expand Specific Solutions05 COF-based anion exchange membranes and devices
COF materials can be fabricated into membranes, films, or structured devices for practical anion exchange applications. These implementations enable continuous separation processes, electrochemical applications, and water treatment systems. The integration of COFs into membrane formats combines their selective anion exchange properties with scalable processing methods, making them suitable for industrial applications in desalination, resource recovery, and environmental remediation.Expand Specific Solutions
Leading Players in COF Research and Commercialization
The development of COFs for selective anion exchange represents an emerging field within the broader porous materials landscape, currently in its early-to-mid development stage with significant growth potential. The market remains relatively nascent but shows promising expansion driven by applications in water treatment, energy storage, and separation technologies. From a technology maturity perspective, the field demonstrates strong academic foundation with leading research institutions driving innovation. Key players include prominent Chinese universities such as Nanchang University, Huazhong University of Science & Technology, Tianjin University, and Fudan University, alongside international institutions like Monash University and the University of California system. Industrial participation is emerging through companies like BASF Corp. and specialized technology firms, while government research organizations such as CSIR provide foundational support. The competitive landscape shows a concentration of expertise in academic institutions, particularly in China, indicating the technology is still in the research-intensive phase with limited commercial deployment but strong potential for future industrial applications.
Fudan University
Technical Solution: Advanced COF design strategies focusing on heteroatom incorporation and defect engineering for enhanced anion exchange performance. Their approach involves systematic modification of COF backbones with nitrogen-rich heterocycles and the introduction of controlled defects to create additional binding sites for anions. The research emphasizes understanding structure-property relationships through computational modeling combined with experimental validation. Their COF materials show remarkable selectivity for perchlorate and nitrate ions, with demonstrated applications in environmental remediation and analytical separation processes.
Strengths: Excellent theoretical foundation and computational modeling capabilities for COF design. Weaknesses: Focus primarily on academic research with limited commercial development experience.
Nankai University
Technical Solution: Pioneered the development of ionic COFs through direct synthesis approaches incorporating charged building blocks for anion exchange applications. Their research focuses on creating COFs with permanent positive charges through the use of viologen-based linkers and pyridinium-containing nodes. The synthesis methodology involves solvothermal conditions with careful control of reaction parameters to achieve high crystallinity and porosity. Their COF materials demonstrate exceptional selectivity for chromate and arsenate removal from aqueous solutions, with exchange capacities reaching up to 2.5 mmol/g and maintaining stability across pH ranges from 2 to 12.
Strengths: Strong fundamental research capabilities and innovative synthesis approaches for ionic COFs. Weaknesses: Limited industrial partnerships and scaling capabilities compared to commercial entities.
Key Innovations in COF Pore Engineering for Anion Binding
Preparation of a ferric acetate coated silica sol by selective anion exchange
PatentInactiveUS4478742A
Innovation
- An anion exchange method is employed using ion-free colloidal silica and an inorganic iron salt in contact with a strong base anion exchange resin in the acetic acid salt form to convert the iron salt into iron acetate and coat it onto silica sols, leveraging commercially available starting materials like NALCOAG 1034-A silica sol and ferric salts, and regenerating the resin with acetic acid to produce iron acetate coated silica sols.
Ion exchange and electrochemical methods and devices employing one-step quaternized and polymerized anion selective polymers
PatentInactiveUS7087654B2
Innovation
- The development of caustic-stable anion selective polymers through substantially simultaneous quaternization and polymerization reactions, using ethylenic tertiary amine monomers, alkylating agents with high boiling points, and cross-linking agents in the presence of polymerization agents, eliminating the need for caustic pretreatment and washing steps, and resulting in polymers that are insoluble in aqueous solutions.
Environmental Regulations for Ion Exchange Materials
The regulatory landscape for ion exchange materials, particularly covalent organic frameworks (COFs) designed for selective anion exchange, is rapidly evolving as environmental agencies worldwide recognize both the potential benefits and risks associated with these advanced materials. Current environmental regulations primarily focus on traditional ion exchange resins and polymeric materials, creating a regulatory gap that COF developers must navigate carefully.
In the United States, the Environmental Protection Agency (EPA) regulates ion exchange materials under multiple frameworks, including the Toxic Substances Control Act (TSCA) and the Safe Drinking Water Act (SDWA). COFs intended for water treatment applications must demonstrate compliance with NSF/ANSI Standard 61 for drinking water system components, which requires extensive toxicological testing to ensure no harmful substances leach into treated water. The EPA's New Chemicals Program under TSCA mandates pre-manufacture notification for novel COF structures, requiring comprehensive environmental fate and transport studies.
European regulations under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) impose stringent requirements on COF manufacturers. The regulation demands detailed safety data sheets, environmental risk assessments, and long-term ecological impact studies. Particularly relevant for anion-selective COFs is the Biocidal Products Regulation (BPR), which governs materials that may exhibit antimicrobial properties during anion capture processes.
Emerging regulatory trends indicate increasing scrutiny of nanoscale materials and their environmental persistence. The OECD Guidelines for Testing of Chemicals now include specific protocols for nanomaterials that may apply to certain COF structures. Environmental agencies are developing new testing methodologies to assess the biodegradability and bioaccumulation potential of synthetic organic frameworks.
Compliance challenges for COF developers include demonstrating controlled release profiles, establishing acceptable daily intake levels for potential leachates, and conducting comprehensive life-cycle assessments. Future regulatory developments are expected to address end-of-life disposal requirements, recycling protocols, and standardized performance metrics for selective anion exchange applications in environmental remediation contexts.
In the United States, the Environmental Protection Agency (EPA) regulates ion exchange materials under multiple frameworks, including the Toxic Substances Control Act (TSCA) and the Safe Drinking Water Act (SDWA). COFs intended for water treatment applications must demonstrate compliance with NSF/ANSI Standard 61 for drinking water system components, which requires extensive toxicological testing to ensure no harmful substances leach into treated water. The EPA's New Chemicals Program under TSCA mandates pre-manufacture notification for novel COF structures, requiring comprehensive environmental fate and transport studies.
European regulations under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) impose stringent requirements on COF manufacturers. The regulation demands detailed safety data sheets, environmental risk assessments, and long-term ecological impact studies. Particularly relevant for anion-selective COFs is the Biocidal Products Regulation (BPR), which governs materials that may exhibit antimicrobial properties during anion capture processes.
Emerging regulatory trends indicate increasing scrutiny of nanoscale materials and their environmental persistence. The OECD Guidelines for Testing of Chemicals now include specific protocols for nanomaterials that may apply to certain COF structures. Environmental agencies are developing new testing methodologies to assess the biodegradability and bioaccumulation potential of synthetic organic frameworks.
Compliance challenges for COF developers include demonstrating controlled release profiles, establishing acceptable daily intake levels for potential leachates, and conducting comprehensive life-cycle assessments. Future regulatory developments are expected to address end-of-life disposal requirements, recycling protocols, and standardized performance metrics for selective anion exchange applications in environmental remediation contexts.
Scale-up Challenges for COF Manufacturing
The transition from laboratory-scale synthesis to industrial-scale production of COFs for selective anion exchange applications presents numerous technical and economic challenges that significantly impact commercial viability. Current COF synthesis methods, predominantly relying on solvothermal reactions and mechanochemical approaches, face substantial scalability limitations due to their inherently batch-oriented nature and stringent reaction conditions.
Process uniformity emerges as a critical bottleneck during scale-up operations. Laboratory synthesis typically involves small reaction volumes with precise temperature and pressure control, enabling homogeneous nucleation and crystal growth. However, maintaining uniform heat and mass transfer across large-scale reactors becomes increasingly difficult, leading to heterogeneous product quality and reduced crystallinity. The challenge intensifies when considering the sensitive nature of COF formation, where slight variations in local reaction conditions can dramatically alter pore structure and anion selectivity properties.
Solvent management represents another significant hurdle in COF manufacturing scale-up. Most COF synthesis protocols require substantial quantities of organic solvents, including DMF, dioxane, and mesitylene, which pose environmental and economic concerns at industrial scales. The recovery and purification of these solvents add complexity to the manufacturing process, while solvent residues can compromise the anion exchange performance of the final product.
Equipment design limitations further complicate scale-up efforts. Traditional laboratory glassware cannot accommodate the mechanical stresses and thermal gradients encountered in large-scale production. Specialized reactor designs incorporating advanced mixing systems, precise temperature control zones, and continuous monitoring capabilities are essential but require substantial capital investment and technical expertise.
Quality control and characterization present additional challenges as production volumes increase. Standard analytical techniques used for COF characterization, such as powder X-ray diffraction and nitrogen adsorption isotherms, become time-intensive and costly when applied to large batches. Developing rapid, inline quality assessment methods remains crucial for ensuring consistent anion exchange selectivity across production runs.
Economic considerations significantly influence scale-up feasibility. The high cost of organic building blocks, specialized equipment requirements, and energy-intensive synthesis conditions contribute to elevated production costs. Achieving cost-effective manufacturing while maintaining the precise structural control necessary for selective anion exchange functionality requires innovative process optimization and potentially alternative synthesis approaches specifically designed for industrial implementation.
Process uniformity emerges as a critical bottleneck during scale-up operations. Laboratory synthesis typically involves small reaction volumes with precise temperature and pressure control, enabling homogeneous nucleation and crystal growth. However, maintaining uniform heat and mass transfer across large-scale reactors becomes increasingly difficult, leading to heterogeneous product quality and reduced crystallinity. The challenge intensifies when considering the sensitive nature of COF formation, where slight variations in local reaction conditions can dramatically alter pore structure and anion selectivity properties.
Solvent management represents another significant hurdle in COF manufacturing scale-up. Most COF synthesis protocols require substantial quantities of organic solvents, including DMF, dioxane, and mesitylene, which pose environmental and economic concerns at industrial scales. The recovery and purification of these solvents add complexity to the manufacturing process, while solvent residues can compromise the anion exchange performance of the final product.
Equipment design limitations further complicate scale-up efforts. Traditional laboratory glassware cannot accommodate the mechanical stresses and thermal gradients encountered in large-scale production. Specialized reactor designs incorporating advanced mixing systems, precise temperature control zones, and continuous monitoring capabilities are essential but require substantial capital investment and technical expertise.
Quality control and characterization present additional challenges as production volumes increase. Standard analytical techniques used for COF characterization, such as powder X-ray diffraction and nitrogen adsorption isotherms, become time-intensive and costly when applied to large batches. Developing rapid, inline quality assessment methods remains crucial for ensuring consistent anion exchange selectivity across production runs.
Economic considerations significantly influence scale-up feasibility. The high cost of organic building blocks, specialized equipment requirements, and energy-intensive synthesis conditions contribute to elevated production costs. Achieving cost-effective manufacturing while maintaining the precise structural control necessary for selective anion exchange functionality requires innovative process optimization and potentially alternative synthesis approaches specifically designed for industrial implementation.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!