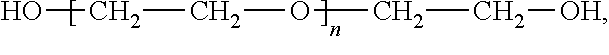Optimize Polyethylene Glycol Splitting for Fractionation
MAR 8, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEG Fractionation Technology Background and Objectives
Polyethylene glycol (PEG) fractionation represents a critical separation technology that has evolved significantly since its initial development in the mid-20th century. Originally discovered as a byproduct of ethylene oxide polymerization, PEG quickly gained recognition for its unique physicochemical properties, including biocompatibility, water solubility, and tunable molecular weight characteristics. The evolution from crude PEG production to sophisticated fractionation techniques reflects the growing demand for high-purity, narrow molecular weight distribution polymers across diverse industrial applications.
The historical development of PEG fractionation technology can be traced through several distinct phases. Early separation methods relied primarily on precipitation and crystallization techniques, which provided limited control over molecular weight distribution. The introduction of chromatographic separation methods in the 1970s marked a significant advancement, enabling more precise molecular weight control. Subsequent developments in membrane technology, supercritical fluid extraction, and advanced distillation techniques have progressively enhanced separation efficiency and product quality.
Current fractionation challenges stem from the inherent polydispersity of PEG polymers and the complex thermodynamic behavior exhibited during separation processes. Traditional splitting methods often result in broad molecular weight distributions, incomplete separation of adjacent molecular weight fractions, and significant energy consumption. These limitations become particularly pronounced when targeting specific molecular weight ranges for pharmaceutical, cosmetic, or specialty chemical applications where narrow polydispersity indices are essential.
The primary technical objectives driving PEG fractionation optimization focus on achieving superior molecular weight precision while maintaining economic viability. Key targets include reducing polydispersity indices below 1.1 for critical applications, improving separation efficiency to minimize product loss, and developing energy-efficient processes that reduce operational costs. Additionally, there is growing emphasis on developing environmentally sustainable fractionation methods that minimize solvent usage and waste generation.
Contemporary research directions emphasize the integration of advanced process control systems, novel separation media, and hybrid fractionation approaches. The convergence of artificial intelligence with traditional separation science offers unprecedented opportunities for real-time optimization and predictive process control. These technological advances aim to establish PEG fractionation as a highly efficient, scalable, and environmentally responsible manufacturing process capable of meeting increasingly stringent quality requirements across multiple industrial sectors.
The historical development of PEG fractionation technology can be traced through several distinct phases. Early separation methods relied primarily on precipitation and crystallization techniques, which provided limited control over molecular weight distribution. The introduction of chromatographic separation methods in the 1970s marked a significant advancement, enabling more precise molecular weight control. Subsequent developments in membrane technology, supercritical fluid extraction, and advanced distillation techniques have progressively enhanced separation efficiency and product quality.
Current fractionation challenges stem from the inherent polydispersity of PEG polymers and the complex thermodynamic behavior exhibited during separation processes. Traditional splitting methods often result in broad molecular weight distributions, incomplete separation of adjacent molecular weight fractions, and significant energy consumption. These limitations become particularly pronounced when targeting specific molecular weight ranges for pharmaceutical, cosmetic, or specialty chemical applications where narrow polydispersity indices are essential.
The primary technical objectives driving PEG fractionation optimization focus on achieving superior molecular weight precision while maintaining economic viability. Key targets include reducing polydispersity indices below 1.1 for critical applications, improving separation efficiency to minimize product loss, and developing energy-efficient processes that reduce operational costs. Additionally, there is growing emphasis on developing environmentally sustainable fractionation methods that minimize solvent usage and waste generation.
Contemporary research directions emphasize the integration of advanced process control systems, novel separation media, and hybrid fractionation approaches. The convergence of artificial intelligence with traditional separation science offers unprecedented opportunities for real-time optimization and predictive process control. These technological advances aim to establish PEG fractionation as a highly efficient, scalable, and environmentally responsible manufacturing process capable of meeting increasingly stringent quality requirements across multiple industrial sectors.
Market Demand Analysis for High-Purity PEG Products
The global polyethylene glycol market demonstrates robust growth driven by increasing demand for high-purity PEG products across multiple industrial sectors. Pharmaceutical applications represent the largest consumption segment, where high-purity PEG serves as excipients, drug delivery systems, and bioconjugation agents. The stringent regulatory requirements in pharmaceutical manufacturing necessitate PEG products with exceptional purity levels, typically exceeding 99.5%, creating substantial market opportunities for optimized fractionation technologies.
Cosmetics and personal care industries constitute another significant demand driver, utilizing high-purity PEG in premium skincare formulations, hair care products, and color cosmetics. Consumer preferences for clean-label and high-quality personal care products have intensified the requirement for ultra-pure PEG variants, particularly those with narrow molecular weight distributions achieved through advanced splitting and fractionation processes.
The electronics and semiconductor sectors present emerging high-value applications for specialized PEG products. High-purity PEG serves as processing aids in semiconductor manufacturing, where even trace impurities can compromise product performance. This application demands extremely pure PEG with controlled molecular weight specifications, driving premium pricing and creating lucrative market niches.
Industrial applications including lubricants, surfactants, and polymer processing continue expanding, with increasing emphasis on product consistency and purity. Manufacturing processes requiring precise performance characteristics favor high-purity PEG over conventional grades, supporting market premiumization trends.
Geographically, North America and Europe maintain strong demand for high-purity PEG products, driven by established pharmaceutical and cosmetics industries with stringent quality standards. Asia-Pacific markets show accelerating growth, particularly in China and India, where expanding pharmaceutical manufacturing and rising consumer spending on premium personal care products fuel demand.
Market dynamics indicate growing price differentiation between standard and high-purity PEG grades, with premium products commanding significantly higher margins. This trend incentivizes investment in advanced fractionation technologies capable of producing consistently high-purity products with optimized molecular weight distributions, validating the commercial importance of polyethylene glycol splitting optimization initiatives.
Cosmetics and personal care industries constitute another significant demand driver, utilizing high-purity PEG in premium skincare formulations, hair care products, and color cosmetics. Consumer preferences for clean-label and high-quality personal care products have intensified the requirement for ultra-pure PEG variants, particularly those with narrow molecular weight distributions achieved through advanced splitting and fractionation processes.
The electronics and semiconductor sectors present emerging high-value applications for specialized PEG products. High-purity PEG serves as processing aids in semiconductor manufacturing, where even trace impurities can compromise product performance. This application demands extremely pure PEG with controlled molecular weight specifications, driving premium pricing and creating lucrative market niches.
Industrial applications including lubricants, surfactants, and polymer processing continue expanding, with increasing emphasis on product consistency and purity. Manufacturing processes requiring precise performance characteristics favor high-purity PEG over conventional grades, supporting market premiumization trends.
Geographically, North America and Europe maintain strong demand for high-purity PEG products, driven by established pharmaceutical and cosmetics industries with stringent quality standards. Asia-Pacific markets show accelerating growth, particularly in China and India, where expanding pharmaceutical manufacturing and rising consumer spending on premium personal care products fuel demand.
Market dynamics indicate growing price differentiation between standard and high-purity PEG grades, with premium products commanding significantly higher margins. This trend incentivizes investment in advanced fractionation technologies capable of producing consistently high-purity products with optimized molecular weight distributions, validating the commercial importance of polyethylene glycol splitting optimization initiatives.
Current PEG Splitting Challenges and Technical Barriers
Polyethylene glycol splitting for fractionation faces significant molecular weight distribution challenges that fundamentally impact separation efficiency. The inherent polydispersity of PEG molecules creates overlapping molecular weight ranges, making precise fractionation extremely difficult. Current splitting methodologies struggle to achieve sharp molecular weight cutoffs, resulting in broad transition zones between fractions that compromise product purity and yield.
Thermal degradation represents a critical barrier during PEG splitting processes, particularly when elevated temperatures are employed to enhance separation kinetics. The ether linkages in PEG chains are susceptible to oxidative and thermal breakdown, leading to chain scission and the formation of lower molecular weight fragments. This degradation not only reduces overall yield but also introduces unwanted impurities that contaminate target fractions.
Solvent selection and compatibility issues pose substantial technical hurdles in PEG fractionation systems. Many conventional solvents exhibit limited selectivity for specific PEG molecular weight ranges, while others may cause precipitation or phase separation at critical processing conditions. The challenge intensifies when considering solvent recovery and recycling, as residual solvent traces can significantly impact downstream applications.
Viscosity-related processing difficulties emerge as molecular weight increases, creating operational challenges in conventional separation equipment. High molecular weight PEG fractions exhibit non-Newtonian flow behavior that complicates pumping, mixing, and heat transfer operations. These rheological properties often necessitate elevated processing temperatures, which paradoxically increase the risk of thermal degradation.
Crystallization and solidification phenomena during cooling stages present additional technical barriers, particularly for higher molecular weight fractions. Uncontrolled crystallization can lead to equipment fouling, inconsistent product morphology, and difficulties in downstream handling. The crystallization kinetics are highly sensitive to cooling rates, concentration gradients, and the presence of impurities.
Analytical characterization limitations hinder process optimization and quality control efforts. Traditional molecular weight determination methods often lack the resolution required to accurately assess fractionation efficiency, particularly in the transition regions between adjacent fractions. This analytical gap makes it challenging to optimize process parameters and validate separation performance.
Scale-up considerations introduce additional complexity, as laboratory-scale fractionation methods often fail to translate effectively to industrial production volumes. Heat and mass transfer limitations become more pronounced at larger scales, while maintaining uniform processing conditions across larger equipment volumes presents significant engineering challenges.
Thermal degradation represents a critical barrier during PEG splitting processes, particularly when elevated temperatures are employed to enhance separation kinetics. The ether linkages in PEG chains are susceptible to oxidative and thermal breakdown, leading to chain scission and the formation of lower molecular weight fragments. This degradation not only reduces overall yield but also introduces unwanted impurities that contaminate target fractions.
Solvent selection and compatibility issues pose substantial technical hurdles in PEG fractionation systems. Many conventional solvents exhibit limited selectivity for specific PEG molecular weight ranges, while others may cause precipitation or phase separation at critical processing conditions. The challenge intensifies when considering solvent recovery and recycling, as residual solvent traces can significantly impact downstream applications.
Viscosity-related processing difficulties emerge as molecular weight increases, creating operational challenges in conventional separation equipment. High molecular weight PEG fractions exhibit non-Newtonian flow behavior that complicates pumping, mixing, and heat transfer operations. These rheological properties often necessitate elevated processing temperatures, which paradoxically increase the risk of thermal degradation.
Crystallization and solidification phenomena during cooling stages present additional technical barriers, particularly for higher molecular weight fractions. Uncontrolled crystallization can lead to equipment fouling, inconsistent product morphology, and difficulties in downstream handling. The crystallization kinetics are highly sensitive to cooling rates, concentration gradients, and the presence of impurities.
Analytical characterization limitations hinder process optimization and quality control efforts. Traditional molecular weight determination methods often lack the resolution required to accurately assess fractionation efficiency, particularly in the transition regions between adjacent fractions. This analytical gap makes it challenging to optimize process parameters and validate separation performance.
Scale-up considerations introduce additional complexity, as laboratory-scale fractionation methods often fail to translate effectively to industrial production volumes. Heat and mass transfer limitations become more pronounced at larger scales, while maintaining uniform processing conditions across larger equipment volumes presents significant engineering challenges.
Existing PEG Splitting and Purification Solutions
01 Catalytic splitting of polyethylene glycol using metal catalysts
Metal catalysts such as platinum, palladium, or nickel-based catalysts can be employed to facilitate the splitting or degradation of polyethylene glycol chains. These catalysts promote selective bond cleavage under controlled temperature and pressure conditions, enabling efficient conversion to lower molecular weight products or monomers. The catalytic approach offers advantages in terms of reaction selectivity and energy efficiency compared to thermal methods.- Catalytic splitting methods for polyethylene glycol: Various catalytic approaches can be employed to optimize the splitting of polyethylene glycol. These methods involve the use of specific catalysts that facilitate the breaking of ether bonds in the polymer chain. The catalytic process can be controlled by adjusting reaction parameters such as temperature, pressure, and catalyst concentration to achieve desired molecular weight distributions and conversion rates. Different types of catalysts including acidic, basic, and metal-based catalysts have been investigated for their effectiveness in PEG degradation.
- Thermal degradation optimization of polyethylene glycol: Thermal splitting involves the controlled heating of polyethylene glycol under specific conditions to break down the polymer chains. Optimization of this process includes determining optimal temperature ranges, heating rates, and residence times to maximize yield and minimize unwanted byproducts. The thermal degradation process can be enhanced through the use of inert atmospheres or vacuum conditions to prevent oxidation and improve product quality. Process parameters can be adjusted to target specific molecular weight fractions.
- Enzymatic degradation of polyethylene glycol: Enzymatic methods offer a biological approach to splitting polyethylene glycol chains. Specific enzymes can selectively cleave the polymer at particular sites, providing controlled degradation. This method is particularly advantageous for producing specific oligomers or monomers with high selectivity. The enzymatic process can be optimized by adjusting factors such as enzyme concentration, pH, temperature, and reaction time to achieve maximum efficiency and desired product distribution.
- Chemical oxidation methods for PEG splitting: Chemical oxidation provides an effective route for breaking down polyethylene glycol through the use of oxidizing agents. Various oxidants can be employed to selectively attack and cleave the ether linkages in the polymer backbone. The optimization involves selecting appropriate oxidizing agents, controlling reaction conditions, and managing the oxidation rate to prevent over-oxidation. This approach can be tailored to produce specific degradation products with desired functional groups.
- Hydrolysis-based splitting techniques: Hydrolytic splitting of polyethylene glycol involves the cleavage of ether bonds through reaction with water under controlled conditions. This process can be catalyzed by acids or bases to enhance the reaction rate. Optimization parameters include pH control, temperature management, water-to-polymer ratio, and reaction time. The hydrolysis method can be designed to achieve specific degrees of depolymerization and produce oligomers with targeted molecular weights. This approach is particularly useful for recycling and recovery applications.
02 Oxidative degradation methods for polyethylene glycol splitting
Oxidative degradation techniques utilize oxidizing agents or oxidative conditions to break down polyethylene glycol molecules. This approach involves the use of chemical oxidants, photocatalytic oxidation, or electrochemical oxidation to cleave the ether bonds in the polymer chain. The method can be optimized by controlling parameters such as oxidant concentration, pH, temperature, and reaction time to achieve desired molecular weight reduction and product distribution.Expand Specific Solutions03 Enzymatic splitting of polyethylene glycol
Enzymatic methods employ specific enzymes capable of degrading polyethylene glycol through biological catalysis. These biocatalysts offer high specificity and operate under mild conditions, making them environmentally friendly alternatives. The enzymatic splitting process can be optimized by selecting appropriate enzyme types, adjusting enzyme concentration, controlling reaction temperature and pH, and using co-factors or additives to enhance enzyme activity and stability.Expand Specific Solutions04 Thermal cracking optimization for polyethylene glycol
Thermal cracking involves heating polyethylene glycol to elevated temperatures to induce chain scission through pyrolysis. Optimization of this process includes controlling heating rates, residence time, reactor design, and the use of inert atmospheres to minimize unwanted side reactions. Advanced thermal methods may incorporate microwave heating or plasma technology to improve energy efficiency and product selectivity while reducing reaction time.Expand Specific Solutions05 Hydrolytic splitting under acidic or alkaline conditions
Hydrolytic splitting utilizes acidic or alkaline aqueous solutions to hydrolyze the ether linkages in polyethylene glycol. The process can be optimized by adjusting the concentration of acid or base, reaction temperature, pressure, and residence time. Supercritical water conditions may also be employed to enhance the hydrolysis rate. This method is particularly effective for producing specific oligomers or converting polyethylene glycol to ethylene glycol and other valuable chemicals.Expand Specific Solutions
Major Players in PEG Processing and Chemical Industry
The polyethylene glycol (PEG) splitting and fractionation technology sector represents a mature yet evolving market within the broader petrochemical industry. The competitive landscape is dominated by established petrochemical giants including China Petroleum & Chemical Corp. (Sinopec), ExxonMobil Chemical Patents, Chevron Phillips Chemical, and NOVA Chemicals, alongside specialized chemical companies like NOF Corp. and Sanyo Chemical Industries. The market demonstrates significant scale with multi-billion dollar revenues across major players, driven by diverse applications spanning pharmaceuticals, cosmetics, and industrial processes. Technology maturity varies considerably, with traditional petrochemical companies leveraging established refining capabilities while specialized firms like NOF Corp. and research institutions such as Korea Research Institute of Chemical Technology focus on advanced fractionation techniques and high-purity PEG derivatives, indicating ongoing innovation opportunities in optimization processes.
China Petroleum & Chemical Corp.
Technical Solution: SINOPEC has developed advanced distillation technologies for polyethylene glycol fractionation, utilizing multi-stage vacuum distillation systems with optimized temperature and pressure profiles. Their approach incorporates molecular distillation techniques combined with reactive distillation processes to achieve high-purity PEG separation across different molecular weight ranges. The company employs proprietary catalyst systems and heat integration strategies to minimize energy consumption during the splitting process, achieving separation efficiencies above 95% for various PEG grades while maintaining product quality standards for industrial applications.
Strengths: Extensive industrial experience and integrated petrochemical operations. Weaknesses: High energy consumption in traditional distillation processes.
Dow Global Technologies LLC
Technical Solution: Dow has pioneered membrane-based separation technologies for PEG fractionation, utilizing selective permeation membranes that separate PEG molecules based on molecular weight differences. Their process combines ultrafiltration and nanofiltration techniques with optimized membrane materials to achieve precise molecular weight cutoffs. The technology incorporates continuous processing capabilities with automated control systems that monitor viscosity, molecular weight distribution, and purity levels in real-time. This approach reduces energy requirements by up to 40% compared to conventional distillation methods while maintaining high product quality and enabling flexible production of various PEG grades.
Strengths: Energy-efficient membrane technology and advanced process control. Weaknesses: High membrane replacement costs and limited scalability for very high molecular weight PEGs.
Core Patents in Advanced PEG Fractionation Methods
Process for preparing and separating monodisperse polyethylene glycol
PatentInactiveUS20160075624A1
Innovation
- A process involving the modification of industrial-grade polyethylene glycol using a compound of general formula R-B, followed by silica gel column chromatography to separate derivatives based on polarity differences, and subsequent hydrolysis to achieve monodisperse polyethylene glycol with a narrow molecular weight distribution.
Ultra high molecular weight polyethylene fractions having narrow molecular weight distributions and methods of making and using the same
PatentInactiveEP1709088A1
Innovation
- A method involving the dissolution of UHMWPE in a solvent, followed by fractionation using a column with a support, where the temperature is controlled to precipitate fractions with narrow molecular weight distributions, allowing for the recovery of polyethylene fractions with a polydispersity index (PDI) less than 2.3 and weight-average molecular weight greater than 1,000,000 g/mol, using a solvent gradient to isolate specific fractions.
Environmental Regulations for Chemical Processing
The optimization of polyethylene glycol splitting for fractionation operates within a complex regulatory framework that governs chemical processing operations worldwide. Environmental regulations have become increasingly stringent as governments recognize the potential impacts of chemical manufacturing on air quality, water resources, and soil contamination. These regulations directly influence the design, operation, and optimization strategies employed in PEG fractionation processes.
Air emission standards represent a critical regulatory consideration for PEG splitting operations. The process typically involves thermal treatment and potential volatile organic compound emissions, which are subject to strict monitoring under regulations such as the Clean Air Act in the United States and similar legislation globally. Facilities must implement appropriate emission control technologies and maintain compliance with permitted discharge limits, often requiring continuous monitoring systems and regular reporting to regulatory authorities.
Water discharge regulations significantly impact PEG fractionation processes, particularly regarding wastewater treatment and disposal. The splitting process may generate aqueous waste streams containing residual PEG compounds, catalysts, or other chemical additives. Regulatory frameworks like the Clean Water Act establish stringent limits on biochemical oxygen demand, chemical oxygen demand, and specific pollutant concentrations in industrial discharge. These requirements often necessitate advanced wastewater treatment systems, increasing both capital and operational costs.
Waste management regulations govern the handling and disposal of solid waste generated during PEG splitting operations. Regulatory compliance requires proper classification of waste materials, implementation of waste minimization strategies, and adherence to hazardous waste disposal protocols where applicable. The Resource Conservation and Recovery Act and international equivalents establish comprehensive frameworks for waste tracking, storage, and ultimate disposal methods.
Chemical safety regulations, including those established by OSHA and international safety standards, mandate specific operational protocols for PEG processing facilities. These regulations encompass worker safety requirements, emergency response procedures, and chemical inventory reporting obligations. Compliance often requires extensive documentation, regular safety training programs, and implementation of process safety management systems that can influence optimization strategies and operational flexibility.
Air emission standards represent a critical regulatory consideration for PEG splitting operations. The process typically involves thermal treatment and potential volatile organic compound emissions, which are subject to strict monitoring under regulations such as the Clean Air Act in the United States and similar legislation globally. Facilities must implement appropriate emission control technologies and maintain compliance with permitted discharge limits, often requiring continuous monitoring systems and regular reporting to regulatory authorities.
Water discharge regulations significantly impact PEG fractionation processes, particularly regarding wastewater treatment and disposal. The splitting process may generate aqueous waste streams containing residual PEG compounds, catalysts, or other chemical additives. Regulatory frameworks like the Clean Water Act establish stringent limits on biochemical oxygen demand, chemical oxygen demand, and specific pollutant concentrations in industrial discharge. These requirements often necessitate advanced wastewater treatment systems, increasing both capital and operational costs.
Waste management regulations govern the handling and disposal of solid waste generated during PEG splitting operations. Regulatory compliance requires proper classification of waste materials, implementation of waste minimization strategies, and adherence to hazardous waste disposal protocols where applicable. The Resource Conservation and Recovery Act and international equivalents establish comprehensive frameworks for waste tracking, storage, and ultimate disposal methods.
Chemical safety regulations, including those established by OSHA and international safety standards, mandate specific operational protocols for PEG processing facilities. These regulations encompass worker safety requirements, emergency response procedures, and chemical inventory reporting obligations. Compliance often requires extensive documentation, regular safety training programs, and implementation of process safety management systems that can influence optimization strategies and operational flexibility.
Process Safety Standards for PEG Manufacturing
Process safety standards for polyethylene glycol manufacturing represent a critical framework governing the safe operation of PEG production facilities, particularly during splitting and fractionation processes. These standards encompass comprehensive guidelines that address hazard identification, risk assessment, and mitigation strategies specific to the thermal and chemical processes involved in PEG molecular weight separation.
The foundation of PEG manufacturing safety standards rests on established international frameworks including OSHA Process Safety Management regulations, API recommended practices, and IEC functional safety standards. These frameworks mandate systematic approaches to process hazard analysis, with particular emphasis on thermal runaway scenarios that can occur during high-temperature splitting operations where PEG chains are selectively broken to achieve desired molecular weight distributions.
Temperature control represents the most critical safety parameter in PEG fractionation processes. Safety standards require implementation of multiple independent temperature monitoring systems with fail-safe mechanisms to prevent overheating beyond 280°C, where significant thermal degradation and potential explosive decomposition can occur. Emergency cooling systems must be designed with sufficient capacity to handle worst-case thermal excursions during splitting operations.
Pressure management protocols constitute another essential component of safety standards, addressing the vapor pressure dynamics during fractionation column operations. Standards mandate pressure relief systems sized for emergency venting scenarios, with particular attention to the handling of hot PEG vapors that can cause severe thermal burns upon contact with personnel or equipment.
Chemical exposure prevention measures focus on minimizing worker contact with hot PEG materials and potential degradation byproducts formed during splitting processes. Ventilation systems must meet stringent air change requirements to prevent accumulation of organic vapors, while personal protective equipment specifications address the unique thermal and chemical hazards associated with molten polymer handling.
Emergency response procedures specific to PEG manufacturing incidents require specialized training protocols covering thermal burn treatment, chemical spill containment, and fire suppression techniques appropriate for high-molecular-weight organic materials. These procedures must account for the unique flow characteristics of molten PEG and its tendency to adhere to surfaces at elevated temperatures.
The foundation of PEG manufacturing safety standards rests on established international frameworks including OSHA Process Safety Management regulations, API recommended practices, and IEC functional safety standards. These frameworks mandate systematic approaches to process hazard analysis, with particular emphasis on thermal runaway scenarios that can occur during high-temperature splitting operations where PEG chains are selectively broken to achieve desired molecular weight distributions.
Temperature control represents the most critical safety parameter in PEG fractionation processes. Safety standards require implementation of multiple independent temperature monitoring systems with fail-safe mechanisms to prevent overheating beyond 280°C, where significant thermal degradation and potential explosive decomposition can occur. Emergency cooling systems must be designed with sufficient capacity to handle worst-case thermal excursions during splitting operations.
Pressure management protocols constitute another essential component of safety standards, addressing the vapor pressure dynamics during fractionation column operations. Standards mandate pressure relief systems sized for emergency venting scenarios, with particular attention to the handling of hot PEG vapors that can cause severe thermal burns upon contact with personnel or equipment.
Chemical exposure prevention measures focus on minimizing worker contact with hot PEG materials and potential degradation byproducts formed during splitting processes. Ventilation systems must meet stringent air change requirements to prevent accumulation of organic vapors, while personal protective equipment specifications address the unique thermal and chemical hazards associated with molten polymer handling.
Emergency response procedures specific to PEG manufacturing incidents require specialized training protocols covering thermal burn treatment, chemical spill containment, and fire suppression techniques appropriate for high-molecular-weight organic materials. These procedures must account for the unique flow characteristics of molten PEG and its tendency to adhere to surfaces at elevated temperatures.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!