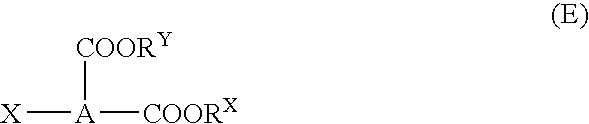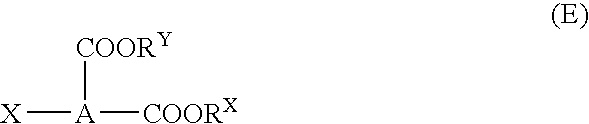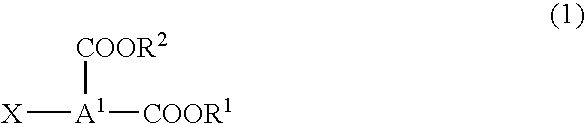Optimize Reaction Conditions for Dodecyl Acid Esterification
MAR 19, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Dodecyl Acid Esterification Background and Objectives
Dodecyl acid esterification represents a critical chemical transformation in the production of various industrial compounds, particularly in the synthesis of surfactants, lubricants, and specialty chemicals. This process involves the formation of ester bonds between dodecyl acid (lauric acid) and alcohols, creating valuable intermediates for downstream applications. The reaction has gained significant attention due to the growing demand for bio-based chemicals and sustainable manufacturing processes.
The historical development of esterification processes dates back to the early 20th century, with initial focus on simple acid-alcohol reactions using mineral acid catalysts. Over the decades, the field has evolved from basic homogeneous catalysis to sophisticated heterogeneous systems, enzymatic approaches, and continuous flow processes. The introduction of solid acid catalysts in the 1980s marked a pivotal advancement, addressing environmental concerns and simplifying product separation.
Current technological trends emphasize green chemistry principles, with increasing adoption of renewable feedstocks and environmentally benign catalysts. The shift toward sustainable processes has driven innovation in catalyst design, reaction engineering, and process intensification. Recent developments include the use of ionic liquids, deep eutectic solvents, and microreactor technologies to enhance reaction efficiency and selectivity.
The primary technical objectives for optimizing dodecyl acid esterification encompass several key areas. Maximizing conversion rates while maintaining high selectivity remains paramount, as incomplete conversion leads to product contamination and increased purification costs. Temperature and pressure optimization is crucial for balancing reaction kinetics with energy consumption and equipment requirements.
Catalyst selection and optimization represent another critical objective, focusing on developing systems that offer high activity, stability, and recyclability. The goal extends to minimizing catalyst deactivation and enabling easy separation from reaction products. Additionally, reducing reaction time through process intensification techniques can significantly improve economic viability.
Water removal strategies constitute a fundamental challenge, as esterification reactions are equilibrium-limited by water formation. Effective water management through reactive distillation, membrane separation, or molecular sieves is essential for achieving high conversions. The integration of separation processes with reaction systems offers opportunities for process optimization and cost reduction.
Environmental sustainability objectives include minimizing waste generation, reducing energy consumption, and eliminating hazardous solvents or catalysts. The development of continuous processes over traditional batch operations aligns with these goals while improving productivity and product consistency.
The historical development of esterification processes dates back to the early 20th century, with initial focus on simple acid-alcohol reactions using mineral acid catalysts. Over the decades, the field has evolved from basic homogeneous catalysis to sophisticated heterogeneous systems, enzymatic approaches, and continuous flow processes. The introduction of solid acid catalysts in the 1980s marked a pivotal advancement, addressing environmental concerns and simplifying product separation.
Current technological trends emphasize green chemistry principles, with increasing adoption of renewable feedstocks and environmentally benign catalysts. The shift toward sustainable processes has driven innovation in catalyst design, reaction engineering, and process intensification. Recent developments include the use of ionic liquids, deep eutectic solvents, and microreactor technologies to enhance reaction efficiency and selectivity.
The primary technical objectives for optimizing dodecyl acid esterification encompass several key areas. Maximizing conversion rates while maintaining high selectivity remains paramount, as incomplete conversion leads to product contamination and increased purification costs. Temperature and pressure optimization is crucial for balancing reaction kinetics with energy consumption and equipment requirements.
Catalyst selection and optimization represent another critical objective, focusing on developing systems that offer high activity, stability, and recyclability. The goal extends to minimizing catalyst deactivation and enabling easy separation from reaction products. Additionally, reducing reaction time through process intensification techniques can significantly improve economic viability.
Water removal strategies constitute a fundamental challenge, as esterification reactions are equilibrium-limited by water formation. Effective water management through reactive distillation, membrane separation, or molecular sieves is essential for achieving high conversions. The integration of separation processes with reaction systems offers opportunities for process optimization and cost reduction.
Environmental sustainability objectives include minimizing waste generation, reducing energy consumption, and eliminating hazardous solvents or catalysts. The development of continuous processes over traditional batch operations aligns with these goals while improving productivity and product consistency.
Market Demand for Dodecyl Ester Products
The global market for dodecyl ester products demonstrates robust growth driven by expanding applications across multiple industrial sectors. These versatile compounds serve as essential intermediates in personal care formulations, industrial lubricants, plasticizers, and specialty chemical applications. The cosmetics and personal care industry represents the largest consumption segment, where dodecyl esters function as emollients, conditioning agents, and texture enhancers in skincare products, hair care formulations, and color cosmetics.
Industrial applications constitute another significant demand driver, particularly in the lubricant sector where dodecyl esters provide superior thermal stability and biodegradability compared to conventional petroleum-based alternatives. The automotive and machinery industries increasingly favor these bio-based esters for hydraulic fluids, gear oils, and metalworking fluids due to stringent environmental regulations and sustainability initiatives.
The plasticizer market presents substantial opportunities for dodecyl ester products, especially in flexible PVC applications where they offer improved low-temperature performance and reduced volatility. Food packaging, medical devices, and consumer goods manufacturing sectors drive consistent demand for these safer plasticizer alternatives as regulatory pressure mounts against traditional phthalate-based compounds.
Pharmaceutical and nutraceutical industries utilize dodecyl esters as excipients and drug delivery enhancers, creating a specialized but high-value market segment. The growing trend toward controlled-release formulations and transdermal drug delivery systems supports steady demand growth in this sector.
Geographically, Asia-Pacific leads global consumption due to rapid industrialization and expanding personal care markets in China, India, and Southeast Asian countries. North America and Europe maintain strong demand driven by premium personal care products and stringent environmental regulations favoring bio-based chemicals. The shift toward sustainable and renewable feedstocks creates additional market momentum as manufacturers seek alternatives to petroleum-derived chemicals.
Market dynamics indicate increasing preference for high-purity, low-odor dodecyl ester variants, particularly in consumer-facing applications. This trend emphasizes the importance of optimizing esterification reaction conditions to achieve superior product quality and meet evolving market specifications while maintaining cost competitiveness in diverse application segments.
Industrial applications constitute another significant demand driver, particularly in the lubricant sector where dodecyl esters provide superior thermal stability and biodegradability compared to conventional petroleum-based alternatives. The automotive and machinery industries increasingly favor these bio-based esters for hydraulic fluids, gear oils, and metalworking fluids due to stringent environmental regulations and sustainability initiatives.
The plasticizer market presents substantial opportunities for dodecyl ester products, especially in flexible PVC applications where they offer improved low-temperature performance and reduced volatility. Food packaging, medical devices, and consumer goods manufacturing sectors drive consistent demand for these safer plasticizer alternatives as regulatory pressure mounts against traditional phthalate-based compounds.
Pharmaceutical and nutraceutical industries utilize dodecyl esters as excipients and drug delivery enhancers, creating a specialized but high-value market segment. The growing trend toward controlled-release formulations and transdermal drug delivery systems supports steady demand growth in this sector.
Geographically, Asia-Pacific leads global consumption due to rapid industrialization and expanding personal care markets in China, India, and Southeast Asian countries. North America and Europe maintain strong demand driven by premium personal care products and stringent environmental regulations favoring bio-based chemicals. The shift toward sustainable and renewable feedstocks creates additional market momentum as manufacturers seek alternatives to petroleum-derived chemicals.
Market dynamics indicate increasing preference for high-purity, low-odor dodecyl ester variants, particularly in consumer-facing applications. This trend emphasizes the importance of optimizing esterification reaction conditions to achieve superior product quality and meet evolving market specifications while maintaining cost competitiveness in diverse application segments.
Current Esterification Challenges and Limitations
Dodecyl acid esterification faces significant thermodynamic limitations that constrain reaction efficiency and product yield. The equilibrium nature of esterification reactions creates an inherent challenge where forward and reverse reactions compete simultaneously. This equilibrium typically favors the reactants rather than the desired ester products, particularly in aqueous environments where hydrolysis reactions become prominent. The presence of water as a byproduct further shifts the equilibrium backward, necessitating continuous water removal or specialized reaction conditions to drive the reaction toward completion.
Kinetic barriers present another substantial challenge in dodecyl acid esterification processes. The long-chain fatty acid structure of dodecyl acid creates steric hindrance that impedes the approach of alcohol molecules to the carboxyl group. This molecular crowding effect becomes more pronounced at higher concentrations, leading to reduced reaction rates and extended processing times. Additionally, the viscosity of dodecyl acid increases significantly at lower temperatures, further limiting molecular mobility and collision frequency between reactants.
Catalyst-related limitations significantly impact the efficiency of dodecyl acid esterification. Traditional acid catalysts, while effective in promoting protonation of the carboxyl group, often suffer from deactivation due to water formation during the reaction. Homogeneous catalysts face separation challenges, requiring additional purification steps that increase process complexity and costs. Heterogeneous catalysts, though easier to separate, frequently exhibit mass transfer limitations and reduced activity due to pore blockage by the long-chain fatty acid molecules.
Temperature control presents a critical challenge in optimizing dodecyl acid esterification conditions. While elevated temperatures generally increase reaction rates and help overcome activation energy barriers, they also promote undesirable side reactions such as thermal decomposition and oxidation of the fatty acid chains. These side reactions not only reduce product yield but also generate impurities that compromise product quality and require additional purification steps.
Mass transfer limitations become particularly problematic in dodecyl acid esterification due to the amphiphilic nature of the reactants and products. The formation of emulsions or phase separation can occur, creating interfacial barriers that limit reactant contact and reduce overall reaction efficiency. This challenge is exacerbated when using heterogeneous catalysts, where reactants must diffuse through catalyst pores that may become blocked by the bulky dodecyl acid molecules.
Product separation and purification represent ongoing challenges in dodecyl acid esterification processes. The similar boiling points and chemical properties of reactants and products make conventional separation techniques less effective. Unreacted dodecyl acid and alcohol must be efficiently separated from the desired ester product while minimizing product loss and maintaining economic viability of the overall process.
Kinetic barriers present another substantial challenge in dodecyl acid esterification processes. The long-chain fatty acid structure of dodecyl acid creates steric hindrance that impedes the approach of alcohol molecules to the carboxyl group. This molecular crowding effect becomes more pronounced at higher concentrations, leading to reduced reaction rates and extended processing times. Additionally, the viscosity of dodecyl acid increases significantly at lower temperatures, further limiting molecular mobility and collision frequency between reactants.
Catalyst-related limitations significantly impact the efficiency of dodecyl acid esterification. Traditional acid catalysts, while effective in promoting protonation of the carboxyl group, often suffer from deactivation due to water formation during the reaction. Homogeneous catalysts face separation challenges, requiring additional purification steps that increase process complexity and costs. Heterogeneous catalysts, though easier to separate, frequently exhibit mass transfer limitations and reduced activity due to pore blockage by the long-chain fatty acid molecules.
Temperature control presents a critical challenge in optimizing dodecyl acid esterification conditions. While elevated temperatures generally increase reaction rates and help overcome activation energy barriers, they also promote undesirable side reactions such as thermal decomposition and oxidation of the fatty acid chains. These side reactions not only reduce product yield but also generate impurities that compromise product quality and require additional purification steps.
Mass transfer limitations become particularly problematic in dodecyl acid esterification due to the amphiphilic nature of the reactants and products. The formation of emulsions or phase separation can occur, creating interfacial barriers that limit reactant contact and reduce overall reaction efficiency. This challenge is exacerbated when using heterogeneous catalysts, where reactants must diffuse through catalyst pores that may become blocked by the bulky dodecyl acid molecules.
Product separation and purification represent ongoing challenges in dodecyl acid esterification processes. The similar boiling points and chemical properties of reactants and products make conventional separation techniques less effective. Unreacted dodecyl acid and alcohol must be efficiently separated from the desired ester product while minimizing product loss and maintaining economic viability of the overall process.
Existing Dodecyl Acid Esterification Methods
01 Catalyst selection for dodecyl acid esterification
Various catalysts can be employed to facilitate the esterification reaction of dodecyl acid, including acid catalysts, enzyme catalysts, and solid acid catalysts. The choice of catalyst significantly affects the reaction rate, yield, and selectivity. Common catalysts include sulfuric acid, p-toluenesulfonic acid, lipases, and heterogeneous catalysts such as ion exchange resins. The catalyst concentration and type should be optimized based on the specific esterification system to achieve efficient conversion.- Catalyst selection for dodecyl acid esterification: Various catalysts can be employed to facilitate the esterification reaction of dodecyl acid, including acid catalysts, enzyme catalysts, and solid acid catalysts. The choice of catalyst significantly affects the reaction rate, yield, and selectivity. Common catalysts include sulfuric acid, p-toluenesulfonic acid, lipases, and heterogeneous catalysts such as ion exchange resins. The catalyst concentration and type should be optimized based on the specific esterification system to achieve efficient conversion.
- Temperature control in esterification reactions: Temperature is a critical parameter in dodecyl acid esterification reactions, typically ranging from room temperature to elevated temperatures. Higher temperatures generally increase reaction rates and conversion efficiency, but excessive heat may lead to side reactions or product degradation. The optimal temperature range is usually between 60-150°C depending on the catalyst system and reactants used. Temperature control must be balanced with energy efficiency and product quality considerations.
- Molar ratio optimization of reactants: The molar ratio between dodecyl acid and the alcohol component significantly influences the esterification equilibrium and conversion rate. Excess alcohol is often used to shift the equilibrium toward ester formation and improve yields. Typical molar ratios range from 1:1 to 1:5 or higher, depending on the specific reaction system. The optimal ratio must consider both economic factors and the ease of separating excess reactants from the final product.
- Water removal techniques during esterification: Since esterification is a reversible reaction that produces water as a byproduct, continuous removal of water is essential to drive the reaction toward completion. Various methods can be employed including azeotropic distillation, use of molecular sieves, Dean-Stark apparatus, or vacuum conditions. Effective water removal significantly improves conversion rates and reduces reaction time. The choice of water removal method depends on the scale of operation and the specific reaction conditions.
- Reaction time and pressure conditions: The duration of the esterification reaction varies depending on catalyst type, temperature, and desired conversion level, typically ranging from several hours to over 24 hours. Pressure conditions can also be adjusted, with some processes operating under atmospheric pressure while others employ reduced pressure to facilitate water removal or elevated pressure to increase reactant solubility. Optimization of reaction time and pressure helps balance production efficiency with energy costs and equipment requirements.
02 Temperature control in esterification process
Temperature is a critical parameter in dodecyl acid esterification reactions. The reaction temperature typically ranges from ambient temperature to elevated temperatures depending on the reactants and catalysts used. Higher temperatures generally increase reaction rates but may also promote side reactions or catalyst deactivation. Optimal temperature control ensures maximum conversion while maintaining product quality and minimizing energy consumption. Temperature profiles may be adjusted during different stages of the reaction to optimize yield.Expand Specific Solutions03 Molar ratio optimization of reactants
The molar ratio between dodecyl acid and the alcohol component is crucial for achieving high conversion rates in esterification reactions. Excess alcohol is often used to shift the equilibrium toward ester formation and compensate for alcohol loss through evaporation. The optimal molar ratio depends on factors such as catalyst type, reaction temperature, and desired conversion level. Proper ratio selection can improve reaction efficiency and reduce the need for extensive purification steps.Expand Specific Solutions04 Water removal techniques during esterification
Since esterification is a reversible reaction that produces water as a byproduct, continuous removal of water is essential to drive the reaction toward completion. Various methods can be employed including azeotropic distillation, use of molecular sieves, vacuum application, or Dean-Stark apparatus. Effective water removal increases the equilibrium conversion and reduces reaction time. The choice of water removal method depends on the reaction scale, equipment availability, and economic considerations.Expand Specific Solutions05 Reaction time and pressure conditions
The duration of the esterification reaction and the pressure conditions applied significantly influence the conversion efficiency and product quality. Reaction times can vary from several hours to continuous processes depending on the catalyst activity and reaction conditions. Atmospheric pressure is commonly used, but reduced pressure can facilitate water removal and lower the required reaction temperature. Pressure control may also prevent the loss of volatile reactants and improve overall process economics.Expand Specific Solutions
Key Players in Fatty Acid Esterification Industry
The dodecyl acid esterification optimization field represents a mature industrial chemistry sector with significant market presence, primarily driven by oleochemical and petrochemical applications. The industry has reached a stable development stage, characterized by established production processes and well-defined market segments spanning personal care, lubricants, and specialty chemicals. Market size reflects steady demand from downstream industries, with growth tied to consumer goods and industrial applications. Technology maturity varies significantly among key players, with established chemical giants like China Petroleum & Chemical Corp., PetroChina, and Wanhua Chemical Group demonstrating advanced process optimization capabilities through extensive R&D infrastructure. Japanese companies including New Japan Chemical and Kansai Paint contribute specialized oleochemical expertise, while pharmaceutical players like Pfizer, Eli Lilly, and Takeda bring sophisticated analytical and purification technologies. Research institutions such as Dalian Institute of Chemical Physics, Xi'an Petroleum University, and Nanyang Technological University provide fundamental research support, indicating strong academic-industry collaboration for process innovation and catalyst development in this established yet continuously evolving field.
China Petroleum & Chemical Corp.
Technical Solution: Sinopec has developed advanced catalytic esterification processes for fatty acid production, utilizing heterogeneous acid catalysts such as sulfonic acid-functionalized resins and solid acid catalysts. Their technology focuses on optimizing reaction temperature (150-200°C), pressure conditions (2-5 bar), and catalyst loading to achieve high conversion rates of dodecyl acid esterification. The company employs continuous flow reactors with integrated separation systems to enhance product purity and reduce reaction time. Their process includes real-time monitoring of acid value and ester content to maintain optimal reaction conditions throughout the esterification process.
Strengths: Large-scale industrial experience, integrated refining capabilities, cost-effective production. Weaknesses: Limited flexibility for specialty applications, environmental concerns with traditional processes.
Sinopec Research Institute of Petroleum Processing
Technical Solution: The institute has developed innovative esterification technologies using novel solid acid catalysts including zeolite-based and metal oxide catalysts for dodecyl acid esterification. Their research focuses on reaction kinetics optimization, achieving conversion rates above 95% through precise control of molar ratios, temperature profiles (160-180°C), and residence time distribution. They have pioneered microreactor technology for process intensification and developed predictive models for reaction optimization. The institute's approach includes advanced separation techniques using reactive distillation columns to simultaneously conduct esterification and product purification, significantly reducing energy consumption and improving overall process efficiency.
Strengths: Advanced R&D capabilities, innovative catalyst development, process optimization expertise. Weaknesses: Limited commercial scale implementation, higher development costs.
Core Patents in Esterification Optimization
Method and apparatus for preparing fatty acid alkyl ester using fatty acid
PatentWO2007126166A1
Innovation
- A method and apparatus for esterifying fatty acids with alcohol at high temperatures (200 to 350°C) and atmospheric pressure to 10 bar without using catalysts, involving a two-reactor system for complete conversion and distillation to remove impurities and recover excess alcohol, thereby achieving high conversion ratios and eliminating glycerin production.
Dicarboxylic diester, process for producing the same, and refrigerating machine lubricating oil comprising the ester
PatentInactiveUS7282601B2
Innovation
- Development of alicyclic dicarboxylic acid diesters and mixed diesters with specific structural properties, prepared through esterification or ester interchange reactions using specific alcohols and catalysts, which exhibit enhanced stability and adjustability for various viscosity grades.
Environmental Regulations for Chemical Processing
The chemical processing industry faces increasingly stringent environmental regulations that directly impact dodecyl acid esterification processes. These regulations encompass air quality standards, water discharge limits, waste management protocols, and greenhouse gas emission controls. The Environmental Protection Agency (EPA) and international regulatory bodies have established comprehensive frameworks governing volatile organic compound (VOC) emissions, which are particularly relevant to esterification reactions involving organic acids and alcohols.
Air emission standards represent a critical regulatory dimension for dodecyl acid esterification operations. The Clean Air Act mandates strict limits on VOC emissions, requiring facilities to implement best available control technology (BACT) for processes generating organic vapors. Esterification reactions typically produce water vapor and trace organic compounds, necessitating proper ventilation systems and emission control equipment. Facilities must maintain continuous emission monitoring systems and submit regular compliance reports to regulatory authorities.
Water discharge regulations significantly influence process design and waste treatment strategies. The Clean Water Act establishes effluent limitations for chemical manufacturing facilities, requiring comprehensive treatment of process wastewater before discharge. Esterification processes generate aqueous waste streams containing residual acids, alcohols, and catalyst materials that must be treated to meet stringent discharge standards for chemical oxygen demand, total organic carbon, and specific pollutant concentrations.
Waste management protocols govern the handling and disposal of solid and liquid waste streams generated during dodecyl acid esterification. The Resource Conservation and Recovery Act (RCRA) classifies certain organic waste materials as hazardous waste, requiring specialized handling, storage, and disposal procedures. Catalyst recovery and recycling programs have become essential for regulatory compliance and cost optimization, particularly for processes utilizing heavy metal catalysts or acidic materials.
Chemical safety regulations under the Occupational Safety and Health Administration (OSHA) mandate comprehensive safety protocols for handling dodecyl acid and associated reagents. These regulations require detailed safety data sheets, worker training programs, and emergency response procedures. Process safety management standards apply to facilities handling significant quantities of flammable or reactive materials, necessitating hazard analysis and risk assessment documentation.
Emerging regulations focus on sustainability metrics and carbon footprint reduction. The proposed climate disclosure rules require detailed reporting of scope 1, 2, and 3 emissions, driving optimization of energy-intensive esterification processes. Green chemistry principles are increasingly incorporated into regulatory frameworks, encouraging the adoption of environmentally benign catalysts and solvent-free reaction conditions.
Air emission standards represent a critical regulatory dimension for dodecyl acid esterification operations. The Clean Air Act mandates strict limits on VOC emissions, requiring facilities to implement best available control technology (BACT) for processes generating organic vapors. Esterification reactions typically produce water vapor and trace organic compounds, necessitating proper ventilation systems and emission control equipment. Facilities must maintain continuous emission monitoring systems and submit regular compliance reports to regulatory authorities.
Water discharge regulations significantly influence process design and waste treatment strategies. The Clean Water Act establishes effluent limitations for chemical manufacturing facilities, requiring comprehensive treatment of process wastewater before discharge. Esterification processes generate aqueous waste streams containing residual acids, alcohols, and catalyst materials that must be treated to meet stringent discharge standards for chemical oxygen demand, total organic carbon, and specific pollutant concentrations.
Waste management protocols govern the handling and disposal of solid and liquid waste streams generated during dodecyl acid esterification. The Resource Conservation and Recovery Act (RCRA) classifies certain organic waste materials as hazardous waste, requiring specialized handling, storage, and disposal procedures. Catalyst recovery and recycling programs have become essential for regulatory compliance and cost optimization, particularly for processes utilizing heavy metal catalysts or acidic materials.
Chemical safety regulations under the Occupational Safety and Health Administration (OSHA) mandate comprehensive safety protocols for handling dodecyl acid and associated reagents. These regulations require detailed safety data sheets, worker training programs, and emergency response procedures. Process safety management standards apply to facilities handling significant quantities of flammable or reactive materials, necessitating hazard analysis and risk assessment documentation.
Emerging regulations focus on sustainability metrics and carbon footprint reduction. The proposed climate disclosure rules require detailed reporting of scope 1, 2, and 3 emissions, driving optimization of energy-intensive esterification processes. Green chemistry principles are increasingly incorporated into regulatory frameworks, encouraging the adoption of environmentally benign catalysts and solvent-free reaction conditions.
Green Chemistry Approaches in Esterification
Green chemistry principles have revolutionized esterification processes by emphasizing environmental sustainability, atom economy, and energy efficiency. The application of these principles to dodecyl acid esterification represents a paradigm shift from traditional harsh reaction conditions toward more benign and sustainable synthetic approaches. This transformation addresses growing environmental concerns while maintaining or improving reaction efficiency and product quality.
Solvent-free esterification has emerged as a cornerstone of green chemistry approaches, eliminating the need for volatile organic compounds that pose environmental and health risks. Direct condensation reactions between dodecyl acid and alcohols under solvent-free conditions significantly reduce waste generation and simplify product purification processes. This approach aligns with the green chemistry principle of preventing waste rather than treating it after formation.
Biocatalytic esterification using lipases represents another significant advancement in sustainable synthesis. Enzymatic catalysts operate under mild reaction conditions, typically at temperatures below 80°C and atmospheric pressure, dramatically reducing energy consumption compared to traditional acid-catalyzed processes. Lipases demonstrate excellent selectivity and can be immobilized for repeated use, further enhancing the sustainability profile of the esterification process.
Microwave-assisted synthesis has gained prominence as an energy-efficient alternative for dodecyl acid esterification. This technology enables rapid heating through dielectric heating mechanisms, reducing reaction times from hours to minutes while maintaining high conversion rates. The selective heating nature of microwave irradiation minimizes side reactions and improves overall atom economy.
Ionic liquids serve as environmentally benign reaction media, offering negligible vapor pressure and excellent thermal stability. These designer solvents can be tailored to optimize reaction conditions while facilitating easy product separation and catalyst recovery. The recyclability of ionic liquids addresses sustainability concerns associated with traditional organic solvents.
Continuous flow chemistry represents an emerging green approach that enables precise control over reaction parameters while minimizing waste generation. Flow reactors provide enhanced heat and mass transfer, allowing for more efficient esterification processes with reduced environmental impact and improved safety profiles compared to batch operations.
Solvent-free esterification has emerged as a cornerstone of green chemistry approaches, eliminating the need for volatile organic compounds that pose environmental and health risks. Direct condensation reactions between dodecyl acid and alcohols under solvent-free conditions significantly reduce waste generation and simplify product purification processes. This approach aligns with the green chemistry principle of preventing waste rather than treating it after formation.
Biocatalytic esterification using lipases represents another significant advancement in sustainable synthesis. Enzymatic catalysts operate under mild reaction conditions, typically at temperatures below 80°C and atmospheric pressure, dramatically reducing energy consumption compared to traditional acid-catalyzed processes. Lipases demonstrate excellent selectivity and can be immobilized for repeated use, further enhancing the sustainability profile of the esterification process.
Microwave-assisted synthesis has gained prominence as an energy-efficient alternative for dodecyl acid esterification. This technology enables rapid heating through dielectric heating mechanisms, reducing reaction times from hours to minutes while maintaining high conversion rates. The selective heating nature of microwave irradiation minimizes side reactions and improves overall atom economy.
Ionic liquids serve as environmentally benign reaction media, offering negligible vapor pressure and excellent thermal stability. These designer solvents can be tailored to optimize reaction conditions while facilitating easy product separation and catalyst recovery. The recyclability of ionic liquids addresses sustainability concerns associated with traditional organic solvents.
Continuous flow chemistry represents an emerging green approach that enables precise control over reaction parameters while minimizing waste generation. Flow reactors provide enhanced heat and mass transfer, allowing for more efficient esterification processes with reduced environmental impact and improved safety profiles compared to batch operations.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!