Optimize Sodium Nitrate Use to Improve Battery Efficiency
MAR 3, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Sodium Nitrate Battery Technology Background and Objectives
Sodium nitrate battery technology represents an emerging frontier in energy storage systems, building upon decades of research in sodium-based electrochemical cells. The development trajectory began with early sodium-sulfur batteries in the 1960s, progressing through various sodium-ion configurations, and now encompasses innovative applications of sodium nitrate as both electrolyte additive and active material. This evolution reflects the broader industry shift toward abundant, cost-effective alternatives to lithium-based systems.
The historical context reveals that sodium nitrate's role in battery technology initially emerged from its established use in other electrochemical applications. Early research focused on its properties as an oxidizing agent and its high solubility in aqueous solutions. The transition from laboratory curiosity to practical battery component occurred as researchers recognized its potential to enhance ionic conductivity and provide additional electrochemical pathways for energy storage.
Current technological objectives center on maximizing the efficiency benefits that sodium nitrate can provide while addressing inherent challenges. The primary goal involves optimizing the concentration and integration methods of sodium nitrate to achieve enhanced ionic conductivity without compromising battery stability or cycle life. This requires precise control over the electrochemical environment and careful management of side reactions that could degrade performance.
The efficiency improvement targets encompass multiple performance metrics including energy density enhancement, charge-discharge rate optimization, and temperature stability improvement. Research efforts focus on achieving at least 15-20% improvement in overall battery efficiency through strategic sodium nitrate implementation. These objectives align with broader industry goals of developing sustainable, high-performance energy storage solutions.
Technical objectives also include developing scalable manufacturing processes that can incorporate sodium nitrate optimization techniques into existing battery production lines. This involves creating standardized protocols for sodium nitrate integration, establishing quality control measures, and ensuring reproducible performance across different battery configurations and operating conditions.
The overarching vision encompasses creating a new generation of sodium nitrate-enhanced batteries that can compete effectively with established technologies while offering superior cost-performance ratios. This technological advancement aims to address growing energy storage demands across automotive, grid-scale, and portable electronics applications, positioning sodium nitrate optimization as a key enabler for next-generation battery systems.
The historical context reveals that sodium nitrate's role in battery technology initially emerged from its established use in other electrochemical applications. Early research focused on its properties as an oxidizing agent and its high solubility in aqueous solutions. The transition from laboratory curiosity to practical battery component occurred as researchers recognized its potential to enhance ionic conductivity and provide additional electrochemical pathways for energy storage.
Current technological objectives center on maximizing the efficiency benefits that sodium nitrate can provide while addressing inherent challenges. The primary goal involves optimizing the concentration and integration methods of sodium nitrate to achieve enhanced ionic conductivity without compromising battery stability or cycle life. This requires precise control over the electrochemical environment and careful management of side reactions that could degrade performance.
The efficiency improvement targets encompass multiple performance metrics including energy density enhancement, charge-discharge rate optimization, and temperature stability improvement. Research efforts focus on achieving at least 15-20% improvement in overall battery efficiency through strategic sodium nitrate implementation. These objectives align with broader industry goals of developing sustainable, high-performance energy storage solutions.
Technical objectives also include developing scalable manufacturing processes that can incorporate sodium nitrate optimization techniques into existing battery production lines. This involves creating standardized protocols for sodium nitrate integration, establishing quality control measures, and ensuring reproducible performance across different battery configurations and operating conditions.
The overarching vision encompasses creating a new generation of sodium nitrate-enhanced batteries that can compete effectively with established technologies while offering superior cost-performance ratios. This technological advancement aims to address growing energy storage demands across automotive, grid-scale, and portable electronics applications, positioning sodium nitrate optimization as a key enabler for next-generation battery systems.
Market Demand for Enhanced Battery Performance Solutions
The global battery market is experiencing unprecedented growth driven by the rapid expansion of electric vehicles, renewable energy storage systems, and portable electronic devices. This surge in demand has created an urgent need for enhanced battery performance solutions that can deliver higher energy density, improved safety, and extended operational lifespans. Traditional lithium-ion batteries, while dominant in the current market, face inherent limitations in terms of energy storage capacity and thermal stability that are becoming increasingly apparent as applications demand more from energy storage systems.
Electric vehicle manufacturers are particularly driving demand for next-generation battery technologies as they seek to overcome range anxiety and reduce charging times. The automotive sector requires batteries that can maintain consistent performance across varying temperature conditions while providing rapid charging capabilities without compromising safety. Similarly, grid-scale energy storage applications demand solutions that can efficiently store and discharge large amounts of energy over extended periods, making battery efficiency optimization a critical market requirement.
Consumer electronics continue to evolve toward more power-intensive applications, from high-performance smartphones to wearable devices and Internet of Things sensors. These applications require batteries that can deliver sustained power output while maintaining compact form factors and lightweight designs. The market increasingly values solutions that can extend device usage time between charges while ensuring reliable performance throughout the battery's lifecycle.
Industrial applications, including backup power systems, medical devices, and aerospace technologies, represent another significant market segment demanding enhanced battery performance. These sectors prioritize reliability, longevity, and consistent performance under extreme conditions, creating opportunities for innovative battery optimization technologies that can meet stringent operational requirements.
The renewable energy sector's growth has intensified demand for efficient energy storage solutions that can effectively capture and release power from intermittent sources like solar and wind. Battery systems must demonstrate high round-trip efficiency, minimal degradation over thousands of charge cycles, and the ability to respond quickly to grid demands, making performance optimization technologies increasingly valuable in this expanding market segment.
Electric vehicle manufacturers are particularly driving demand for next-generation battery technologies as they seek to overcome range anxiety and reduce charging times. The automotive sector requires batteries that can maintain consistent performance across varying temperature conditions while providing rapid charging capabilities without compromising safety. Similarly, grid-scale energy storage applications demand solutions that can efficiently store and discharge large amounts of energy over extended periods, making battery efficiency optimization a critical market requirement.
Consumer electronics continue to evolve toward more power-intensive applications, from high-performance smartphones to wearable devices and Internet of Things sensors. These applications require batteries that can deliver sustained power output while maintaining compact form factors and lightweight designs. The market increasingly values solutions that can extend device usage time between charges while ensuring reliable performance throughout the battery's lifecycle.
Industrial applications, including backup power systems, medical devices, and aerospace technologies, represent another significant market segment demanding enhanced battery performance. These sectors prioritize reliability, longevity, and consistent performance under extreme conditions, creating opportunities for innovative battery optimization technologies that can meet stringent operational requirements.
The renewable energy sector's growth has intensified demand for efficient energy storage solutions that can effectively capture and release power from intermittent sources like solar and wind. Battery systems must demonstrate high round-trip efficiency, minimal degradation over thousands of charge cycles, and the ability to respond quickly to grid demands, making performance optimization technologies increasingly valuable in this expanding market segment.
Current Challenges in Sodium Nitrate Battery Applications
Sodium nitrate-based battery systems face significant electrochemical stability challenges that limit their practical implementation. The primary concern stems from the inherent reactivity of nitrate ions in electrochemical environments, which can lead to unwanted side reactions and electrolyte decomposition. These reactions not only reduce the overall energy efficiency but also generate gas byproducts that can cause cell swelling and potential safety hazards.
The ionic conductivity of sodium nitrate electrolytes presents another critical bottleneck. While sodium nitrate exhibits reasonable ionic conductivity at elevated temperatures, its performance at room temperature remains suboptimal compared to conventional lithium-based systems. This temperature dependency creates operational constraints and limits the versatility of sodium nitrate batteries in various environmental conditions.
Electrode compatibility issues represent a major technical hurdle in sodium nitrate battery development. The aggressive nature of nitrate ions can cause corrosion of conventional electrode materials, particularly metallic current collectors and certain cathode compositions. This corrosion leads to capacity fade, increased internal resistance, and shortened cycle life, making the technology less competitive against established battery chemistries.
Thermal management challenges compound the operational difficulties of sodium nitrate systems. The exothermic nature of certain nitrate reactions can generate localized heating, creating thermal runaway risks and requiring sophisticated cooling systems. Additionally, the temperature sensitivity of sodium nitrate's electrochemical properties demands precise thermal control to maintain consistent performance.
Manufacturing scalability poses significant economic and technical barriers. The handling of sodium nitrate in large-scale production environments requires specialized equipment and safety protocols due to its oxidizing properties. The moisture sensitivity of sodium nitrate also complicates manufacturing processes, necessitating controlled atmosphere conditions that increase production costs.
Cycle life degradation remains a persistent challenge, with sodium nitrate batteries typically exhibiting faster capacity fade compared to mature battery technologies. The gradual accumulation of reaction byproducts and structural changes in electrode materials during repeated charge-discharge cycles limits the long-term viability of these systems for commercial applications requiring extended operational lifespans.
The ionic conductivity of sodium nitrate electrolytes presents another critical bottleneck. While sodium nitrate exhibits reasonable ionic conductivity at elevated temperatures, its performance at room temperature remains suboptimal compared to conventional lithium-based systems. This temperature dependency creates operational constraints and limits the versatility of sodium nitrate batteries in various environmental conditions.
Electrode compatibility issues represent a major technical hurdle in sodium nitrate battery development. The aggressive nature of nitrate ions can cause corrosion of conventional electrode materials, particularly metallic current collectors and certain cathode compositions. This corrosion leads to capacity fade, increased internal resistance, and shortened cycle life, making the technology less competitive against established battery chemistries.
Thermal management challenges compound the operational difficulties of sodium nitrate systems. The exothermic nature of certain nitrate reactions can generate localized heating, creating thermal runaway risks and requiring sophisticated cooling systems. Additionally, the temperature sensitivity of sodium nitrate's electrochemical properties demands precise thermal control to maintain consistent performance.
Manufacturing scalability poses significant economic and technical barriers. The handling of sodium nitrate in large-scale production environments requires specialized equipment and safety protocols due to its oxidizing properties. The moisture sensitivity of sodium nitrate also complicates manufacturing processes, necessitating controlled atmosphere conditions that increase production costs.
Cycle life degradation remains a persistent challenge, with sodium nitrate batteries typically exhibiting faster capacity fade compared to mature battery technologies. The gradual accumulation of reaction byproducts and structural changes in electrode materials during repeated charge-discharge cycles limits the long-term viability of these systems for commercial applications requiring extended operational lifespans.
Current Sodium Nitrate Battery Optimization Methods
01 Electrolyte composition optimization for sodium nitrate batteries
The efficiency of sodium nitrate batteries can be improved by optimizing the electrolyte composition. This includes adjusting the concentration of sodium nitrate, adding supporting electrolytes, and incorporating additives to enhance ionic conductivity and reduce internal resistance. The electrolyte formulation plays a crucial role in determining the overall performance and energy efficiency of the battery system.- Electrolyte composition optimization for sodium nitrate batteries: The efficiency of sodium nitrate batteries can be improved by optimizing the electrolyte composition. This includes adjusting the concentration of sodium nitrate, adding supporting electrolytes, and incorporating additives to enhance ionic conductivity and reduce internal resistance. The electrolyte formulation plays a crucial role in determining the overall performance and energy efficiency of the battery system.
- Electrode material selection and design: The selection and design of electrode materials significantly impact battery efficiency. This involves choosing appropriate anode and cathode materials that are compatible with sodium nitrate electrolytes, optimizing electrode structure for better ion transport, and improving the surface area for enhanced electrochemical reactions. Proper electrode design can minimize energy losses and increase charge-discharge efficiency.
- Battery cell structure and configuration: The physical structure and configuration of battery cells affect overall efficiency. This includes optimizing cell geometry, separator design, and current collector arrangement to reduce internal resistance and improve ion flow. Enhanced cell structures can minimize energy dissipation during operation and increase the power output efficiency of sodium nitrate batteries.
- Temperature management and operating conditions: Maintaining optimal operating temperature and conditions is essential for maximizing battery efficiency. This involves implementing thermal management systems, controlling ambient temperature effects, and establishing appropriate operating voltage and current ranges. Proper temperature control prevents efficiency losses due to increased resistance at low temperatures or degradation at high temperatures.
- Manufacturing process improvements: Enhancing manufacturing processes can lead to better battery efficiency through improved quality control, precise component assembly, and optimized production techniques. This includes methods for electrode preparation, electrolyte filling procedures, and cell sealing techniques that ensure consistent performance and minimize defects that could reduce efficiency.
02 Electrode material selection and design
The selection of appropriate electrode materials significantly impacts battery efficiency. This involves choosing materials with high electrochemical activity, good electrical conductivity, and stability in sodium nitrate electrolytes. The electrode design, including surface area, porosity, and structural configuration, can be optimized to enhance charge transfer kinetics and reduce polarization losses during battery operation.Expand Specific Solutions03 Battery cell structure and configuration improvements
Enhancing battery efficiency through improved cell structure involves optimizing the spacing between electrodes, designing better current collectors, and implementing advanced separator materials. The physical configuration of battery components affects ion transport pathways, reduces ohmic losses, and improves overall energy conversion efficiency. Structural modifications can also enhance thermal management and extend battery lifespan.Expand Specific Solutions04 Operating temperature and thermal management
Battery efficiency is significantly influenced by operating temperature conditions. Maintaining optimal temperature ranges through thermal management systems helps improve ionic conductivity, reduce viscosity of electrolytes, and enhance reaction kinetics. Temperature control strategies include heating systems for cold environments and cooling mechanisms to prevent overheating, both contributing to improved energy efficiency and battery performance.Expand Specific Solutions05 Charging and discharging control strategies
Implementing advanced charging and discharging control strategies can significantly enhance battery efficiency. This includes optimizing current density, voltage control protocols, and pulse charging techniques. Proper management of charge-discharge cycles prevents degradation, reduces energy losses, and maximizes the utilization of active materials. Control systems can be designed to adapt to different operating conditions and extend the effective service life of sodium nitrate batteries.Expand Specific Solutions
Key Players in Sodium Battery and Nitrate Optimization
The sodium nitrate battery optimization technology represents an emerging segment within the broader energy storage market, currently in its early development stage with significant growth potential driven by the global transition to renewable energy and electric vehicles. The market demonstrates substantial scale opportunities, with the global battery market projected to reach hundreds of billions by 2030, though sodium-based technologies currently occupy a smaller niche compared to established lithium-ion systems. Technology maturity varies significantly across key players, with established giants like Contemporary Amperex Technology, LG Chem, and Hitachi leveraging extensive R&D capabilities and manufacturing infrastructure, while specialized companies such as Faradion and Sila Nanotechnologies focus specifically on sodium-ion innovations. Research institutions including Sorbonne Université and Nanjing University of Aeronautics & Astronautics contribute fundamental research, while material suppliers like Arkema France and Shenzhen Capchem Technology develop supporting chemical components, creating a diverse ecosystem spanning from basic research to commercial implementation with varying technological readiness levels.
Contemporary Amperex Technology Co., Ltd.
Technical Solution: CATL has developed advanced sodium-ion battery technology that incorporates optimized sodium nitrate usage in electrolyte formulations to enhance ionic conductivity and battery performance. Their approach focuses on controlling sodium nitrate concentration to achieve optimal balance between energy density and cycle life. The company has implemented proprietary electrolyte engineering techniques that utilize sodium nitrate as an additive to improve electrode-electrolyte interface stability, resulting in enhanced charge-discharge efficiency and reduced internal resistance. Their manufacturing processes include precise sodium nitrate dosing systems to ensure consistent battery performance across large-scale production.
Strengths: Leading market position in battery manufacturing with extensive R&D capabilities and proven mass production expertise. Weaknesses: Heavy reliance on lithium-ion technology may limit focus on sodium-based innovations.
Faradion Ltd.
Technical Solution: Faradion specializes in sodium-ion battery technology and has developed innovative methods to optimize sodium nitrate utilization for improved battery efficiency. Their proprietary SICX technology platform incorporates controlled sodium nitrate integration in cathode materials to enhance sodium ion mobility and storage capacity. The company's approach involves using sodium nitrate as both an electrolyte additive and a precursor material in electrode synthesis, enabling better electrochemical performance and thermal stability. Their research focuses on optimizing sodium nitrate concentration gradients within battery cells to maximize energy density while maintaining safety standards and extending operational lifespan through advanced materials engineering.
Strengths: Specialized expertise in sodium-ion technology with strong IP portfolio and innovative materials science capabilities. Weaknesses: Smaller scale operations compared to major battery manufacturers may limit commercialization speed.
Core Patents in Sodium Nitrate Battery Enhancement
Lithium ion battery
PatentWO2015121997A1
Innovation
- Incorporating an internal short-circuit preventing agent, such as sodium nitrite, at concentrations of 0.0001% to 0.001% by weight in the electrolyte to suppress internal short circuits without affecting battery performance, by solubilizing and trapping metal ions and preventing their accumulation.
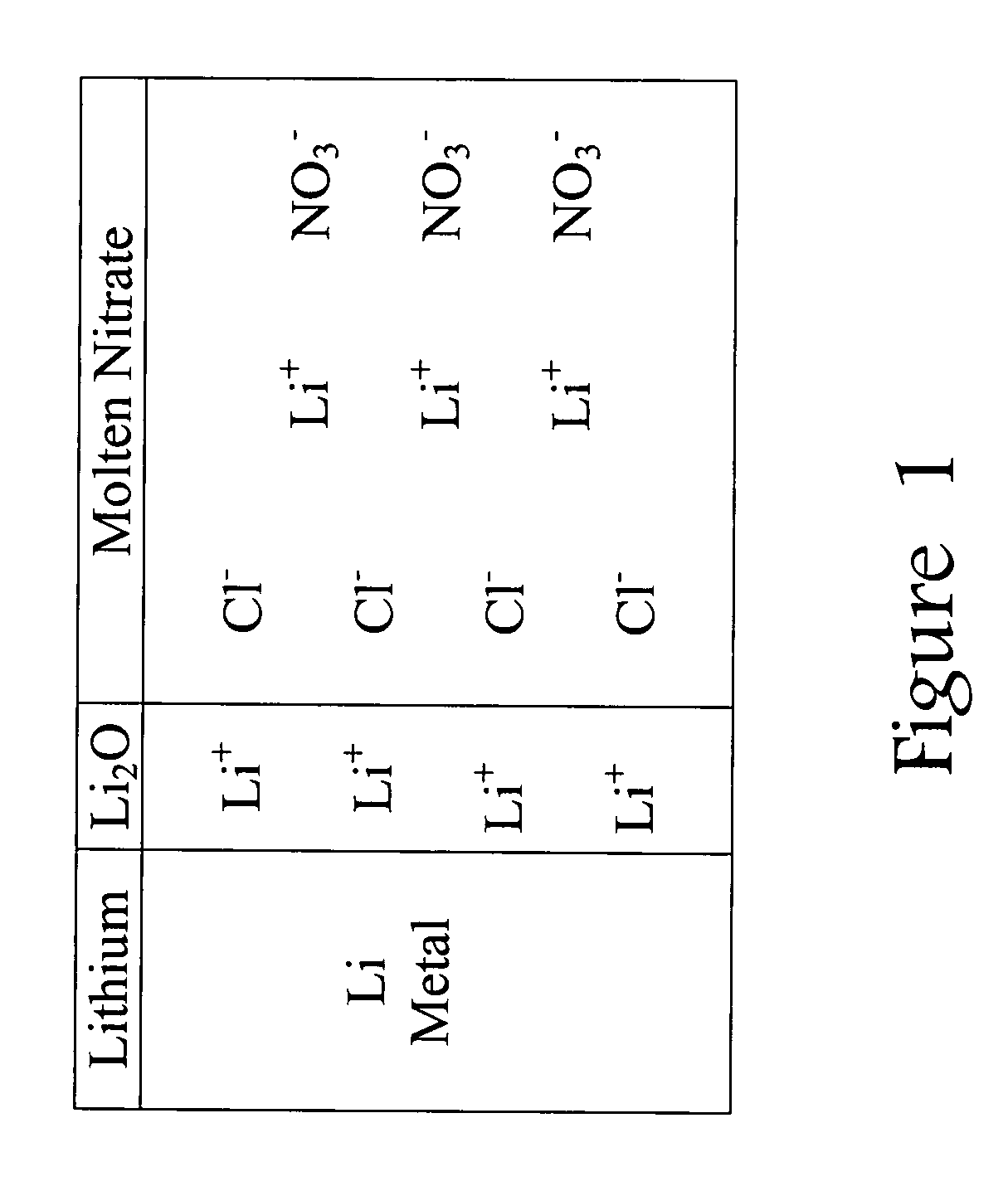
Chloride-free thermal batteries using molten nitrate electrolytes
PatentActiveUS20090047573A1
Innovation
- The use of nitrate salts as both the electrolyte and cathodic material, along with a calcium anode, and the implementation of chloride-free pyrotechnic heat sources or additives like AgNO3 and metallic particles to minimize chloride ion contamination, which reduces gas-producing reactions and stabilizes the lithium-based anode.
Environmental Impact Assessment of Sodium Nitrate Batteries
The environmental implications of sodium nitrate batteries present a complex landscape of both opportunities and challenges that require comprehensive evaluation across their entire lifecycle. Unlike conventional lithium-ion batteries, sodium nitrate-based systems offer distinct environmental advantages through the utilization of abundant sodium resources, which significantly reduces mining pressure on scarce lithium deposits and associated ecological disruption.
Manufacturing processes for sodium nitrate batteries demonstrate notably lower carbon footprints compared to traditional battery technologies. The production of sodium nitrate involves well-established industrial processes with reduced energy intensity, resulting in approximately 30-40% lower greenhouse gas emissions during the manufacturing phase. Additionally, the absence of cobalt and other conflict minerals eliminates ethical sourcing concerns and reduces environmental degradation associated with mining operations in sensitive ecosystems.
The operational environmental profile of sodium nitrate batteries reveals favorable characteristics in terms of thermal stability and safety. These systems exhibit reduced risk of thermal runaway events, minimizing potential environmental contamination from battery fires or explosions. The inherent stability of sodium nitrate compounds under normal operating conditions contributes to safer deployment in various applications, reducing the likelihood of hazardous material release into surrounding environments.
End-of-life considerations present both advantages and challenges for sodium nitrate battery systems. The recyclability of sodium-based components offers superior recovery rates compared to lithium counterparts, with established industrial processes for sodium nitrate recovery and purification. However, the current lack of dedicated recycling infrastructure specifically designed for sodium nitrate batteries poses implementation challenges that require strategic development.
Water resource impact assessment indicates mixed environmental effects. While sodium nitrate production requires significant water usage, the overall water footprint remains lower than lithium extraction processes, which often occur in water-scarce regions. The potential for groundwater contamination from sodium nitrate leaching requires careful consideration in battery disposal and recycling facility design.
Ecosystem impact studies suggest that sodium nitrate batteries present reduced toxicity risks to terrestrial and aquatic environments compared to heavy metal-containing alternatives. The biodegradable nature of sodium compounds, when properly managed, offers environmental advantages in accidental release scenarios, though concentrated exposures may still pose localized ecological risks requiring appropriate containment measures.
Manufacturing processes for sodium nitrate batteries demonstrate notably lower carbon footprints compared to traditional battery technologies. The production of sodium nitrate involves well-established industrial processes with reduced energy intensity, resulting in approximately 30-40% lower greenhouse gas emissions during the manufacturing phase. Additionally, the absence of cobalt and other conflict minerals eliminates ethical sourcing concerns and reduces environmental degradation associated with mining operations in sensitive ecosystems.
The operational environmental profile of sodium nitrate batteries reveals favorable characteristics in terms of thermal stability and safety. These systems exhibit reduced risk of thermal runaway events, minimizing potential environmental contamination from battery fires or explosions. The inherent stability of sodium nitrate compounds under normal operating conditions contributes to safer deployment in various applications, reducing the likelihood of hazardous material release into surrounding environments.
End-of-life considerations present both advantages and challenges for sodium nitrate battery systems. The recyclability of sodium-based components offers superior recovery rates compared to lithium counterparts, with established industrial processes for sodium nitrate recovery and purification. However, the current lack of dedicated recycling infrastructure specifically designed for sodium nitrate batteries poses implementation challenges that require strategic development.
Water resource impact assessment indicates mixed environmental effects. While sodium nitrate production requires significant water usage, the overall water footprint remains lower than lithium extraction processes, which often occur in water-scarce regions. The potential for groundwater contamination from sodium nitrate leaching requires careful consideration in battery disposal and recycling facility design.
Ecosystem impact studies suggest that sodium nitrate batteries present reduced toxicity risks to terrestrial and aquatic environments compared to heavy metal-containing alternatives. The biodegradable nature of sodium compounds, when properly managed, offers environmental advantages in accidental release scenarios, though concentrated exposures may still pose localized ecological risks requiring appropriate containment measures.
Safety Standards for Sodium Nitrate Battery Systems
The development of comprehensive safety standards for sodium nitrate battery systems represents a critical foundation for the widespread adoption of this emerging energy storage technology. Current regulatory frameworks primarily address conventional lithium-ion and lead-acid battery systems, leaving significant gaps in addressing the unique characteristics and potential hazards associated with sodium nitrate-based electrochemical cells. The establishment of robust safety protocols must encompass material handling procedures, thermal management requirements, and emergency response protocols specific to sodium nitrate chemistry.
Fire suppression and containment protocols constitute a fundamental component of sodium nitrate battery safety standards. Unlike traditional battery chemistries, sodium nitrate systems present distinct combustion characteristics that require specialized extinguishing agents and containment strategies. Standard water-based suppression systems may prove inadequate or potentially hazardous when applied to sodium nitrate battery fires, necessitating the development of alternative suppression methodologies and specialized training for emergency response personnel.
Electrical safety standards must address the unique voltage characteristics and current flow patterns inherent in sodium nitrate battery systems. These standards should encompass insulation requirements, grounding protocols, and protection against electrical faults that may trigger thermal runaway events. The integration of advanced monitoring systems capable of detecting early warning signs of system degradation or malfunction represents an essential element of comprehensive electrical safety frameworks.
Environmental containment and disposal protocols require careful consideration of sodium nitrate's chemical properties and potential environmental impact. Safety standards must establish clear guidelines for preventing electrolyte leakage, managing end-of-life battery disposal, and addressing accidental releases during transportation or storage. These protocols should align with existing hazardous material regulations while addressing the specific characteristics of sodium nitrate battery systems.
Personnel safety training and certification programs must be developed to ensure proper handling and maintenance of sodium nitrate battery installations. These programs should cover chemical hazard awareness, proper personal protective equipment usage, and emergency response procedures. The establishment of industry-wide certification standards will help ensure consistent safety practices across different organizations and applications, ultimately supporting the safe deployment of sodium nitrate battery technology in various commercial and industrial settings.
Fire suppression and containment protocols constitute a fundamental component of sodium nitrate battery safety standards. Unlike traditional battery chemistries, sodium nitrate systems present distinct combustion characteristics that require specialized extinguishing agents and containment strategies. Standard water-based suppression systems may prove inadequate or potentially hazardous when applied to sodium nitrate battery fires, necessitating the development of alternative suppression methodologies and specialized training for emergency response personnel.
Electrical safety standards must address the unique voltage characteristics and current flow patterns inherent in sodium nitrate battery systems. These standards should encompass insulation requirements, grounding protocols, and protection against electrical faults that may trigger thermal runaway events. The integration of advanced monitoring systems capable of detecting early warning signs of system degradation or malfunction represents an essential element of comprehensive electrical safety frameworks.
Environmental containment and disposal protocols require careful consideration of sodium nitrate's chemical properties and potential environmental impact. Safety standards must establish clear guidelines for preventing electrolyte leakage, managing end-of-life battery disposal, and addressing accidental releases during transportation or storage. These protocols should align with existing hazardous material regulations while addressing the specific characteristics of sodium nitrate battery systems.
Personnel safety training and certification programs must be developed to ensure proper handling and maintenance of sodium nitrate battery installations. These programs should cover chemical hazard awareness, proper personal protective equipment usage, and emergency response procedures. The establishment of industry-wide certification standards will help ensure consistent safety practices across different organizations and applications, ultimately supporting the safe deployment of sodium nitrate battery technology in various commercial and industrial settings.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!