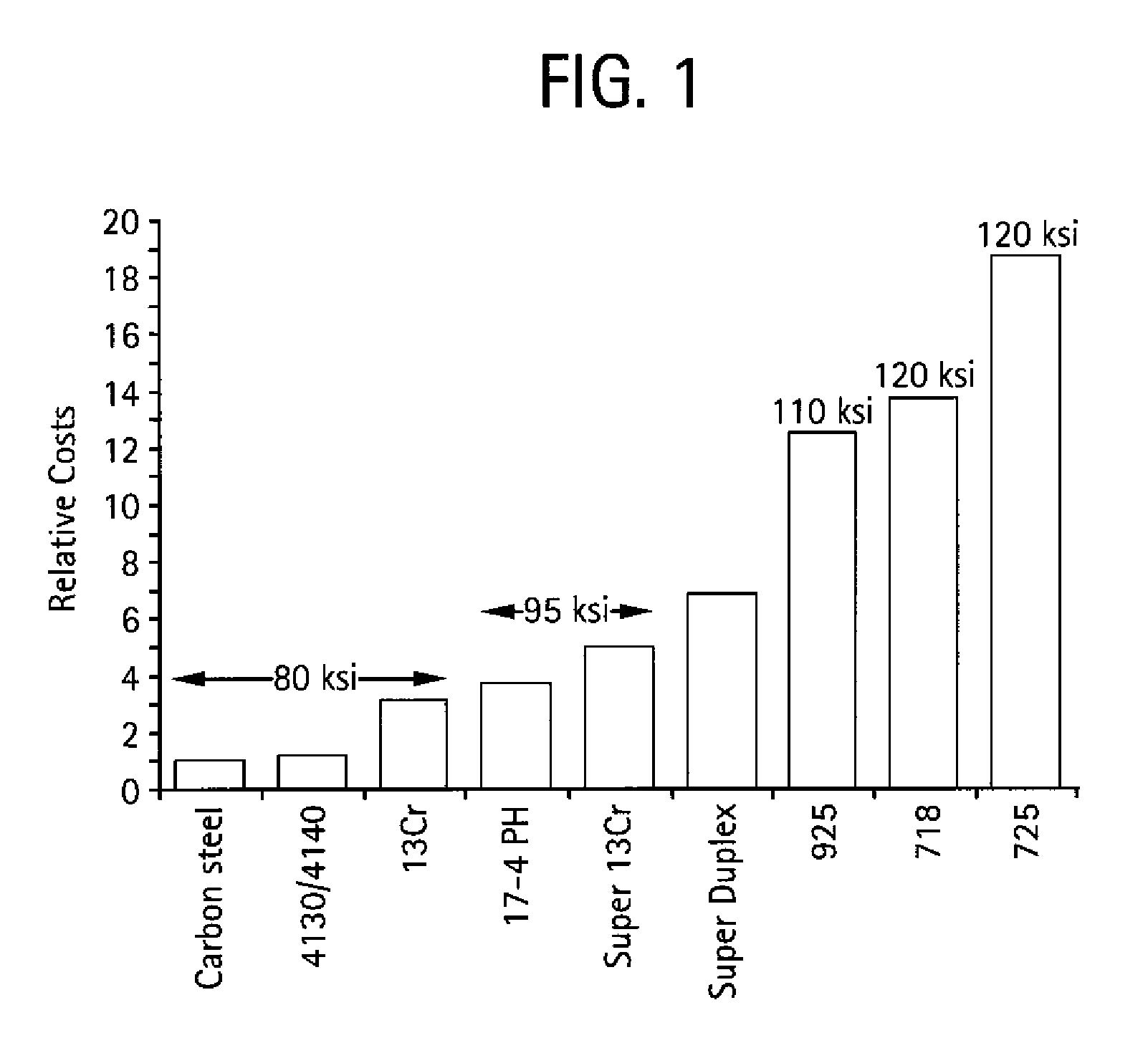
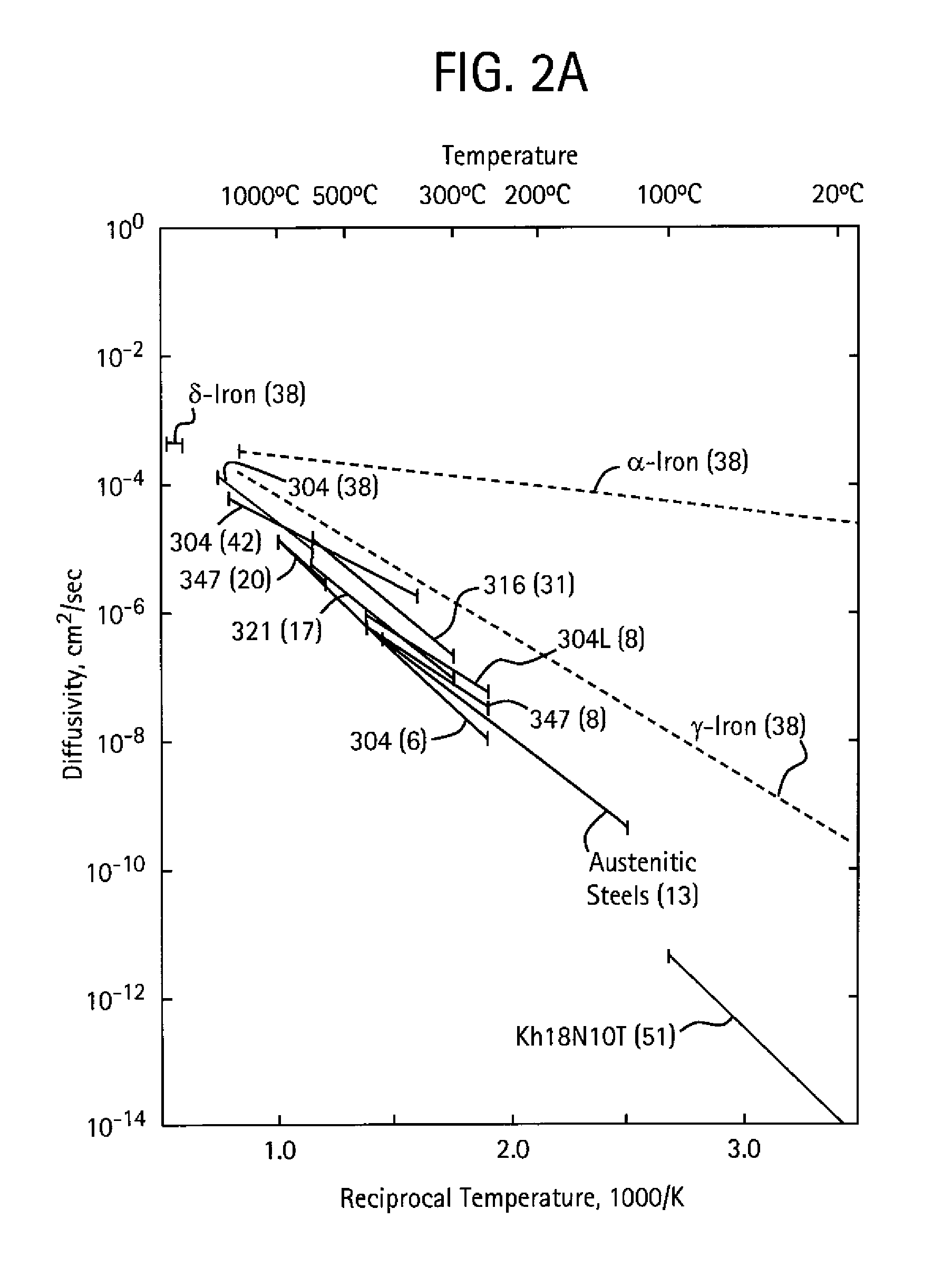
Optimizing Interstitial Alloy Composition for Corrosion Resistance
APR 28, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Interstitial Alloy Development Background and Corrosion Goals
Interstitial alloys represent a fundamental class of metallic materials where small atoms such as carbon, nitrogen, boron, or hydrogen occupy the interstitial sites within the crystal lattice of host metals. This atomic arrangement creates unique mechanical and chemical properties that have been exploited for centuries, with early examples including carbon steel and case-hardened iron tools. The systematic understanding of interstitial alloying began in the early 20th century with advances in metallurgical science and X-ray crystallography.
The evolution of interstitial alloy technology has progressed through distinct phases, beginning with empirical approaches in traditional steelmaking to modern computational materials design. The development of stainless steels in the 1910s marked a pivotal moment, demonstrating how controlled interstitial carbon content combined with chromium additions could dramatically enhance corrosion resistance. Subsequent decades witnessed the emergence of specialized interstitial alloys for aerospace, chemical processing, and marine applications.
Contemporary interstitial alloy development leverages advanced characterization techniques including atom probe tomography, high-resolution electron microscopy, and synchrotron radiation studies. These tools enable precise control over interstitial atom distribution and concentration, facilitating the design of alloys with tailored corrosion resistance properties. The integration of computational thermodynamics and first-principles calculations has accelerated the discovery of novel interstitial compositions.
Current technological objectives focus on achieving superior corrosion resistance through optimized interstitial element selection and distribution control. Primary goals include developing alloys that maintain structural integrity in aggressive environments such as high-temperature oxidizing atmospheres, chloride-rich solutions, and acidic conditions. The target is to create materials that exhibit passive film stability, reduced susceptibility to localized corrosion phenomena, and enhanced repassivation kinetics.
Advanced manufacturing techniques including powder metallurgy, additive manufacturing, and surface engineering enable precise control over interstitial atom incorporation and spatial distribution. These approaches support the development of gradient compositions and functionally graded materials that optimize corrosion resistance while maintaining mechanical performance requirements for specific applications.
The evolution of interstitial alloy technology has progressed through distinct phases, beginning with empirical approaches in traditional steelmaking to modern computational materials design. The development of stainless steels in the 1910s marked a pivotal moment, demonstrating how controlled interstitial carbon content combined with chromium additions could dramatically enhance corrosion resistance. Subsequent decades witnessed the emergence of specialized interstitial alloys for aerospace, chemical processing, and marine applications.
Contemporary interstitial alloy development leverages advanced characterization techniques including atom probe tomography, high-resolution electron microscopy, and synchrotron radiation studies. These tools enable precise control over interstitial atom distribution and concentration, facilitating the design of alloys with tailored corrosion resistance properties. The integration of computational thermodynamics and first-principles calculations has accelerated the discovery of novel interstitial compositions.
Current technological objectives focus on achieving superior corrosion resistance through optimized interstitial element selection and distribution control. Primary goals include developing alloys that maintain structural integrity in aggressive environments such as high-temperature oxidizing atmospheres, chloride-rich solutions, and acidic conditions. The target is to create materials that exhibit passive film stability, reduced susceptibility to localized corrosion phenomena, and enhanced repassivation kinetics.
Advanced manufacturing techniques including powder metallurgy, additive manufacturing, and surface engineering enable precise control over interstitial atom incorporation and spatial distribution. These approaches support the development of gradient compositions and functionally graded materials that optimize corrosion resistance while maintaining mechanical performance requirements for specific applications.
Market Demand for Corrosion-Resistant Alloy Materials
The global demand for corrosion-resistant alloy materials has experienced substantial growth across multiple industrial sectors, driven by the increasing need for durable materials that can withstand harsh environmental conditions. Industries such as oil and gas, chemical processing, marine engineering, and aerospace have become primary consumers of advanced corrosion-resistant alloys, as these sectors face escalating challenges from aggressive chemical environments, extreme temperatures, and prolonged exposure to corrosive media.
The oil and gas industry represents one of the largest market segments for corrosion-resistant alloys, particularly in offshore drilling operations, pipeline systems, and refinery equipment. The harsh conditions encountered in deep-water exploration and the processing of increasingly sour crude oil have necessitated the development of superior alloy compositions that can maintain structural integrity over extended operational periods.
Chemical processing facilities have similarly driven demand for specialized alloy materials capable of resisting various corrosive chemicals, acids, and high-temperature environments. The pharmaceutical and petrochemical industries specifically require materials that not only resist corrosion but also maintain purity standards, creating a niche market for premium alloy compositions.
The marine industry has witnessed growing demand for corrosion-resistant materials due to expanding offshore wind energy projects, shipbuilding activities, and underwater infrastructure development. Seawater's highly corrosive nature, combined with mechanical stress and temperature variations, has created specific requirements for alloy compositions optimized for marine applications.
Aerospace and defense sectors continue to seek advanced materials that can withstand extreme environmental conditions while maintaining lightweight properties. The increasing focus on fuel efficiency and performance has intensified the demand for innovative alloy compositions that balance corrosion resistance with mechanical properties.
Infrastructure development in emerging economies has further expanded the market, as governments and private entities invest in long-lasting materials for bridges, buildings, and transportation systems. The growing emphasis on sustainability and lifecycle cost optimization has shifted procurement strategies toward higher-performance materials that offer extended service life despite higher initial costs.
Market trends indicate a strong preference for customized alloy solutions tailored to specific application requirements, rather than generic corrosion-resistant materials. This shift has created opportunities for specialized alloy manufacturers to develop targeted compositions that address particular corrosion mechanisms and environmental challenges.
The oil and gas industry represents one of the largest market segments for corrosion-resistant alloys, particularly in offshore drilling operations, pipeline systems, and refinery equipment. The harsh conditions encountered in deep-water exploration and the processing of increasingly sour crude oil have necessitated the development of superior alloy compositions that can maintain structural integrity over extended operational periods.
Chemical processing facilities have similarly driven demand for specialized alloy materials capable of resisting various corrosive chemicals, acids, and high-temperature environments. The pharmaceutical and petrochemical industries specifically require materials that not only resist corrosion but also maintain purity standards, creating a niche market for premium alloy compositions.
The marine industry has witnessed growing demand for corrosion-resistant materials due to expanding offshore wind energy projects, shipbuilding activities, and underwater infrastructure development. Seawater's highly corrosive nature, combined with mechanical stress and temperature variations, has created specific requirements for alloy compositions optimized for marine applications.
Aerospace and defense sectors continue to seek advanced materials that can withstand extreme environmental conditions while maintaining lightweight properties. The increasing focus on fuel efficiency and performance has intensified the demand for innovative alloy compositions that balance corrosion resistance with mechanical properties.
Infrastructure development in emerging economies has further expanded the market, as governments and private entities invest in long-lasting materials for bridges, buildings, and transportation systems. The growing emphasis on sustainability and lifecycle cost optimization has shifted procurement strategies toward higher-performance materials that offer extended service life despite higher initial costs.
Market trends indicate a strong preference for customized alloy solutions tailored to specific application requirements, rather than generic corrosion-resistant materials. This shift has created opportunities for specialized alloy manufacturers to develop targeted compositions that address particular corrosion mechanisms and environmental challenges.
Current Challenges in Interstitial Alloy Corrosion Performance
Interstitial alloys face significant corrosion performance challenges that stem from their complex microstructural characteristics and the intricate relationship between interstitial elements and the host matrix. The primary challenge lies in achieving uniform distribution of interstitial atoms such as carbon, nitrogen, and boron within the metallic lattice, as localized concentration variations create galvanic cells that accelerate corrosion initiation and propagation.
The solubility limits of interstitial elements present a fundamental constraint in alloy design. When these limits are exceeded, precipitation of secondary phases occurs, leading to compositional heterogeneity and the formation of preferential corrosion sites. These precipitates often exhibit different electrochemical potentials compared to the matrix, establishing micro-galvanic couples that compromise the overall corrosion resistance of the alloy system.
Grain boundary segregation represents another critical challenge, where interstitial elements tend to accumulate at grain boundaries, altering the local chemistry and creating susceptible pathways for intergranular corrosion. This phenomenon is particularly pronounced in high-temperature applications where diffusion kinetics favor segregation processes, leading to premature failure of protective oxide layers.
The interaction between multiple interstitial elements introduces additional complexity, as synergistic or antagonistic effects can occur depending on the specific combination and concentration ratios. Carbon and nitrogen co-doping, for instance, can lead to competitive precipitation reactions that affect the stability and protective nature of surface films, making it difficult to predict long-term corrosion behavior.
Processing-induced defects such as dislocations, vacancies, and stacking faults serve as preferential sites for interstitial atom trapping, creating non-equilibrium distributions that persist even after thermal treatments. These defects act as stress concentrators and provide easy diffusion paths for aggressive species, significantly reducing the alloy's resistance to localized corrosion modes including pitting and crevice corrosion.
Environmental factors further complicate the corrosion performance of interstitial alloys. Temperature fluctuations can cause redistribution of interstitial elements, while the presence of specific aggressive ions can selectively attack regions with higher interstitial concentrations. The challenge is compounded by the difficulty in establishing reliable accelerated testing protocols that accurately predict long-term performance under service conditions.
The solubility limits of interstitial elements present a fundamental constraint in alloy design. When these limits are exceeded, precipitation of secondary phases occurs, leading to compositional heterogeneity and the formation of preferential corrosion sites. These precipitates often exhibit different electrochemical potentials compared to the matrix, establishing micro-galvanic couples that compromise the overall corrosion resistance of the alloy system.
Grain boundary segregation represents another critical challenge, where interstitial elements tend to accumulate at grain boundaries, altering the local chemistry and creating susceptible pathways for intergranular corrosion. This phenomenon is particularly pronounced in high-temperature applications where diffusion kinetics favor segregation processes, leading to premature failure of protective oxide layers.
The interaction between multiple interstitial elements introduces additional complexity, as synergistic or antagonistic effects can occur depending on the specific combination and concentration ratios. Carbon and nitrogen co-doping, for instance, can lead to competitive precipitation reactions that affect the stability and protective nature of surface films, making it difficult to predict long-term corrosion behavior.
Processing-induced defects such as dislocations, vacancies, and stacking faults serve as preferential sites for interstitial atom trapping, creating non-equilibrium distributions that persist even after thermal treatments. These defects act as stress concentrators and provide easy diffusion paths for aggressive species, significantly reducing the alloy's resistance to localized corrosion modes including pitting and crevice corrosion.
Environmental factors further complicate the corrosion performance of interstitial alloys. Temperature fluctuations can cause redistribution of interstitial elements, while the presence of specific aggressive ions can selectively attack regions with higher interstitial concentrations. The challenge is compounded by the difficulty in establishing reliable accelerated testing protocols that accurately predict long-term performance under service conditions.
Current Interstitial Alloy Optimization Solutions
01 Alloy composition optimization for interstitial corrosion resistance
Specific alloy compositions can be designed to enhance resistance to interstitial corrosion by controlling the distribution and concentration of alloying elements. The optimization involves balancing carbon, nitrogen, and other interstitial elements to minimize the formation of carbides and nitrides at grain boundaries, which are primary sites for corrosion initiation. Proper compositional control helps maintain the integrity of the protective oxide layer and reduces susceptibility to localized corrosion attacks.- Alloy composition optimization for interstitial corrosion resistance: Specific alloy compositions can be designed to enhance resistance to interstitial corrosion by controlling the distribution and concentration of alloying elements. The optimization involves balancing carbon, nitrogen, and other interstitial elements to minimize the formation of carbides and nitrides at grain boundaries, which are primary sites for corrosion initiation. Proper compositional control helps maintain the integrity of the protective oxide layer and reduces susceptibility to localized corrosion attacks.
- Heat treatment processes for improved corrosion resistance: Controlled heat treatment processes can significantly improve the interstitial corrosion resistance of alloys by modifying the microstructure and redistributing interstitial elements. These processes include solution annealing, controlled cooling rates, and specific temperature profiles that help dissolve harmful precipitates and create a more uniform distribution of elements throughout the matrix. The heat treatment parameters are carefully optimized to achieve the desired balance between mechanical properties and corrosion resistance.
- Surface modification techniques for enhanced protection: Various surface modification methods can be employed to improve the interstitial corrosion resistance of alloys. These techniques involve creating protective surface layers, modifying surface chemistry, or introducing beneficial surface stresses that inhibit corrosion initiation. The modifications can include surface alloying, coating applications, or mechanical treatments that alter the surface microstructure to provide better resistance to corrosive environments.
- Microstructural control and grain boundary engineering: The control of microstructural features, particularly grain boundaries and precipitate distribution, plays a crucial role in enhancing interstitial corrosion resistance. Engineering approaches focus on refining grain size, controlling precipitate morphology, and optimizing the distribution of secondary phases to minimize corrosion-susceptible sites. This involves careful control of processing parameters during manufacturing to achieve the desired microstructural characteristics that provide superior corrosion performance.
- Protective coating systems and barrier layers: Advanced protective coating systems and barrier layers can be applied to provide enhanced interstitial corrosion resistance. These systems work by creating physical and chemical barriers that prevent corrosive species from reaching the underlying alloy substrate. The coatings may incorporate corrosion inhibitors, provide cathodic protection, or form stable passive layers that maintain their protective properties under various environmental conditions. The selection and application of these protective systems are tailored to specific service environments and performance requirements.
02 Heat treatment processes for improved corrosion resistance
Controlled heat treatment processes can significantly improve the interstitial corrosion resistance of alloys by optimizing the microstructure and phase distribution. These processes involve specific temperature profiles, cooling rates, and annealing treatments that help dissolve harmful precipitates and redistribute interstitial elements uniformly throughout the matrix. The heat treatment also promotes the formation of stable phases that enhance the overall corrosion resistance properties.Expand Specific Solutions03 Surface modification techniques for enhanced protection
Various surface modification methods can be employed to improve the interstitial corrosion resistance of alloys. These techniques include surface alloying, coating applications, and surface treatment processes that create protective barriers or modify the surface chemistry. The modifications help prevent the initiation of corrosion at the surface level and provide additional protection against aggressive environments that could lead to interstitial corrosion.Expand Specific Solutions04 Microstructural control and grain boundary engineering
Controlling the microstructure and engineering grain boundaries play crucial roles in enhancing interstitial corrosion resistance. This involves managing grain size, grain boundary character, and the distribution of secondary phases along grain boundaries. Proper microstructural control helps minimize the segregation of harmful elements at grain boundaries and reduces the formation of corrosion-susceptible phases. Advanced processing techniques can be used to achieve optimal grain boundary structures that resist corrosion initiation and propagation.Expand Specific Solutions05 Protective coating systems and barrier layers
Development of specialized protective coating systems and barrier layers provides effective protection against interstitial corrosion. These systems include metallic coatings, ceramic layers, and composite coatings that act as physical and chemical barriers between the base alloy and corrosive environments. The coatings are designed to be chemically inert, mechanically stable, and capable of self-healing properties to maintain long-term protection against various forms of corrosion including interstitial attacks.Expand Specific Solutions
Major Players in Advanced Alloy and Materials Industry
The interstitial alloy composition optimization for corrosion resistance represents a mature technology field experiencing steady growth, driven by increasing demands from aerospace, automotive, and energy sectors. The market demonstrates significant expansion potential, particularly in high-performance applications requiring enhanced durability. Technology maturity varies considerably across market players, with established steel manufacturers like NIPPON STEEL CORP., JFE Steel Corp., and Sanyo Special Steel leading in traditional alloy development, while companies such as QuesTek Innovations LLC pioneer computational materials design approaches. Research institutions including Southwest Research Institute and Yokohama National University contribute fundamental breakthroughs in alloy science. Aluminum specialists like Novelis Inc., UACJ Corp., and Speira GmbH focus on lightweight corrosion-resistant solutions, while specialty steel producers such as voestalpine BÖHLER Edelstahl and Nippon Yakin Kogyo advance high-performance interstitial alloys for demanding industrial applications.
Southwest Research Institute
Technical Solution: Southwest Research Institute conducts comprehensive research on interstitial alloy optimization through systematic studies of carbon, nitrogen, and boron effects on corrosion mechanisms. Their approach combines electrochemical testing, surface analysis, and microstructural characterization to understand how interstitial elements influence passive film formation and stability. The institute develops predictive models for corrosion behavior based on interstitial element distribution and develops processing guidelines for achieving optimal compositions. Their research covers various alloy systems including stainless steels, aluminum alloys, and titanium alloys, with emphasis on understanding the fundamental relationships between interstitial chemistry and corrosion resistance.
Strengths: Strong research capabilities and comprehensive testing facilities for corrosion evaluation. Weaknesses: Focus on research rather than commercial production and longer development timelines.
ATI Properties, Inc.
Technical Solution: ATI focuses on optimizing interstitial alloy compositions in specialty metals, particularly titanium and nickel-based alloys. Their approach involves controlling oxygen, nitrogen, and carbon content to enhance corrosion resistance in aerospace and chemical processing applications. The company utilizes vacuum arc remelting and electron beam melting technologies to achieve precise interstitial element control. Their research emphasizes the role of interstitial elements in forming stable passive films and reducing susceptibility to localized corrosion. ATI's proprietary alloy designs incorporate optimized interstitial chemistry to improve performance in chloride-containing environments and high-temperature oxidizing conditions.
Strengths: Expertise in specialty metals and advanced melting technologies for precise composition control. Weaknesses: Higher material costs compared to conventional alloys and limited applicability to high-volume applications.
Core Patents in Corrosion-Resistant Alloy Composition
Interstitially strengthened high carbon and high nitrogen austenitic alloys, oilfield apparatus comprising same, and methods of making and using same
PatentInactiveUS7658883B2
Innovation
- Development of carbon-plus-nitrogen austenitic ferrous alloys with high interstitial carbon and nitrogen content, ranging from 0.4 to 1.2 wt.%, that are predominantly austenitic, offering enhanced strength, toughness, and corrosion resistance without the need for cold working, and utilizing chromium, manganese, and molybdenum to improve nitrogen solubility and corrosion resistance.
Corrosion and wear resistant alloy
PatentInactiveUS6702905B1
Innovation
- An iron-based alloy composition with specific weight percentages of boron, carbon, vanadium, chromium, niobium, molybdenum, and optional cobalt, silicon, and nickel, heat-treated to achieve a martensitic microstructure with primary and secondary carbides, offering enhanced hot hardness, wear resistance, and corrosion resistance.
Environmental Standards for Corrosion-Resistant Materials
Environmental standards for corrosion-resistant materials have evolved significantly over the past decades, driven by increasing awareness of material performance requirements across diverse industrial applications. These standards establish critical benchmarks for evaluating the effectiveness of interstitial alloy compositions in resisting various forms of environmental degradation.
The International Organization for Standardization (ISO) has developed comprehensive frameworks, particularly ISO 17025 and ISO 9227, which define testing methodologies for corrosion resistance evaluation. These standards specify exposure conditions, testing durations, and acceptance criteria that directly influence alloy composition optimization strategies. The American Society for Testing and Materials (ASTM) complements these with standards such as ASTM G48 for pitting corrosion resistance and ASTM G61 for cyclic polarization measurements.
Regional environmental regulations significantly impact material selection criteria. The European Union's REACH regulation restricts certain alloying elements, necessitating alternative interstitial compositions that maintain corrosion resistance while ensuring regulatory compliance. Similarly, the U.S. Environmental Protection Agency's guidelines for marine applications establish specific performance thresholds that influence nickel, chromium, and nitrogen content optimization in austenitic stainless steels.
Industry-specific standards further refine these requirements. The aerospace sector adheres to ASTM F3039 for biocompatible alloys, while the petrochemical industry follows NACE MR0175 for sour service applications. These specialized standards often mandate enhanced resistance to specific corrosive environments, driving research toward novel interstitial element combinations such as nitrogen-enhanced duplex stainless steels.
Emerging environmental standards increasingly emphasize lifecycle assessment and sustainability metrics. The ISO 14040 series now influences material selection by incorporating environmental impact considerations beyond mere corrosion performance. This shift promotes the development of lean alloy compositions that achieve superior corrosion resistance through optimized interstitial element distribution rather than increased alloying content.
Testing protocol standardization ensures reproducible evaluation of optimized compositions. Salt spray testing duration, solution chemistry, and specimen preparation methods are strictly regulated, enabling reliable comparison of different interstitial alloy formulations across research institutions and manufacturing facilities worldwide.
The International Organization for Standardization (ISO) has developed comprehensive frameworks, particularly ISO 17025 and ISO 9227, which define testing methodologies for corrosion resistance evaluation. These standards specify exposure conditions, testing durations, and acceptance criteria that directly influence alloy composition optimization strategies. The American Society for Testing and Materials (ASTM) complements these with standards such as ASTM G48 for pitting corrosion resistance and ASTM G61 for cyclic polarization measurements.
Regional environmental regulations significantly impact material selection criteria. The European Union's REACH regulation restricts certain alloying elements, necessitating alternative interstitial compositions that maintain corrosion resistance while ensuring regulatory compliance. Similarly, the U.S. Environmental Protection Agency's guidelines for marine applications establish specific performance thresholds that influence nickel, chromium, and nitrogen content optimization in austenitic stainless steels.
Industry-specific standards further refine these requirements. The aerospace sector adheres to ASTM F3039 for biocompatible alloys, while the petrochemical industry follows NACE MR0175 for sour service applications. These specialized standards often mandate enhanced resistance to specific corrosive environments, driving research toward novel interstitial element combinations such as nitrogen-enhanced duplex stainless steels.
Emerging environmental standards increasingly emphasize lifecycle assessment and sustainability metrics. The ISO 14040 series now influences material selection by incorporating environmental impact considerations beyond mere corrosion performance. This shift promotes the development of lean alloy compositions that achieve superior corrosion resistance through optimized interstitial element distribution rather than increased alloying content.
Testing protocol standardization ensures reproducible evaluation of optimized compositions. Salt spray testing duration, solution chemistry, and specimen preparation methods are strictly regulated, enabling reliable comparison of different interstitial alloy formulations across research institutions and manufacturing facilities worldwide.
Sustainability Considerations in Alloy Manufacturing
The development of corrosion-resistant interstitial alloys presents significant sustainability challenges that must be addressed throughout the manufacturing lifecycle. Traditional alloy production processes often involve energy-intensive extraction methods, high-temperature processing, and the use of rare earth elements, contributing to substantial carbon footprints and environmental degradation.
Raw material sourcing represents a critical sustainability bottleneck in interstitial alloy manufacturing. Many corrosion-resistant alloys require elements such as chromium, nickel, and molybdenum, which are often extracted through environmentally intensive mining operations. The geographic concentration of these resources in specific regions creates supply chain vulnerabilities and geopolitical dependencies that impact long-term sustainability planning.
Energy consumption during alloy processing constitutes another major environmental concern. High-temperature melting, controlled atmosphere processing, and precision heat treatment cycles required for optimizing interstitial element distribution demand substantial energy inputs. Modern manufacturing facilities are increasingly adopting renewable energy sources and implementing energy recovery systems to reduce their environmental impact while maintaining the precise thermal control necessary for achieving desired corrosion resistance properties.
Waste management and circular economy principles are becoming integral to sustainable alloy manufacturing. Advanced recycling technologies enable the recovery of valuable alloying elements from end-of-life products, reducing dependence on virgin materials. However, the complex composition of interstitial alloys presents challenges in separation and purification processes, requiring innovative metallurgical techniques to maintain material quality standards.
Water usage and treatment represent additional sustainability considerations, particularly in cooling systems and surface treatment processes. Closed-loop water systems and advanced filtration technologies are being implemented to minimize freshwater consumption and prevent contamination of local water sources.
The industry is increasingly adopting life cycle assessment methodologies to evaluate the environmental impact of different alloy compositions and manufacturing routes. This approach enables manufacturers to make informed decisions about trade-offs between corrosion performance and environmental sustainability, driving innovation toward more eco-friendly production methods while maintaining the superior corrosion resistance required for critical applications.
Raw material sourcing represents a critical sustainability bottleneck in interstitial alloy manufacturing. Many corrosion-resistant alloys require elements such as chromium, nickel, and molybdenum, which are often extracted through environmentally intensive mining operations. The geographic concentration of these resources in specific regions creates supply chain vulnerabilities and geopolitical dependencies that impact long-term sustainability planning.
Energy consumption during alloy processing constitutes another major environmental concern. High-temperature melting, controlled atmosphere processing, and precision heat treatment cycles required for optimizing interstitial element distribution demand substantial energy inputs. Modern manufacturing facilities are increasingly adopting renewable energy sources and implementing energy recovery systems to reduce their environmental impact while maintaining the precise thermal control necessary for achieving desired corrosion resistance properties.
Waste management and circular economy principles are becoming integral to sustainable alloy manufacturing. Advanced recycling technologies enable the recovery of valuable alloying elements from end-of-life products, reducing dependence on virgin materials. However, the complex composition of interstitial alloys presents challenges in separation and purification processes, requiring innovative metallurgical techniques to maintain material quality standards.
Water usage and treatment represent additional sustainability considerations, particularly in cooling systems and surface treatment processes. Closed-loop water systems and advanced filtration technologies are being implemented to minimize freshwater consumption and prevent contamination of local water sources.
The industry is increasingly adopting life cycle assessment methodologies to evaluate the environmental impact of different alloy compositions and manufacturing routes. This approach enables manufacturers to make informed decisions about trade-offs between corrosion performance and environmental sustainability, driving innovation toward more eco-friendly production methods while maintaining the superior corrosion resistance required for critical applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!