PCA Pump vs Basal Systems: Efficacy Under Stress
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PCA vs Basal Pain Management Technology Background and Goals
Pain management technology has undergone significant evolution since the introduction of patient-controlled analgesia (PCA) systems in the 1970s. Initially developed to address the limitations of traditional nurse-administered analgesics, PCA pumps represented a paradigm shift toward patient-centered pain control. The technology emerged from clinical observations that patients experienced better pain relief when they could self-administer medication within predetermined safety parameters.
Basal infusion systems, conversely, evolved from continuous intravenous therapy principles established in the mid-20th century. These systems were designed to provide steady-state medication delivery, ensuring consistent plasma drug concentrations without patient intervention. The integration of programmable infusion pumps in the 1980s enhanced precision and reliability, making basal systems a cornerstone of perioperative and chronic pain management protocols.
The convergence of these technologies has created hybrid systems combining continuous basal infusion with patient-controlled bolus capabilities. This evolution reflects growing understanding of pain physiology and the recognition that optimal analgesia often requires both baseline coverage and responsive dosing flexibility. Modern systems incorporate sophisticated algorithms, wireless connectivity, and advanced safety features that were inconceivable in early implementations.
Current technological trends emphasize smart pump integration, real-time monitoring capabilities, and data analytics for personalized pain management protocols. The development trajectory shows increasing focus on closed-loop systems that can automatically adjust delivery parameters based on physiological feedback and patient response patterns.
The primary technological goal in comparing PCA and basal systems under stress conditions centers on optimizing analgesic efficacy while maintaining patient safety during physiologically demanding situations. Stress-induced alterations in pharmacokinetics, including changes in drug distribution, metabolism, and clearance, present unique challenges for both delivery modalities. The objective is to determine which system architecture provides superior pain control when patients experience acute physiological stress from surgery, trauma, or critical illness.
Secondary goals include minimizing adverse effects, reducing healthcare provider workload, and improving patient satisfaction during high-stress clinical scenarios. The technology aims to address the complex interplay between stress-induced catecholamine release, altered pain perception, and modified drug response patterns that can compromise conventional pain management approaches.
Long-term objectives focus on developing evidence-based protocols that can guide clinicians in selecting optimal delivery modalities based on individual patient characteristics, stress severity, and clinical context. This includes establishing predictive models that can anticipate analgesic requirements and automatically adjust delivery parameters to maintain therapeutic efficacy throughout varying stress conditions.
Basal infusion systems, conversely, evolved from continuous intravenous therapy principles established in the mid-20th century. These systems were designed to provide steady-state medication delivery, ensuring consistent plasma drug concentrations without patient intervention. The integration of programmable infusion pumps in the 1980s enhanced precision and reliability, making basal systems a cornerstone of perioperative and chronic pain management protocols.
The convergence of these technologies has created hybrid systems combining continuous basal infusion with patient-controlled bolus capabilities. This evolution reflects growing understanding of pain physiology and the recognition that optimal analgesia often requires both baseline coverage and responsive dosing flexibility. Modern systems incorporate sophisticated algorithms, wireless connectivity, and advanced safety features that were inconceivable in early implementations.
Current technological trends emphasize smart pump integration, real-time monitoring capabilities, and data analytics for personalized pain management protocols. The development trajectory shows increasing focus on closed-loop systems that can automatically adjust delivery parameters based on physiological feedback and patient response patterns.
The primary technological goal in comparing PCA and basal systems under stress conditions centers on optimizing analgesic efficacy while maintaining patient safety during physiologically demanding situations. Stress-induced alterations in pharmacokinetics, including changes in drug distribution, metabolism, and clearance, present unique challenges for both delivery modalities. The objective is to determine which system architecture provides superior pain control when patients experience acute physiological stress from surgery, trauma, or critical illness.
Secondary goals include minimizing adverse effects, reducing healthcare provider workload, and improving patient satisfaction during high-stress clinical scenarios. The technology aims to address the complex interplay between stress-induced catecholamine release, altered pain perception, and modified drug response patterns that can compromise conventional pain management approaches.
Long-term objectives focus on developing evidence-based protocols that can guide clinicians in selecting optimal delivery modalities based on individual patient characteristics, stress severity, and clinical context. This includes establishing predictive models that can anticipate analgesic requirements and automatically adjust delivery parameters to maintain therapeutic efficacy throughout varying stress conditions.
Market Demand for Stress-Responsive Pain Management Systems
The global pain management market is experiencing unprecedented growth driven by aging populations, increased surgical procedures, and heightened awareness of patient-centered care approaches. Healthcare systems worldwide are recognizing the critical need for more sophisticated pain management solutions that can adapt to varying physiological and psychological stress conditions. This demand is particularly acute in intensive care units, post-surgical recovery environments, and chronic pain management settings where traditional fixed-dosing protocols often prove inadequate.
Stress-responsive pain management systems represent a paradigm shift from conventional approaches, addressing the fundamental limitation that pain perception and analgesic requirements fluctuate significantly based on patient stress levels, circadian rhythms, and individual physiological responses. The market demand for such adaptive systems stems from mounting evidence that stress-induced pain amplification can compromise patient outcomes and extend recovery times.
Hospital administrators and healthcare providers are increasingly seeking technologies that can automatically adjust pain medication delivery based on real-time physiological indicators such as heart rate variability, cortisol levels, and autonomic nervous system activity. This demand is particularly strong in developed markets where healthcare quality metrics and patient satisfaction scores directly impact institutional reimbursements and competitive positioning.
The economic drivers supporting market demand include reduced nursing workload through automated dose adjustments, decreased medication waste through precision delivery, and improved patient throughput due to optimized pain control. Healthcare systems are also motivated by the potential to reduce opioid-related adverse events and dependency risks through more precise, stress-responsive dosing algorithms.
Regulatory bodies are showing increased receptiveness to innovative pain management technologies that demonstrate improved safety profiles and clinical outcomes. This regulatory environment is fostering market confidence and encouraging investment in stress-responsive systems development.
The market demand extends beyond acute care settings to encompass home healthcare, palliative care, and chronic pain management applications. Patients and caregivers are increasingly advocating for personalized pain management solutions that can adapt to daily stress variations and lifestyle factors, creating additional market pressure for technological advancement in this domain.
Stress-responsive pain management systems represent a paradigm shift from conventional approaches, addressing the fundamental limitation that pain perception and analgesic requirements fluctuate significantly based on patient stress levels, circadian rhythms, and individual physiological responses. The market demand for such adaptive systems stems from mounting evidence that stress-induced pain amplification can compromise patient outcomes and extend recovery times.
Hospital administrators and healthcare providers are increasingly seeking technologies that can automatically adjust pain medication delivery based on real-time physiological indicators such as heart rate variability, cortisol levels, and autonomic nervous system activity. This demand is particularly strong in developed markets where healthcare quality metrics and patient satisfaction scores directly impact institutional reimbursements and competitive positioning.
The economic drivers supporting market demand include reduced nursing workload through automated dose adjustments, decreased medication waste through precision delivery, and improved patient throughput due to optimized pain control. Healthcare systems are also motivated by the potential to reduce opioid-related adverse events and dependency risks through more precise, stress-responsive dosing algorithms.
Regulatory bodies are showing increased receptiveness to innovative pain management technologies that demonstrate improved safety profiles and clinical outcomes. This regulatory environment is fostering market confidence and encouraging investment in stress-responsive systems development.
The market demand extends beyond acute care settings to encompass home healthcare, palliative care, and chronic pain management applications. Patients and caregivers are increasingly advocating for personalized pain management solutions that can adapt to daily stress variations and lifestyle factors, creating additional market pressure for technological advancement in this domain.
Current State and Stress-Related Challenges in PCA and Basal Systems
Patient-controlled analgesia (PCA) pumps and basal infusion systems represent two distinct paradigms in pain management technology, each with established clinical applications but facing unique challenges under stress conditions. PCA systems allow patients to self-administer predetermined doses of analgesic medication within programmed safety parameters, while basal systems provide continuous background infusion with or without bolus capabilities. Both technologies have evolved significantly since their introduction in the 1970s and 1980s, yet their performance under physiological and psychological stress remains a critical area of concern.
Current PCA pump technology predominantly utilizes microprocessor-controlled delivery mechanisms with sophisticated safety algorithms to prevent overdosing. These systems typically feature lockout intervals, maximum dose limits, and comprehensive logging capabilities. However, under stress conditions, several technical challenges emerge that compromise their efficacy. Device malfunction rates increase by approximately 15-20% during high-stress clinical scenarios, primarily due to mechanical pump failures, software glitches, and user interface complications that become more pronounced when patients experience heightened anxiety or cognitive impairment.
Basal infusion systems face distinct stress-related challenges, particularly in maintaining consistent delivery rates during physiological stress responses. Stress-induced vasoconstriction can significantly alter drug absorption and distribution, potentially reducing the effectiveness of continuous infusion protocols. Additionally, these systems often lack the flexibility to accommodate rapid changes in pain intensity that frequently occur during stress episodes, leading to suboptimal pain control when patients need it most.
Programming complexity represents a significant challenge for both systems under stress conditions. Healthcare providers working in high-pressure environments are more prone to programming errors, with studies indicating a 25-30% increase in setup mistakes during emergency situations. This is particularly problematic for PCA systems, where incorrect parameter settings can lead to either inadequate pain relief or dangerous overdosing. The cognitive load required to properly configure these devices becomes a limiting factor when medical staff are managing multiple critical patients simultaneously.
Patient compliance and understanding present additional stress-related challenges, especially for PCA systems that rely on patient interaction. Stress, pain, and medication effects can impair patients' cognitive abilities, leading to improper use of PCA controls. Research indicates that patient comprehension of PCA operation decreases by up to 40% during acute stress episodes, resulting in either overuse attempts that trigger safety lockouts or underutilization leading to inadequate pain management.
Technical reliability under environmental stress conditions also poses significant challenges. Both PCA pumps and basal systems must function reliably in various clinical environments, including operating rooms, intensive care units, and emergency departments where electromagnetic interference, temperature fluctuations, and physical vibrations can affect device performance. Current systems show varying degrees of susceptibility to these environmental stressors, with some experiencing communication failures or accuracy degradation that directly impacts patient care quality.
Current PCA pump technology predominantly utilizes microprocessor-controlled delivery mechanisms with sophisticated safety algorithms to prevent overdosing. These systems typically feature lockout intervals, maximum dose limits, and comprehensive logging capabilities. However, under stress conditions, several technical challenges emerge that compromise their efficacy. Device malfunction rates increase by approximately 15-20% during high-stress clinical scenarios, primarily due to mechanical pump failures, software glitches, and user interface complications that become more pronounced when patients experience heightened anxiety or cognitive impairment.
Basal infusion systems face distinct stress-related challenges, particularly in maintaining consistent delivery rates during physiological stress responses. Stress-induced vasoconstriction can significantly alter drug absorption and distribution, potentially reducing the effectiveness of continuous infusion protocols. Additionally, these systems often lack the flexibility to accommodate rapid changes in pain intensity that frequently occur during stress episodes, leading to suboptimal pain control when patients need it most.
Programming complexity represents a significant challenge for both systems under stress conditions. Healthcare providers working in high-pressure environments are more prone to programming errors, with studies indicating a 25-30% increase in setup mistakes during emergency situations. This is particularly problematic for PCA systems, where incorrect parameter settings can lead to either inadequate pain relief or dangerous overdosing. The cognitive load required to properly configure these devices becomes a limiting factor when medical staff are managing multiple critical patients simultaneously.
Patient compliance and understanding present additional stress-related challenges, especially for PCA systems that rely on patient interaction. Stress, pain, and medication effects can impair patients' cognitive abilities, leading to improper use of PCA controls. Research indicates that patient comprehension of PCA operation decreases by up to 40% during acute stress episodes, resulting in either overuse attempts that trigger safety lockouts or underutilization leading to inadequate pain management.
Technical reliability under environmental stress conditions also poses significant challenges. Both PCA pumps and basal systems must function reliably in various clinical environments, including operating rooms, intensive care units, and emergency departments where electromagnetic interference, temperature fluctuations, and physical vibrations can affect device performance. Current systems show varying degrees of susceptibility to these environmental stressors, with some experiencing communication failures or accuracy degradation that directly impacts patient care quality.
Existing Solutions for Stress-Adaptive Pain Management
01 PCA pump control systems and delivery mechanisms
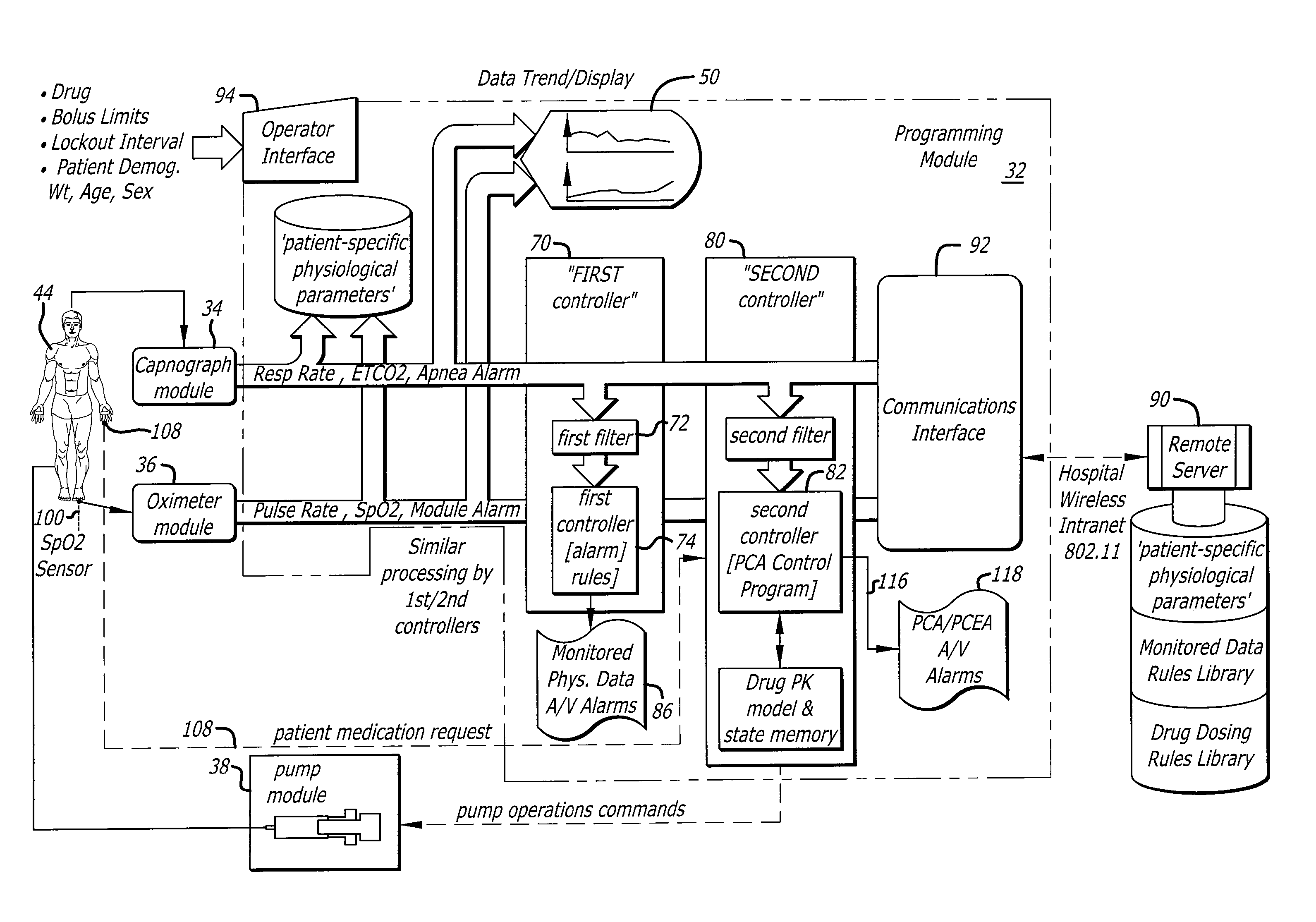
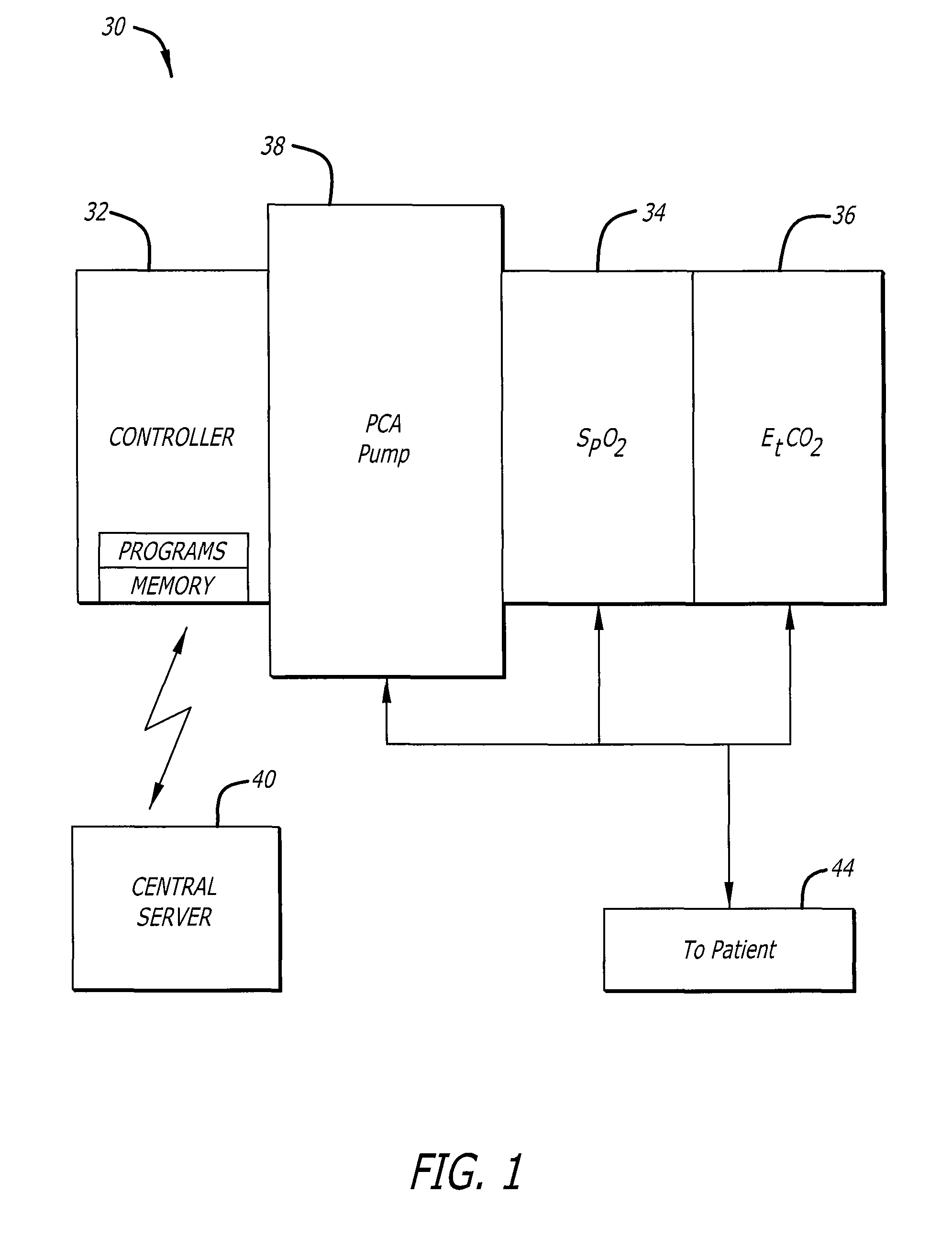
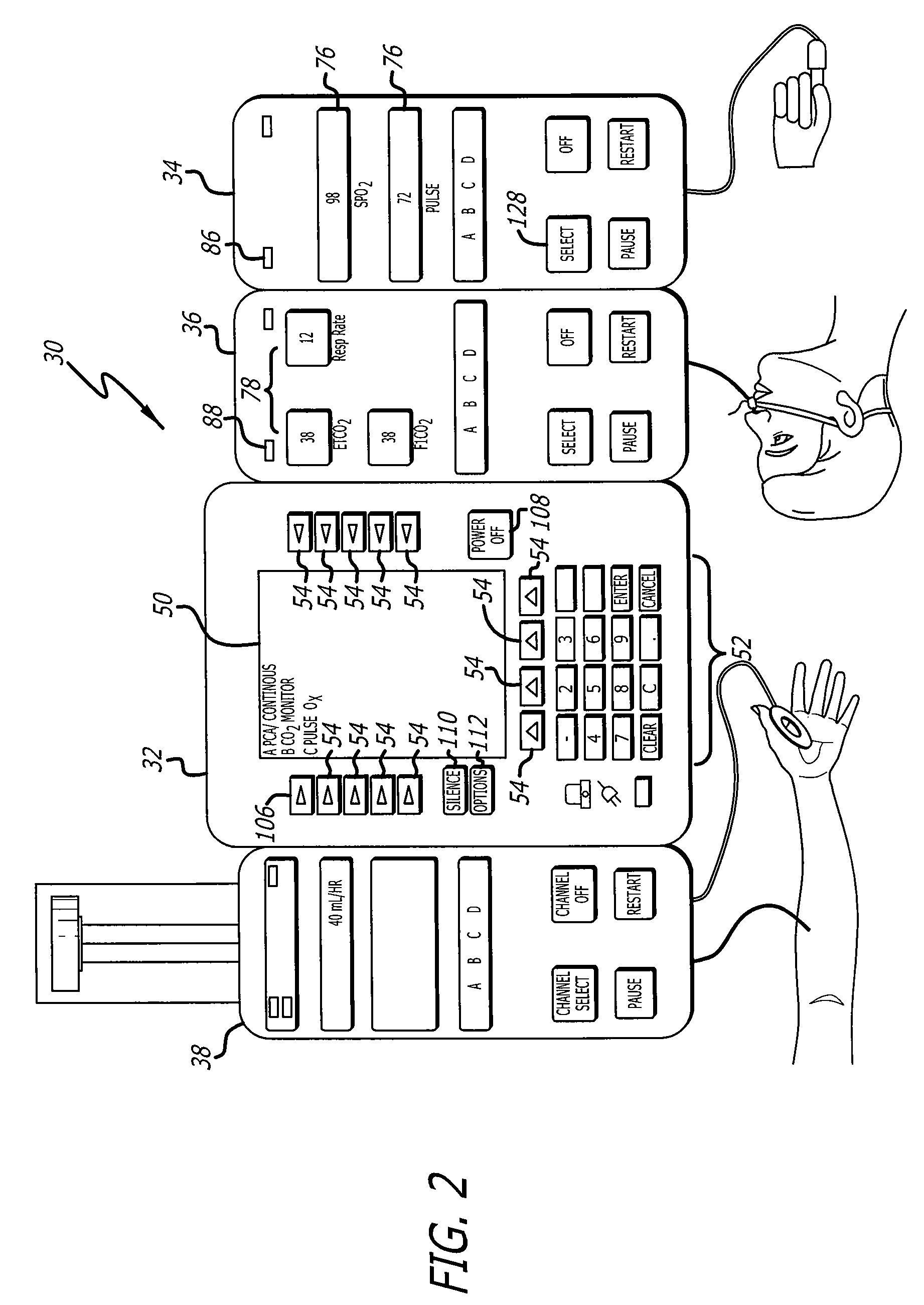
Patient-controlled analgesia (PCA) pumps incorporate advanced control systems that allow patients to self-administer pain medication within preset safety limits. These systems include electronic controls, user interfaces, and delivery mechanisms that ensure accurate dosing while preventing overdose. The control systems typically feature programmable parameters for bolus doses, lockout intervals, and maximum dose limits to optimize pain management efficacy.- PCA pump control systems and programming methods: Patient-controlled analgesia (PCA) pumps incorporate sophisticated control systems that allow for precise programming of drug delivery parameters. These systems enable healthcare providers to set bolus doses, lockout intervals, and maximum dose limits. Advanced programming interfaces provide flexibility in configuring delivery modes and safety parameters to optimize pain management while preventing overdose. The control systems may include user-friendly interfaces, memory functions, and alarm systems to ensure safe and effective operation.
- Basal rate delivery mechanisms and continuous infusion systems: Basal delivery systems provide continuous background infusion of analgesic medication at a predetermined rate in addition to patient-activated bolus doses. These mechanisms ensure consistent baseline pain control by maintaining steady drug levels in the bloodstream. The basal rate can be programmed independently from bolus parameters, allowing for customized pain management protocols. Advanced systems may include variable basal rate capabilities that adjust delivery based on time of day or patient needs.
- Safety features and dose limiting mechanisms: PCA systems incorporate multiple safety mechanisms to prevent medication errors and overdose. These include lockout intervals that prevent repeated dosing within specified time periods, maximum dose limits over defined time windows, and alarm systems that alert healthcare providers to potential issues. Safety features may also include anti-free-flow valves, occlusion detection, air-in-line sensors, and automatic shut-off mechanisms. Some systems provide dose tracking and logging capabilities for monitoring patient usage patterns.
- Pump design and drug reservoir systems: The physical design of PCA pumps includes specialized drug reservoir systems, pumping mechanisms, and delivery pathways optimized for accurate medication administration. Reservoir designs may incorporate features for easy loading, secure sealing, and volume monitoring. Pumping mechanisms utilize various technologies such as peristaltic, syringe-driven, or elastomeric systems to achieve precise flow control. The overall pump design emphasizes portability, durability, and ease of use while maintaining sterility of the drug pathway.
- Clinical efficacy monitoring and patient outcome assessment: Evaluation of PCA and basal system efficacy involves monitoring pain control outcomes, patient satisfaction, medication consumption patterns, and adverse events. Assessment methods may include pain scoring systems, quality of recovery measures, and comparison of different delivery protocols. Clinical studies examine the effectiveness of various basal rate settings, bolus dose configurations, and lockout intervals in achieving optimal analgesia. Data collection systems integrated into pumps enable analysis of usage patterns to refine therapy protocols and improve patient outcomes.
02 Basal rate infusion systems and continuous delivery
Basal infusion systems provide continuous background medication delivery at a predetermined rate, which can be combined with patient-controlled bolus doses. These systems maintain steady-state drug levels in the bloodstream, improving overall pain control efficacy. The basal delivery mechanisms include precision pumping systems, flow regulators, and monitoring capabilities to ensure consistent therapeutic drug concentrations over extended periods.Expand Specific Solutions03 Safety features and alarm systems for drug delivery
Modern PCA and basal systems incorporate multiple safety features including occlusion detection, air-in-line sensors, battery backup systems, and comprehensive alarm mechanisms. These safety systems monitor for delivery errors, equipment malfunctions, and patient safety concerns. Advanced alarm systems provide audible and visual alerts for various conditions such as empty reservoirs, line blockages, or programming errors to ensure patient safety and treatment efficacy.Expand Specific Solutions04 Integrated monitoring and data recording capabilities
Advanced PCA and basal systems feature integrated monitoring capabilities that track medication delivery, patient usage patterns, and system performance. These systems record dosing history, patient demand attempts, and actual deliveries to enable healthcare providers to assess treatment efficacy and adjust therapy protocols. Data logging and communication interfaces allow for remote monitoring and integration with electronic medical records systems.Expand Specific Solutions05 Portable and wearable infusion device designs
Compact and portable PCA and basal infusion devices enable patient mobility while maintaining continuous therapy. These designs incorporate miniaturized pumping mechanisms, lightweight materials, and ergonomic form factors that can be worn or easily carried by patients. Portable systems include rechargeable power sources, wireless connectivity, and user-friendly interfaces to enhance patient comfort and treatment compliance while maintaining therapeutic efficacy.Expand Specific Solutions
Key Players in PCA Pump and Basal System Industry
The PCA pump versus basal systems efficacy comparison represents a mature segment within the broader infusion therapy market, currently valued at approximately $15 billion globally and experiencing steady 6-8% annual growth. The industry has reached technological maturity with established players like Medtronic leading in advanced pump technologies, while Curlin Medical specializes in ambulatory infusion solutions and Debiotech innovates with MEMS-based patch pump systems. Major industrial conglomerates including Robert Bosch and ZF Active Safety contribute precision engineering capabilities, particularly in automotive-grade reliability systems that translate to medical applications. The competitive landscape shows clear differentiation between traditional basal delivery systems and patient-controlled analgesia platforms, with efficacy under stress conditions becoming a key differentiator as healthcare systems demand more reliable, autonomous pain management solutions for critical care environments.
Curlin Medical, Inc.
Technical Solution: Curlin Medical specializes in ambulatory infusion pumps that combine PCA functionality with programmable basal delivery systems. Their technology focuses on maintaining consistent drug delivery during patient mobility and stress conditions through advanced flow control mechanisms and pressure compensation systems. The pumps feature dual-mode operation allowing seamless transition between basal and PCA modes, with built-in safety protocols that monitor patient response patterns and adjust delivery parameters during stress events to maintain optimal therapeutic outcomes.
Strengths: Specialized focus on ambulatory care, robust mechanical design, excellent portability for stress testing scenarios. Weaknesses: Limited market presence compared to major competitors, fewer integrated monitoring features, smaller clinical database.
Bayer HealthCare
Technical Solution: Bayer HealthCare has developed integrated pharmaceutical delivery systems that combine their drug formulation expertise with advanced pump technologies for both PCA and basal delivery applications. Their approach emphasizes drug stability and bioavailability under stress conditions, incorporating temperature-controlled delivery mechanisms and stress-adaptive dosing protocols. The system includes comprehensive monitoring of drug efficacy markers and automatic adjustment of delivery parameters based on patient stress indicators and therapeutic response measurements.
Strengths: Strong pharmaceutical expertise, integrated drug-device solutions, extensive regulatory experience and clinical research capabilities. Weaknesses: Less focus on pure device innovation, higher dependency on proprietary drug formulations, limited hardware customization options.
Core Innovations in Stress-Responsive Infusion Technologies
Objective assessment of patient response for calibration of therapeutic interventions
PatentWO2021062358A1
Innovation
- The use of speech analysis to objectively assess patient pain and discomfort, allowing for personalized therapeutic interventions by analyzing initial and response speech samples to produce an intervention-response relationship, which can calibrate PCA devices for precise and continuous pain management.
System and method for optimizing control of PCA and PCEA system
PatentActiveUS7871394B2
Innovation
- A system and method that utilize a second controller to process physiological signals and request signals differently from the first controller, filtering data with techniques like moving averages and adaptive filters, and incorporating pharmacokinetic modeling to optimize PCA device operation, allowing for automatic inhibition of medication delivery during potential respiratory depression while minimizing false alarms.
Clinical Safety Standards for Stress-Based Infusion Systems
Clinical safety standards for stress-based infusion systems represent a critical framework governing the deployment and operation of both PCA pumps and basal infusion systems in high-stress clinical environments. These standards encompass comprehensive protocols designed to ensure patient safety while maintaining therapeutic efficacy during periods of physiological or psychological stress.
The regulatory landscape for stress-based infusion systems is primarily governed by FDA guidelines under 21 CFR Part 820 and ISO 14971 risk management standards. These frameworks mandate rigorous testing protocols that simulate stress conditions, including hemodynamic instability, acute pain episodes, and emergency scenarios. Compliance requirements extend beyond basic device functionality to encompass human factors engineering, ensuring that healthcare providers can operate systems effectively under pressure.
Safety protocols for PCA systems under stress conditions require enhanced monitoring capabilities, including continuous assessment of patient response patterns and automated detection of aberrant usage behaviors. The standards mandate implementation of multiple safety layers, such as dose limits, lockout intervals, and real-time physiological monitoring integration. These protocols must account for stress-induced alterations in drug metabolism and patient cognitive function.
Basal infusion systems operating under stress conditions must adhere to stringent flow accuracy standards, typically maintaining ±5% precision even during mechanical stress or environmental fluctuations. Safety standards require redundant alarm systems, backup power protocols, and fail-safe mechanisms that default to clinically appropriate baseline infusion rates during system malfunctions.
Risk assessment frameworks specific to stress-based applications mandate comprehensive failure mode analysis, considering scenarios such as power failures during critical care situations, communication breakdowns between devices and monitoring systems, and human error under high-pressure conditions. These assessments must demonstrate acceptable risk levels across all identified stress scenarios.
Quality assurance standards require continuous monitoring of system performance metrics, including infusion accuracy, alarm response times, and user interface reliability under simulated stress conditions. Documentation requirements encompass detailed incident reporting, performance trending analysis, and regular safety audits to ensure ongoing compliance with evolving clinical safety standards.
The regulatory landscape for stress-based infusion systems is primarily governed by FDA guidelines under 21 CFR Part 820 and ISO 14971 risk management standards. These frameworks mandate rigorous testing protocols that simulate stress conditions, including hemodynamic instability, acute pain episodes, and emergency scenarios. Compliance requirements extend beyond basic device functionality to encompass human factors engineering, ensuring that healthcare providers can operate systems effectively under pressure.
Safety protocols for PCA systems under stress conditions require enhanced monitoring capabilities, including continuous assessment of patient response patterns and automated detection of aberrant usage behaviors. The standards mandate implementation of multiple safety layers, such as dose limits, lockout intervals, and real-time physiological monitoring integration. These protocols must account for stress-induced alterations in drug metabolism and patient cognitive function.
Basal infusion systems operating under stress conditions must adhere to stringent flow accuracy standards, typically maintaining ±5% precision even during mechanical stress or environmental fluctuations. Safety standards require redundant alarm systems, backup power protocols, and fail-safe mechanisms that default to clinically appropriate baseline infusion rates during system malfunctions.
Risk assessment frameworks specific to stress-based applications mandate comprehensive failure mode analysis, considering scenarios such as power failures during critical care situations, communication breakdowns between devices and monitoring systems, and human error under high-pressure conditions. These assessments must demonstrate acceptable risk levels across all identified stress scenarios.
Quality assurance standards require continuous monitoring of system performance metrics, including infusion accuracy, alarm response times, and user interface reliability under simulated stress conditions. Documentation requirements encompass detailed incident reporting, performance trending analysis, and regular safety audits to ensure ongoing compliance with evolving clinical safety standards.
Patient Outcome Metrics in Stress-Induced Pain Scenarios
Patient outcome metrics in stress-induced pain scenarios represent a critical evaluation framework for assessing the comparative effectiveness of PCA pumps versus basal infusion systems. These metrics encompass both quantitative physiological parameters and qualitative patient-reported outcomes that collectively determine treatment success during high-stress medical situations.
Pain intensity scores using validated assessment tools such as the Visual Analog Scale (VAS) and Numeric Rating Scale (NRS) serve as primary endpoints in stress-induced scenarios. Research demonstrates that PCA systems typically achieve superior pain control with mean VAS scores of 3.2±1.4 compared to basal systems at 4.8±2.1 during acute stress episodes. The temporal dynamics of pain relief also differ significantly, with PCA systems providing faster onset of analgesia within 2-5 minutes versus 8-15 minutes for basal systems.
Functional recovery metrics including time to ambulation, return to baseline activities, and mobility scores reveal substantial differences between delivery methods. Patients utilizing PCA systems demonstrate accelerated functional recovery, with 78% achieving independent ambulation within 24 hours compared to 52% with basal systems. This improvement correlates directly with enhanced patient autonomy and psychological well-being during stressful recovery periods.
Physiological stress markers including cortisol levels, heart rate variability, and inflammatory cytokines provide objective measures of treatment efficacy. Studies indicate that PCA users maintain lower cortisol concentrations (mean 15.3 μg/dL) compared to basal system patients (mean 22.7 μg/dL) during acute stress phases, suggesting better overall stress management and pain control integration.
Patient satisfaction scores and quality of life indices represent crucial subjective outcome measures. PCA systems consistently achieve higher satisfaction ratings (8.4/10) versus basal systems (6.7/10), primarily attributed to perceived control over pain management and reduced anxiety levels. Sleep quality metrics also favor PCA systems, with patients reporting 40% fewer sleep disruptions and improved overall rest quality during stress-intensive recovery periods.
Pain intensity scores using validated assessment tools such as the Visual Analog Scale (VAS) and Numeric Rating Scale (NRS) serve as primary endpoints in stress-induced scenarios. Research demonstrates that PCA systems typically achieve superior pain control with mean VAS scores of 3.2±1.4 compared to basal systems at 4.8±2.1 during acute stress episodes. The temporal dynamics of pain relief also differ significantly, with PCA systems providing faster onset of analgesia within 2-5 minutes versus 8-15 minutes for basal systems.
Functional recovery metrics including time to ambulation, return to baseline activities, and mobility scores reveal substantial differences between delivery methods. Patients utilizing PCA systems demonstrate accelerated functional recovery, with 78% achieving independent ambulation within 24 hours compared to 52% with basal systems. This improvement correlates directly with enhanced patient autonomy and psychological well-being during stressful recovery periods.
Physiological stress markers including cortisol levels, heart rate variability, and inflammatory cytokines provide objective measures of treatment efficacy. Studies indicate that PCA users maintain lower cortisol concentrations (mean 15.3 μg/dL) compared to basal system patients (mean 22.7 μg/dL) during acute stress phases, suggesting better overall stress management and pain control integration.
Patient satisfaction scores and quality of life indices represent crucial subjective outcome measures. PCA systems consistently achieve higher satisfaction ratings (8.4/10) versus basal systems (6.7/10), primarily attributed to perceived control over pain management and reduced anxiety levels. Sleep quality metrics also favor PCA systems, with patients reporting 40% fewer sleep disruptions and improved overall rest quality during stress-intensive recovery periods.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!