Peroxide Role in Efficient Energy Management Systems
MAR 9, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Peroxide Energy Systems Background and Objectives
Peroxide-based energy systems have emerged as a critical component in the evolution of modern energy management technologies, representing a significant departure from traditional electrochemical storage and conversion methods. The historical development of peroxide chemistry in energy applications traces back to early fuel cell research in the 1960s, where hydrogen peroxide served as both an oxidant and energy carrier. Over subsequent decades, the field has expanded to encompass diverse applications including grid-scale energy storage, portable power systems, and renewable energy integration solutions.
The fundamental appeal of peroxide systems lies in their unique electrochemical properties, offering high energy density, rapid charge-discharge capabilities, and environmental compatibility. Unlike conventional battery technologies that rely on heavy metals or toxic materials, peroxide-based systems utilize abundant and relatively benign chemical compounds. This characteristic has positioned them as attractive alternatives for sustainable energy infrastructure development, particularly in applications requiring high power output and extended operational lifespans.
Contemporary research has identified several key evolutionary trends driving peroxide energy system development. The integration of advanced catalytic materials has significantly improved reaction kinetics and energy conversion efficiency. Simultaneously, innovations in system architecture have enabled better thermal management and enhanced safety protocols. These developments have collectively expanded the operational envelope of peroxide systems, making them viable for increasingly demanding applications.
The primary technical objectives for peroxide energy systems center on achieving optimal balance between energy density, power output, and system longevity. Current research initiatives focus on developing advanced electrode materials that can withstand the corrosive nature of peroxide environments while maintaining high catalytic activity. Additionally, significant effort is directed toward optimizing electrolyte compositions to minimize parasitic reactions and improve overall system efficiency.
System-level objectives encompass the development of intelligent control algorithms capable of managing complex peroxide chemistry in real-time operational scenarios. This includes precise monitoring of peroxide concentration, temperature regulation, and dynamic load balancing to maximize energy utilization efficiency. Furthermore, integration with existing energy infrastructure requires standardized interfaces and communication protocols that enable seamless operation within broader energy management networks.
The strategic vision for peroxide energy systems extends beyond immediate technical improvements to encompass transformative applications in renewable energy storage and grid stabilization. Long-term objectives include achieving cost parity with established energy storage technologies while delivering superior performance characteristics in terms of response time, cycle life, and environmental impact.
The fundamental appeal of peroxide systems lies in their unique electrochemical properties, offering high energy density, rapid charge-discharge capabilities, and environmental compatibility. Unlike conventional battery technologies that rely on heavy metals or toxic materials, peroxide-based systems utilize abundant and relatively benign chemical compounds. This characteristic has positioned them as attractive alternatives for sustainable energy infrastructure development, particularly in applications requiring high power output and extended operational lifespans.
Contemporary research has identified several key evolutionary trends driving peroxide energy system development. The integration of advanced catalytic materials has significantly improved reaction kinetics and energy conversion efficiency. Simultaneously, innovations in system architecture have enabled better thermal management and enhanced safety protocols. These developments have collectively expanded the operational envelope of peroxide systems, making them viable for increasingly demanding applications.
The primary technical objectives for peroxide energy systems center on achieving optimal balance between energy density, power output, and system longevity. Current research initiatives focus on developing advanced electrode materials that can withstand the corrosive nature of peroxide environments while maintaining high catalytic activity. Additionally, significant effort is directed toward optimizing electrolyte compositions to minimize parasitic reactions and improve overall system efficiency.
System-level objectives encompass the development of intelligent control algorithms capable of managing complex peroxide chemistry in real-time operational scenarios. This includes precise monitoring of peroxide concentration, temperature regulation, and dynamic load balancing to maximize energy utilization efficiency. Furthermore, integration with existing energy infrastructure requires standardized interfaces and communication protocols that enable seamless operation within broader energy management networks.
The strategic vision for peroxide energy systems extends beyond immediate technical improvements to encompass transformative applications in renewable energy storage and grid stabilization. Long-term objectives include achieving cost parity with established energy storage technologies while delivering superior performance characteristics in terms of response time, cycle life, and environmental impact.
Market Demand for Peroxide-Based Energy Solutions
The global energy storage market is experiencing unprecedented growth driven by the urgent need for renewable energy integration and grid stabilization solutions. Peroxide-based energy systems are emerging as a compelling alternative to traditional lithium-ion batteries, particularly in large-scale stationary storage applications where safety, longevity, and environmental sustainability are paramount concerns.
Industrial sectors are increasingly seeking energy storage solutions that can operate safely in extreme conditions while maintaining high efficiency over extended periods. Peroxide-based systems address critical pain points in current energy storage technologies, including thermal runaway risks, limited cycle life, and resource scarcity issues associated with rare earth materials. Manufacturing facilities, data centers, and critical infrastructure operators are particularly interested in these systems due to their inherent safety characteristics and stable performance profiles.
The renewable energy sector represents the most significant demand driver for peroxide-based energy solutions. Solar and wind power installations require robust energy storage systems capable of handling frequent charge-discharge cycles while maintaining grid stability. Peroxide systems offer superior cycle stability and can operate effectively across wider temperature ranges compared to conventional battery technologies, making them ideal for utility-scale renewable energy projects.
Grid modernization initiatives worldwide are creating substantial market opportunities for advanced energy storage technologies. Utilities are actively seeking solutions that can provide both energy storage and grid services such as frequency regulation and voltage support. Peroxide-based systems demonstrate excellent response characteristics for these applications, positioning them as valuable assets in smart grid implementations.
The transportation electrification trend is generating additional demand for safer, more sustainable energy storage solutions. While current applications focus primarily on stationary systems, ongoing research into higher energy density peroxide formulations suggests potential expansion into electric vehicle markets, particularly for commercial and industrial vehicle segments where safety and operational reliability are critical requirements.
Emerging markets in developing countries present significant growth opportunities as these regions invest in modern energy infrastructure. The inherent safety and environmental advantages of peroxide-based systems make them particularly attractive for regions with limited technical maintenance capabilities and stringent environmental regulations.
Industrial sectors are increasingly seeking energy storage solutions that can operate safely in extreme conditions while maintaining high efficiency over extended periods. Peroxide-based systems address critical pain points in current energy storage technologies, including thermal runaway risks, limited cycle life, and resource scarcity issues associated with rare earth materials. Manufacturing facilities, data centers, and critical infrastructure operators are particularly interested in these systems due to their inherent safety characteristics and stable performance profiles.
The renewable energy sector represents the most significant demand driver for peroxide-based energy solutions. Solar and wind power installations require robust energy storage systems capable of handling frequent charge-discharge cycles while maintaining grid stability. Peroxide systems offer superior cycle stability and can operate effectively across wider temperature ranges compared to conventional battery technologies, making them ideal for utility-scale renewable energy projects.
Grid modernization initiatives worldwide are creating substantial market opportunities for advanced energy storage technologies. Utilities are actively seeking solutions that can provide both energy storage and grid services such as frequency regulation and voltage support. Peroxide-based systems demonstrate excellent response characteristics for these applications, positioning them as valuable assets in smart grid implementations.
The transportation electrification trend is generating additional demand for safer, more sustainable energy storage solutions. While current applications focus primarily on stationary systems, ongoing research into higher energy density peroxide formulations suggests potential expansion into electric vehicle markets, particularly for commercial and industrial vehicle segments where safety and operational reliability are critical requirements.
Emerging markets in developing countries present significant growth opportunities as these regions invest in modern energy infrastructure. The inherent safety and environmental advantages of peroxide-based systems make them particularly attractive for regions with limited technical maintenance capabilities and stringent environmental regulations.
Current State of Peroxide Energy Management Technologies
Peroxide-based energy management technologies have emerged as a significant component in modern energy storage and conversion systems, with hydrogen peroxide (H2O2) leading the development due to its high energy density and environmental compatibility. Current implementations primarily focus on fuel cell applications, where peroxide serves as both an oxidant and energy carrier, offering theoretical energy densities of up to 2.7 kWh/kg, substantially higher than conventional lithium-ion batteries.
The technology landscape is dominated by direct peroxide fuel cells (DPFCs), which have achieved power densities ranging from 50-150 mW/cm² under optimal operating conditions. These systems utilize platinum-based catalysts at the cathode and various anode materials including carbon-supported metals. Recent advances have demonstrated stable operation for over 1000 hours in laboratory settings, though commercial viability remains limited by catalyst degradation and peroxide decomposition issues.
Industrial applications currently span aerospace propulsion systems, where concentrated peroxide serves as a monopropellant, and stationary energy storage installations for grid stabilization. The aerospace sector has successfully deployed peroxide-based systems in satellite attitude control and rocket propulsion, leveraging the technology's rapid response characteristics and high specific impulse capabilities.
Manufacturing processes for peroxide energy systems have matured significantly, with established production methods for high-purity peroxide solutions exceeding 90% concentration. Key technological barriers include catalyst poisoning from peroxide decomposition products, membrane degradation in fuel cell configurations, and thermal management challenges during high-rate discharge operations.
Current research initiatives focus on developing non-platinum catalysts to reduce system costs, with promising results from transition metal oxides and carbon-based materials. Advanced membrane technologies, including perfluorinated and hydrocarbon-based polymers, are being optimized for peroxide compatibility while maintaining proton conductivity.
Safety protocols and handling procedures have been standardized across the industry, addressing peroxide's inherent reactivity through specialized storage systems, concentration monitoring, and automated safety shutoffs. These developments have enabled broader adoption in controlled industrial environments while maintaining operational safety standards.
The integration of peroxide systems with renewable energy sources represents an emerging application area, where excess solar or wind power drives peroxide synthesis through electrochemical processes, creating a closed-loop energy storage solution with minimal environmental impact.
The technology landscape is dominated by direct peroxide fuel cells (DPFCs), which have achieved power densities ranging from 50-150 mW/cm² under optimal operating conditions. These systems utilize platinum-based catalysts at the cathode and various anode materials including carbon-supported metals. Recent advances have demonstrated stable operation for over 1000 hours in laboratory settings, though commercial viability remains limited by catalyst degradation and peroxide decomposition issues.
Industrial applications currently span aerospace propulsion systems, where concentrated peroxide serves as a monopropellant, and stationary energy storage installations for grid stabilization. The aerospace sector has successfully deployed peroxide-based systems in satellite attitude control and rocket propulsion, leveraging the technology's rapid response characteristics and high specific impulse capabilities.
Manufacturing processes for peroxide energy systems have matured significantly, with established production methods for high-purity peroxide solutions exceeding 90% concentration. Key technological barriers include catalyst poisoning from peroxide decomposition products, membrane degradation in fuel cell configurations, and thermal management challenges during high-rate discharge operations.
Current research initiatives focus on developing non-platinum catalysts to reduce system costs, with promising results from transition metal oxides and carbon-based materials. Advanced membrane technologies, including perfluorinated and hydrocarbon-based polymers, are being optimized for peroxide compatibility while maintaining proton conductivity.
Safety protocols and handling procedures have been standardized across the industry, addressing peroxide's inherent reactivity through specialized storage systems, concentration monitoring, and automated safety shutoffs. These developments have enabled broader adoption in controlled industrial environments while maintaining operational safety standards.
The integration of peroxide systems with renewable energy sources represents an emerging application area, where excess solar or wind power drives peroxide synthesis through electrochemical processes, creating a closed-loop energy storage solution with minimal environmental impact.
Existing Peroxide Energy Management Solutions
01 Peroxide-based energy storage systems
Energy storage systems utilizing peroxide compounds as active materials for electrochemical energy storage. These systems focus on improving energy density and cycle life through optimized peroxide formulations and electrode designs. The technology enables efficient charge-discharge cycles with enhanced stability and performance characteristics for various energy storage applications.- Peroxide-based energy storage systems: Energy storage systems utilizing peroxide compounds as active materials for batteries or fuel cells. These systems focus on improving energy density and conversion efficiency through optimized peroxide formulations and electrode designs. The technology enables better charge-discharge cycles and enhanced overall energy management performance.
- Thermal management in peroxide energy systems: Methods and apparatus for controlling temperature in peroxide-based energy systems to improve efficiency and safety. These approaches include heat dissipation structures, cooling systems, and thermal monitoring mechanisms that prevent overheating and maintain optimal operating conditions. Proper thermal management extends system lifespan and enhances energy conversion efficiency.
- Power management circuits for peroxide systems: Electronic control systems and circuits designed to optimize power distribution and energy flow in peroxide-based energy devices. These circuits regulate charging and discharging processes, balance power loads, and implement intelligent energy management algorithms. The technology improves overall system efficiency and prevents energy waste.
- Peroxide catalyst optimization for energy efficiency: Development of catalytic materials and compositions that enhance peroxide decomposition and energy release rates. These catalysts improve reaction kinetics and energy conversion efficiency while reducing activation energy requirements. The optimization focuses on material selection, surface modification, and nanostructure engineering to maximize performance.
- Monitoring and control systems for peroxide energy management: Integrated monitoring and control technologies that track performance parameters and optimize operational efficiency in peroxide energy systems. These systems employ sensors, data analytics, and feedback control mechanisms to maintain optimal conditions and predict maintenance needs. Real-time monitoring enables proactive adjustments to maximize energy efficiency and system reliability.
02 Thermal management in peroxide energy systems
Methods and apparatus for managing heat generation and dissipation in peroxide-based energy systems. These approaches include cooling mechanisms, temperature monitoring systems, and thermal regulation strategies to maintain optimal operating conditions. The thermal management solutions prevent overheating and improve overall system efficiency and safety during energy conversion processes.Expand Specific Solutions03 Power conversion efficiency optimization
Techniques for enhancing power conversion efficiency in peroxide energy management systems through advanced control algorithms and circuit designs. These methods optimize energy transfer, minimize losses, and improve overall system performance. The optimization strategies include intelligent power management, adaptive control systems, and efficient energy routing mechanisms.Expand Specific Solutions04 Monitoring and control systems for peroxide energy
Integrated monitoring and control systems designed to track performance parameters and manage operational efficiency in peroxide-based energy systems. These systems employ sensors, data analytics, and feedback mechanisms to ensure optimal performance. The technology enables real-time monitoring of energy flow, system health diagnostics, and automated adjustments for maximum efficiency.Expand Specific Solutions05 Safety and stability enhancement mechanisms
Safety features and stability enhancement mechanisms specifically designed for peroxide energy management systems. These include protective circuits, fail-safe mechanisms, and stability control methods to prevent hazardous conditions. The implementations focus on preventing thermal runaway, managing pressure variations, and ensuring safe operation under various load conditions while maintaining high efficiency.Expand Specific Solutions
Key Players in Peroxide Energy Technology Industry
The peroxide role in efficient energy management systems represents an emerging technological domain currently in its early development stage, with significant growth potential driven by increasing demand for sustainable energy solutions. The market is experiencing nascent expansion as organizations recognize peroxide's applications in energy storage, fuel cells, and water treatment systems within energy infrastructure. Technology maturity varies considerably across different applications, with established players like Arkema France SA and Sumitomo Chemical Co., Ltd. providing chemical foundations, while specialized companies such as HPNow ApS and PolyPlus Battery Co., Inc. advance innovative peroxide-based solutions. Research institutions including Tsinghua University, KAIST, and University of Florida contribute fundamental research, supported by government entities like NASA and CNRS. The competitive landscape shows a convergence of chemical manufacturers, energy companies like CHN Energy Investment Group, and technology developers, indicating the sector's transition from research-focused to commercially viable applications with substantial market opportunities ahead.
Electric Power Research Institute, Inc.
Technical Solution: EPRI has pioneered the integration of peroxide-based systems in smart grid applications, developing protocols for using hydrogen peroxide in fuel cell auxiliary power systems and grid stabilization. Their research focuses on peroxide fuel cells that can provide backup power with rapid response times under 30 seconds. The technology includes advanced monitoring systems for peroxide degradation and automated replenishment mechanisms. EPRI's solution emphasizes grid integration capabilities and has demonstrated scalability from residential to utility-scale implementations with efficiency rates exceeding 85% in energy conversion processes.
Advantages: Strong grid integration expertise, extensive testing infrastructure, industry partnerships. Disadvantages: Limited commercial deployment, dependency on peroxide supply chain stability.
ENEOS Corp.
Technical Solution: ENEOS has developed peroxide-based energy storage solutions integrated with their petroleum refining operations, utilizing hydrogen peroxide as an energy buffer for process optimization. Their technology focuses on catalytic peroxide decomposition systems that can provide on-demand steam and power generation with response times under 60 seconds. The system incorporates advanced process control for safe peroxide handling and features heat recovery mechanisms that achieve overall efficiency rates of 78%. ENEOS's approach includes integration with existing industrial infrastructure and demonstrates cost-effective energy management through waste heat utilization and process synergies with refining operations.
Advantages: Industrial scale experience, existing infrastructure integration, cost-effective implementation. Disadvantages: Limited to industrial applications, dependency on refining operations for optimal performance.
Core Peroxide Chemistry Innovations for Energy Systems
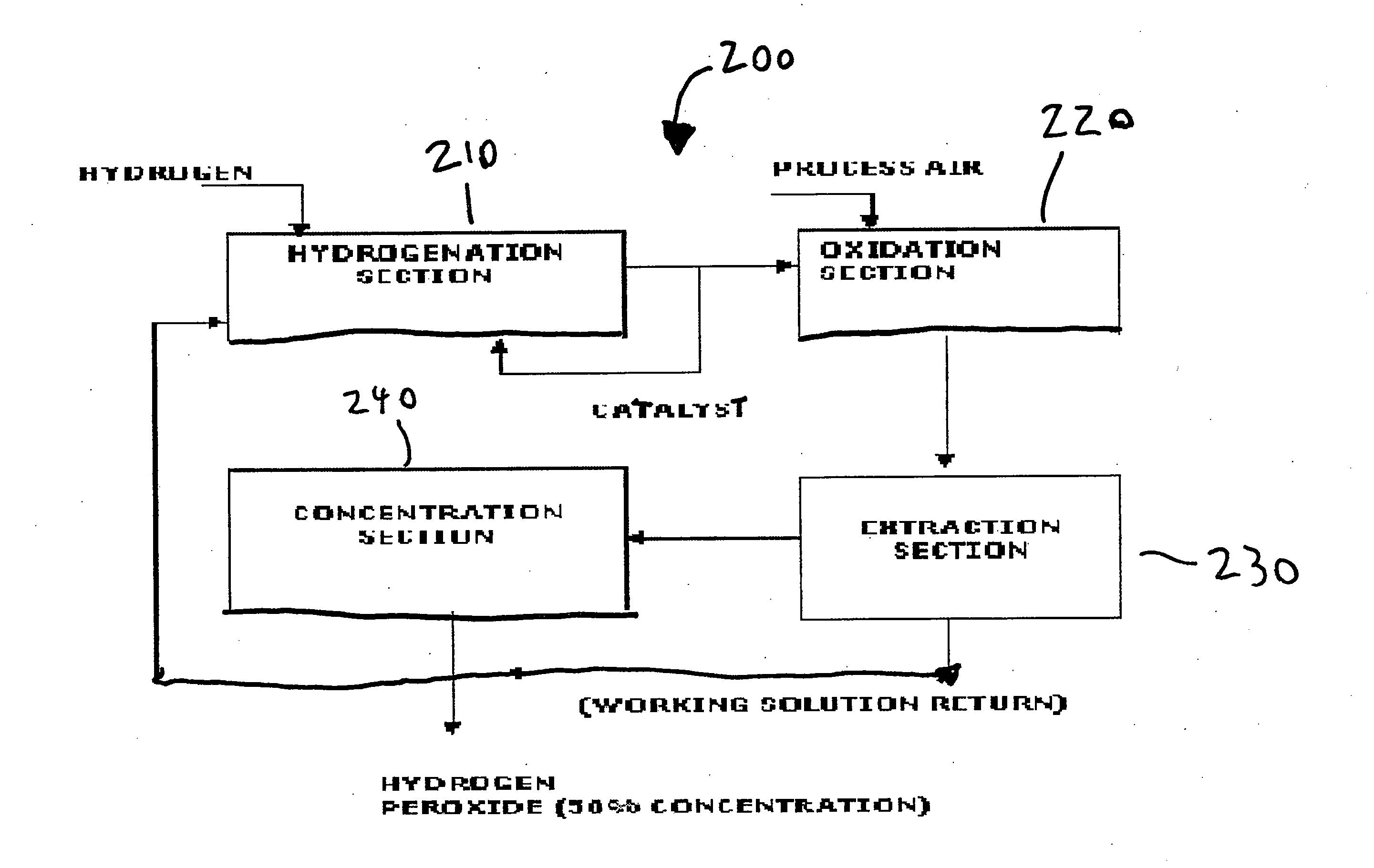
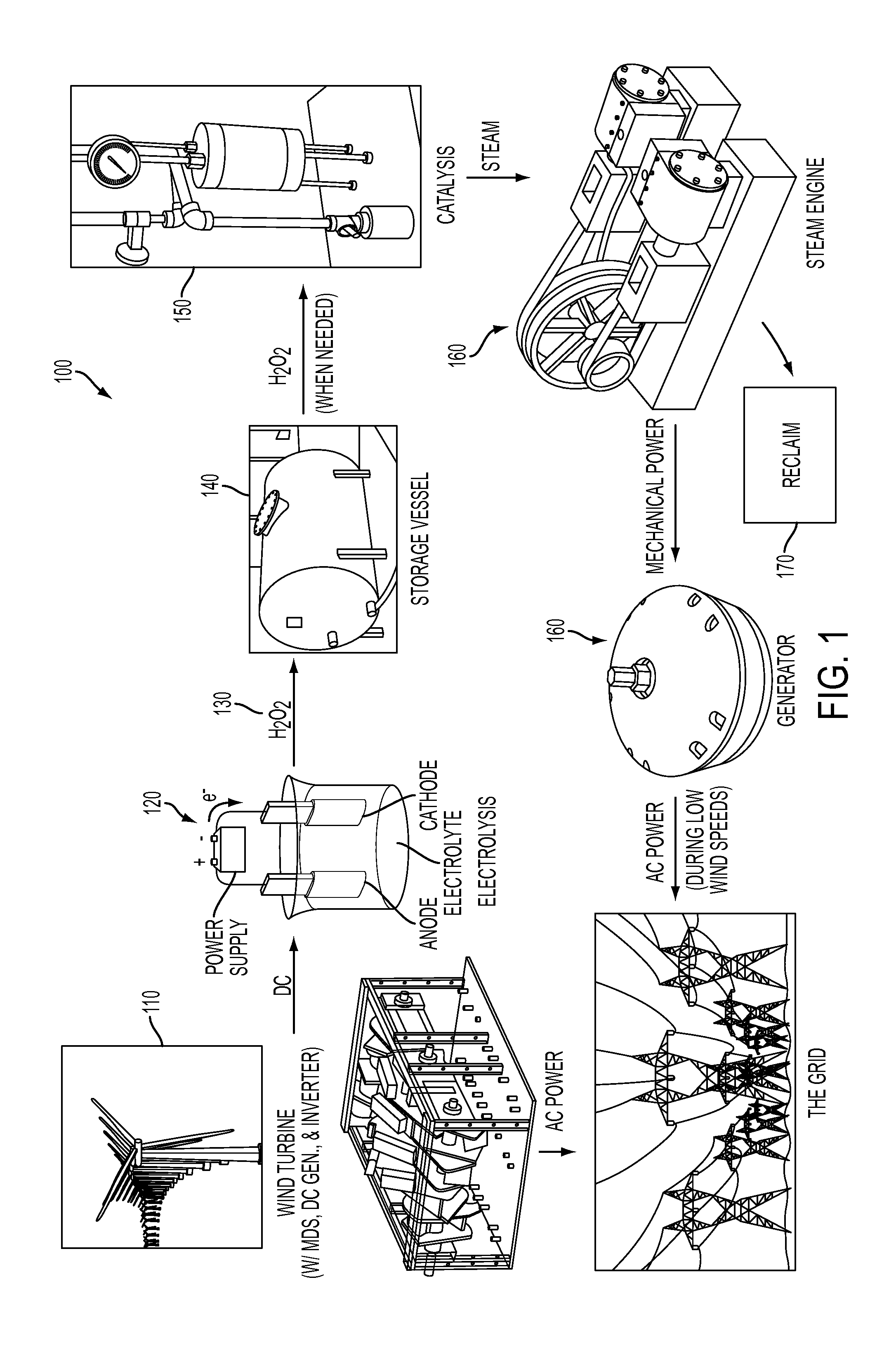
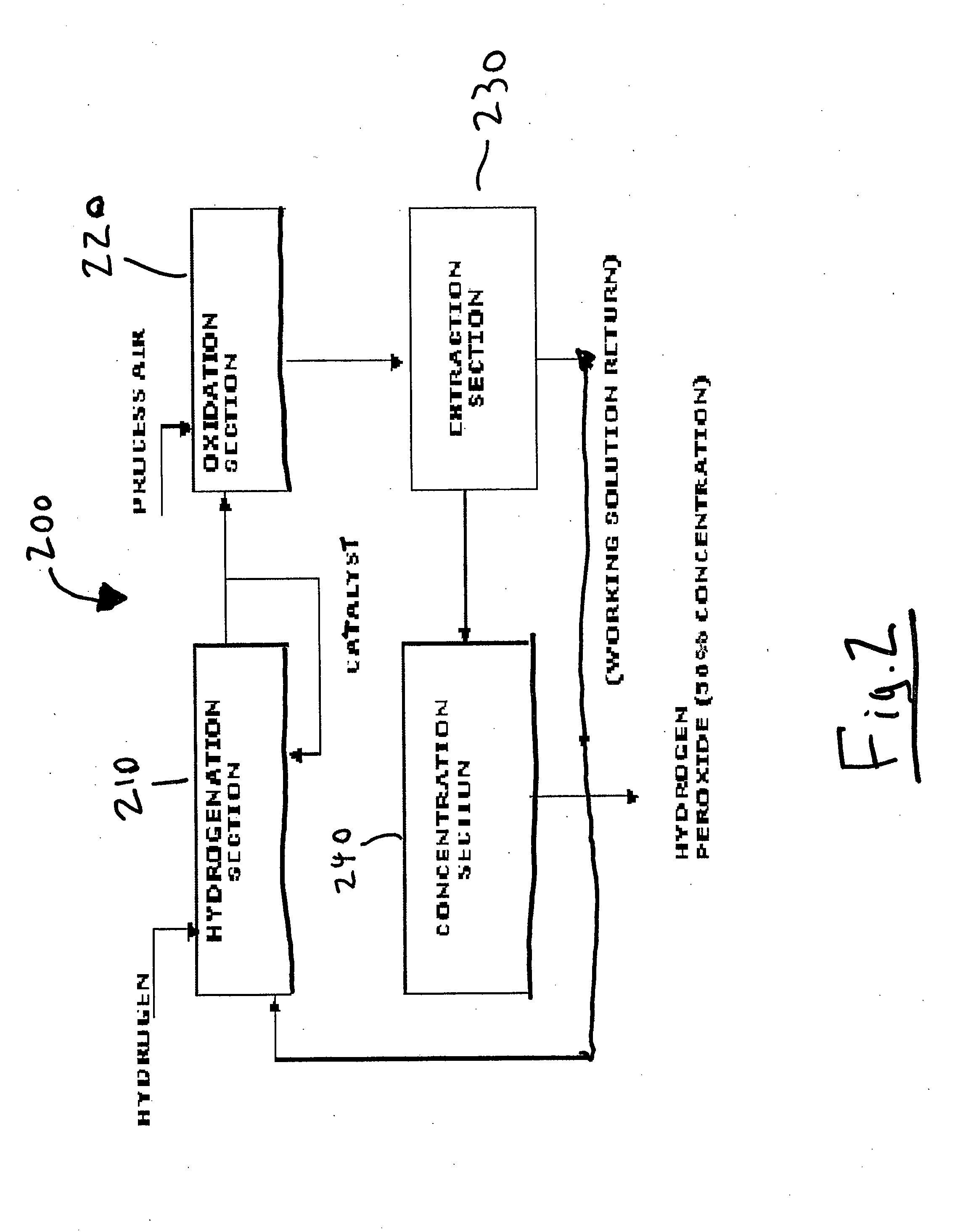
System and method for storing energy and/or generating efficient energy
PatentInactiveUS20120080887A1
Innovation
- A system and method utilizing hydrogen peroxide (H2O2) for energy storage, where energy is generated from wind turbines, stored as H2O2, and later decomposed to produce steam driving a steam engine connected to a magnetic drive assembly to generate electricity on demand.
Fuel cell system using liquid fuel and hydrogen peroxide, and method for operating fuel cell
PatentWO2018101587A1
Innovation
- A fuel cell system utilizing liquid hydrocarbon fuels and hydrogen peroxide as an oxidizing agent, which enables autothermal reforming, water gas conversion, and hydrogen separation membrane processes to produce high-purity hydrogen with reduced energy consumption and noise, eliminating the need for additional oxygen and carbon monoxide removal reactors.
Safety Regulations for Peroxide Energy Applications
The regulatory landscape for peroxide-based energy applications has evolved significantly as these systems transition from laboratory research to commercial deployment. Current safety frameworks primarily stem from traditional chemical handling protocols, which require substantial adaptation to address the unique characteristics of peroxide energy systems. The International Electrotechnical Commission (IEC) and various national standards organizations are developing specialized guidelines that encompass storage, transportation, and operational safety requirements specific to energy applications.
Concentration limits represent a critical regulatory parameter, with most jurisdictions establishing maximum allowable concentrations for different application scenarios. Industrial energy storage systems typically operate under stricter concentration thresholds compared to smaller-scale applications, reflecting the increased risk potential of larger peroxide volumes. These regulations also mandate specific containment materials and design specifications to prevent decomposition reactions that could lead to thermal runaway conditions.
Personnel safety requirements constitute another fundamental regulatory pillar, mandating comprehensive training programs for operators and maintenance personnel. These protocols include emergency response procedures, proper handling techniques, and regular safety assessments. Regulatory bodies increasingly require certification programs that demonstrate competency in peroxide energy system management, particularly for industrial-scale installations.
Environmental protection regulations address potential ecological impacts from peroxide energy systems, establishing discharge limits and waste management protocols. These frameworks recognize that while peroxide decomposition products are generally environmentally benign, concentrated releases could still pose localized risks. Regulatory compliance often requires environmental monitoring systems and contingency plans for accidental releases.
Emerging regulatory trends indicate a shift toward performance-based standards rather than prescriptive requirements, allowing greater innovation flexibility while maintaining safety objectives. International harmonization efforts are underway to establish consistent global standards, facilitating technology transfer and commercial deployment across different markets. These evolving frameworks increasingly incorporate risk-based assessment methodologies that account for system-specific factors rather than applying blanket restrictions.
The regulatory approval process for new peroxide energy applications typically involves multi-stage assessments, including laboratory testing, pilot demonstrations, and comprehensive safety evaluations. Regulatory agencies are developing expedited pathways for technologies that demonstrate superior safety profiles while maintaining rigorous oversight of novel applications that may present unprecedented risk scenarios.
Concentration limits represent a critical regulatory parameter, with most jurisdictions establishing maximum allowable concentrations for different application scenarios. Industrial energy storage systems typically operate under stricter concentration thresholds compared to smaller-scale applications, reflecting the increased risk potential of larger peroxide volumes. These regulations also mandate specific containment materials and design specifications to prevent decomposition reactions that could lead to thermal runaway conditions.
Personnel safety requirements constitute another fundamental regulatory pillar, mandating comprehensive training programs for operators and maintenance personnel. These protocols include emergency response procedures, proper handling techniques, and regular safety assessments. Regulatory bodies increasingly require certification programs that demonstrate competency in peroxide energy system management, particularly for industrial-scale installations.
Environmental protection regulations address potential ecological impacts from peroxide energy systems, establishing discharge limits and waste management protocols. These frameworks recognize that while peroxide decomposition products are generally environmentally benign, concentrated releases could still pose localized risks. Regulatory compliance often requires environmental monitoring systems and contingency plans for accidental releases.
Emerging regulatory trends indicate a shift toward performance-based standards rather than prescriptive requirements, allowing greater innovation flexibility while maintaining safety objectives. International harmonization efforts are underway to establish consistent global standards, facilitating technology transfer and commercial deployment across different markets. These evolving frameworks increasingly incorporate risk-based assessment methodologies that account for system-specific factors rather than applying blanket restrictions.
The regulatory approval process for new peroxide energy applications typically involves multi-stage assessments, including laboratory testing, pilot demonstrations, and comprehensive safety evaluations. Regulatory agencies are developing expedited pathways for technologies that demonstrate superior safety profiles while maintaining rigorous oversight of novel applications that may present unprecedented risk scenarios.
Environmental Impact of Peroxide Energy Systems
Peroxide-based energy systems present a complex environmental profile that requires comprehensive assessment across multiple impact categories. These systems, while offering significant advantages in energy density and storage capabilities, introduce unique environmental considerations that differ substantially from conventional battery technologies and fossil fuel alternatives.
The primary environmental concern stems from hydrogen peroxide production processes, which traditionally rely on energy-intensive anthraquinone oxidation methods. This manufacturing pathway generates considerable carbon emissions, with typical production facilities consuming 2.5-3.5 kWh per kilogram of hydrogen peroxide produced. However, emerging electrochemical synthesis routes demonstrate potential for reducing production-related emissions by up to 40% when powered by renewable energy sources.
Lifecycle assessment studies reveal that peroxide energy systems exhibit favorable environmental performance during operational phases. Unlike lithium-ion batteries, these systems avoid rare earth metal extraction and associated ecological disruption. The absence of heavy metals eliminates long-term soil and groundwater contamination risks, while the primary decomposition products—water and oxygen—pose minimal environmental hazards.
Water resource implications represent another critical consideration. Peroxide energy systems require substantial water inputs for both production and safety management protocols. Industrial-scale implementations typically consume 15-20 liters of process water per kilogram of peroxide handled, raising concerns about water scarcity in deployment regions.
Atmospheric impact assessments indicate mixed environmental outcomes. While operational emissions remain negligible, accidental releases can temporarily elevate local oxygen concentrations, potentially affecting sensitive ecosystems. However, the rapid decomposition characteristics of hydrogen peroxide limit the duration and geographic scope of such impacts.
End-of-life environmental benefits distinguish peroxide systems from conventional energy storage technologies. The absence of toxic materials eliminates specialized disposal requirements, while system components largely consist of recyclable metals and polymers. This characteristic significantly reduces long-term environmental liability compared to battery-based alternatives.
Comparative studies suggest that peroxide energy systems demonstrate superior environmental performance in applications requiring high energy density and infrequent cycling, particularly when integrated with renewable energy sources for peroxide production.
The primary environmental concern stems from hydrogen peroxide production processes, which traditionally rely on energy-intensive anthraquinone oxidation methods. This manufacturing pathway generates considerable carbon emissions, with typical production facilities consuming 2.5-3.5 kWh per kilogram of hydrogen peroxide produced. However, emerging electrochemical synthesis routes demonstrate potential for reducing production-related emissions by up to 40% when powered by renewable energy sources.
Lifecycle assessment studies reveal that peroxide energy systems exhibit favorable environmental performance during operational phases. Unlike lithium-ion batteries, these systems avoid rare earth metal extraction and associated ecological disruption. The absence of heavy metals eliminates long-term soil and groundwater contamination risks, while the primary decomposition products—water and oxygen—pose minimal environmental hazards.
Water resource implications represent another critical consideration. Peroxide energy systems require substantial water inputs for both production and safety management protocols. Industrial-scale implementations typically consume 15-20 liters of process water per kilogram of peroxide handled, raising concerns about water scarcity in deployment regions.
Atmospheric impact assessments indicate mixed environmental outcomes. While operational emissions remain negligible, accidental releases can temporarily elevate local oxygen concentrations, potentially affecting sensitive ecosystems. However, the rapid decomposition characteristics of hydrogen peroxide limit the duration and geographic scope of such impacts.
End-of-life environmental benefits distinguish peroxide systems from conventional energy storage technologies. The absence of toxic materials eliminates specialized disposal requirements, while system components largely consist of recyclable metals and polymers. This characteristic significantly reduces long-term environmental liability compared to battery-based alternatives.
Comparative studies suggest that peroxide energy systems demonstrate superior environmental performance in applications requiring high energy density and infrequent cycling, particularly when integrated with renewable energy sources for peroxide production.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!