Proton Membrane vs Ceramic Electrolytes: Efficacy
MAR 10, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Proton Membrane vs Ceramic Electrolyte Technology Background
The development of proton-conducting electrolytes has emerged as a critical frontier in advanced energy storage and conversion technologies, particularly in fuel cells, electrolyzers, and next-generation batteries. This field encompasses two primary technological pathways: proton exchange membranes (PEMs) and ceramic proton conductors, each representing distinct approaches to achieving efficient proton transport in electrochemical devices.
Proton exchange membrane technology originated in the 1960s with NASA's space program requirements for reliable fuel cell systems. The initial development focused on perfluorosulfonic acid membranes, with DuPont's Nafion becoming the benchmark material. These polymer-based membranes operate effectively at moderate temperatures (60-80°C) and provide excellent proton conductivity in hydrated conditions. The technology has since evolved to include hydrocarbon-based membranes, composite materials, and specialized formulations for various operating conditions.
Ceramic proton conductors represent a fundamentally different approach, utilizing solid oxide materials that exhibit proton conductivity at elevated temperatures (400-800°C). This technology pathway emerged from solid oxide fuel cell research in the 1980s, with perovskite-structured ceramics such as barium cerates and barium zirconates leading the development. These materials offer unique advantages including high-temperature operation capability, chemical stability, and potential for direct hydrocarbon fuel utilization without external reforming.
The technological evolution has been driven by the growing demand for clean energy solutions, particularly in transportation, stationary power generation, and industrial applications. Automotive fuel cell vehicles have primarily adopted PEM technology due to its rapid startup characteristics and moderate operating temperatures. Conversely, ceramic electrolytes have found applications in high-temperature fuel cells and solid oxide electrolysis cells for hydrogen production.
Recent technological advances have focused on addressing fundamental limitations in both pathways. For PEM systems, research emphasizes improving durability, reducing platinum catalyst loading, and enhancing performance under varying humidity conditions. Ceramic electrolyte development concentrates on reducing operating temperatures while maintaining conductivity, improving mechanical properties, and developing cost-effective manufacturing processes.
The convergence of these technologies with emerging applications such as reversible fuel cells, metal-air batteries, and electrochemical synthesis systems has expanded the scope of proton conductor research. Integration with renewable energy systems and grid-scale energy storage applications continues to drive innovation in both membrane and ceramic electrolyte technologies, establishing new performance benchmarks and application requirements.
Proton exchange membrane technology originated in the 1960s with NASA's space program requirements for reliable fuel cell systems. The initial development focused on perfluorosulfonic acid membranes, with DuPont's Nafion becoming the benchmark material. These polymer-based membranes operate effectively at moderate temperatures (60-80°C) and provide excellent proton conductivity in hydrated conditions. The technology has since evolved to include hydrocarbon-based membranes, composite materials, and specialized formulations for various operating conditions.
Ceramic proton conductors represent a fundamentally different approach, utilizing solid oxide materials that exhibit proton conductivity at elevated temperatures (400-800°C). This technology pathway emerged from solid oxide fuel cell research in the 1980s, with perovskite-structured ceramics such as barium cerates and barium zirconates leading the development. These materials offer unique advantages including high-temperature operation capability, chemical stability, and potential for direct hydrocarbon fuel utilization without external reforming.
The technological evolution has been driven by the growing demand for clean energy solutions, particularly in transportation, stationary power generation, and industrial applications. Automotive fuel cell vehicles have primarily adopted PEM technology due to its rapid startup characteristics and moderate operating temperatures. Conversely, ceramic electrolytes have found applications in high-temperature fuel cells and solid oxide electrolysis cells for hydrogen production.
Recent technological advances have focused on addressing fundamental limitations in both pathways. For PEM systems, research emphasizes improving durability, reducing platinum catalyst loading, and enhancing performance under varying humidity conditions. Ceramic electrolyte development concentrates on reducing operating temperatures while maintaining conductivity, improving mechanical properties, and developing cost-effective manufacturing processes.
The convergence of these technologies with emerging applications such as reversible fuel cells, metal-air batteries, and electrochemical synthesis systems has expanded the scope of proton conductor research. Integration with renewable energy systems and grid-scale energy storage applications continues to drive innovation in both membrane and ceramic electrolyte technologies, establishing new performance benchmarks and application requirements.
Market Demand for Advanced Electrolyte Solutions
The global electrolyte market is experiencing unprecedented growth driven by the rapid expansion of energy storage systems, electric vehicles, and fuel cell technologies. Traditional liquid electrolytes face increasing scrutiny due to safety concerns, thermal instability, and limited operating temperature ranges, creating substantial demand for advanced solid-state alternatives.
Proton exchange membrane fuel cells represent a significant market segment where electrolyte performance directly impacts commercial viability. The automotive industry's transition toward hydrogen-powered vehicles has intensified requirements for electrolytes that can operate efficiently across diverse environmental conditions while maintaining long-term durability. Current market drivers include stringent emission regulations, government incentives for clean energy adoption, and corporate sustainability commitments.
Ceramic electrolytes are gaining traction in high-temperature applications, particularly in solid oxide fuel cells and advanced battery systems. Industrial applications requiring robust performance under extreme conditions show strong preference for ceramic-based solutions, despite higher initial costs. The market demand stems from their superior chemical stability and wide electrochemical windows.
Proton-conducting membranes dominate lower-temperature applications, especially in portable electronics and automotive fuel cells. Market acceptance has been driven by their established manufacturing infrastructure and relatively mature supply chains. However, growing concerns about membrane degradation and water management challenges are pushing end-users to explore alternative solutions.
The energy storage sector presents the largest growth opportunity for advanced electrolytes. Grid-scale storage projects require electrolytes capable of thousands of charge-discharge cycles while maintaining safety standards. This application segment shows willingness to adopt premium electrolyte technologies that demonstrate superior lifecycle performance.
Emerging markets in Asia-Pacific exhibit particularly strong demand for next-generation electrolyte solutions, driven by aggressive renewable energy deployment and electric vehicle adoption policies. Regional manufacturers are increasingly seeking electrolyte technologies that can differentiate their products in competitive markets while meeting international safety and performance standards.
Cost-performance optimization remains the primary market driver, with end-users seeking electrolyte solutions that balance initial investment against operational benefits including efficiency gains, extended service life, and reduced maintenance requirements.
Proton exchange membrane fuel cells represent a significant market segment where electrolyte performance directly impacts commercial viability. The automotive industry's transition toward hydrogen-powered vehicles has intensified requirements for electrolytes that can operate efficiently across diverse environmental conditions while maintaining long-term durability. Current market drivers include stringent emission regulations, government incentives for clean energy adoption, and corporate sustainability commitments.
Ceramic electrolytes are gaining traction in high-temperature applications, particularly in solid oxide fuel cells and advanced battery systems. Industrial applications requiring robust performance under extreme conditions show strong preference for ceramic-based solutions, despite higher initial costs. The market demand stems from their superior chemical stability and wide electrochemical windows.
Proton-conducting membranes dominate lower-temperature applications, especially in portable electronics and automotive fuel cells. Market acceptance has been driven by their established manufacturing infrastructure and relatively mature supply chains. However, growing concerns about membrane degradation and water management challenges are pushing end-users to explore alternative solutions.
The energy storage sector presents the largest growth opportunity for advanced electrolytes. Grid-scale storage projects require electrolytes capable of thousands of charge-discharge cycles while maintaining safety standards. This application segment shows willingness to adopt premium electrolyte technologies that demonstrate superior lifecycle performance.
Emerging markets in Asia-Pacific exhibit particularly strong demand for next-generation electrolyte solutions, driven by aggressive renewable energy deployment and electric vehicle adoption policies. Regional manufacturers are increasingly seeking electrolyte technologies that can differentiate their products in competitive markets while meeting international safety and performance standards.
Cost-performance optimization remains the primary market driver, with end-users seeking electrolyte solutions that balance initial investment against operational benefits including efficiency gains, extended service life, and reduced maintenance requirements.
Current State of Proton and Ceramic Electrolyte Technologies
Proton exchange membrane (PEM) electrolytes have achieved significant commercial maturity, particularly in fuel cell applications and emerging electrolyzer systems. Current PEM technologies primarily utilize perfluorosulfonic acid membranes, with Nafion remaining the industry standard despite its high cost and temperature limitations. These membranes typically operate effectively between 60-80°C and demonstrate proton conductivities of 0.1-0.2 S/cm under fully hydrated conditions. However, their performance degrades substantially at temperatures exceeding 100°C due to dehydration, limiting their application scope in high-temperature industrial processes.
Recent developments in PEM technology focus on addressing durability and cost challenges. Advanced membrane formulations incorporating reinforced structures and alternative ionomer chemistries have extended operational lifespans to over 40,000 hours in stationary applications. Manufacturing scalability has improved significantly, with several facilities achieving production capacities exceeding 1 million square meters annually.
Ceramic electrolyte technologies represent a rapidly evolving field with substantial potential for high-temperature applications. Solid oxide electrolytes, particularly yttria-stabilized zirconia (YSZ), dominate current ceramic implementations in fuel cells operating at 700-1000°C. These materials exhibit excellent chemical stability and ionic conductivities approaching 0.1 S/cm at operating temperatures, enabling efficient electrochemical processes without the hydration requirements of PEM systems.
Emerging ceramic electrolyte compositions, including gadolinium-doped ceria and lanthanum gallate-based materials, demonstrate enhanced performance characteristics. These advanced ceramics achieve higher ionic conductivities at reduced operating temperatures, with some formulations showing promising results at 500-600°C. Manufacturing processes have evolved from traditional sintering methods to advanced techniques including tape casting and screen printing, enabling thinner electrolyte layers and improved performance density.
Both technology categories face distinct manufacturing and integration challenges. PEM systems require sophisticated water management and gas handling infrastructure, while ceramic electrolytes demand high-temperature sealing solutions and thermal cycling durability. Current research efforts concentrate on hybrid approaches combining advantages of both technologies, including composite membranes incorporating ceramic nanoparticles and intermediate-temperature ceramic compositions suitable for broader application ranges.
The technological maturity gap between these approaches continues narrowing as ceramic processing advances and PEM durability improvements progress simultaneously.
Recent developments in PEM technology focus on addressing durability and cost challenges. Advanced membrane formulations incorporating reinforced structures and alternative ionomer chemistries have extended operational lifespans to over 40,000 hours in stationary applications. Manufacturing scalability has improved significantly, with several facilities achieving production capacities exceeding 1 million square meters annually.
Ceramic electrolyte technologies represent a rapidly evolving field with substantial potential for high-temperature applications. Solid oxide electrolytes, particularly yttria-stabilized zirconia (YSZ), dominate current ceramic implementations in fuel cells operating at 700-1000°C. These materials exhibit excellent chemical stability and ionic conductivities approaching 0.1 S/cm at operating temperatures, enabling efficient electrochemical processes without the hydration requirements of PEM systems.
Emerging ceramic electrolyte compositions, including gadolinium-doped ceria and lanthanum gallate-based materials, demonstrate enhanced performance characteristics. These advanced ceramics achieve higher ionic conductivities at reduced operating temperatures, with some formulations showing promising results at 500-600°C. Manufacturing processes have evolved from traditional sintering methods to advanced techniques including tape casting and screen printing, enabling thinner electrolyte layers and improved performance density.
Both technology categories face distinct manufacturing and integration challenges. PEM systems require sophisticated water management and gas handling infrastructure, while ceramic electrolytes demand high-temperature sealing solutions and thermal cycling durability. Current research efforts concentrate on hybrid approaches combining advantages of both technologies, including composite membranes incorporating ceramic nanoparticles and intermediate-temperature ceramic compositions suitable for broader application ranges.
The technological maturity gap between these approaches continues narrowing as ceramic processing advances and PEM durability improvements progress simultaneously.
Existing Proton Membrane and Ceramic Electrolyte Solutions
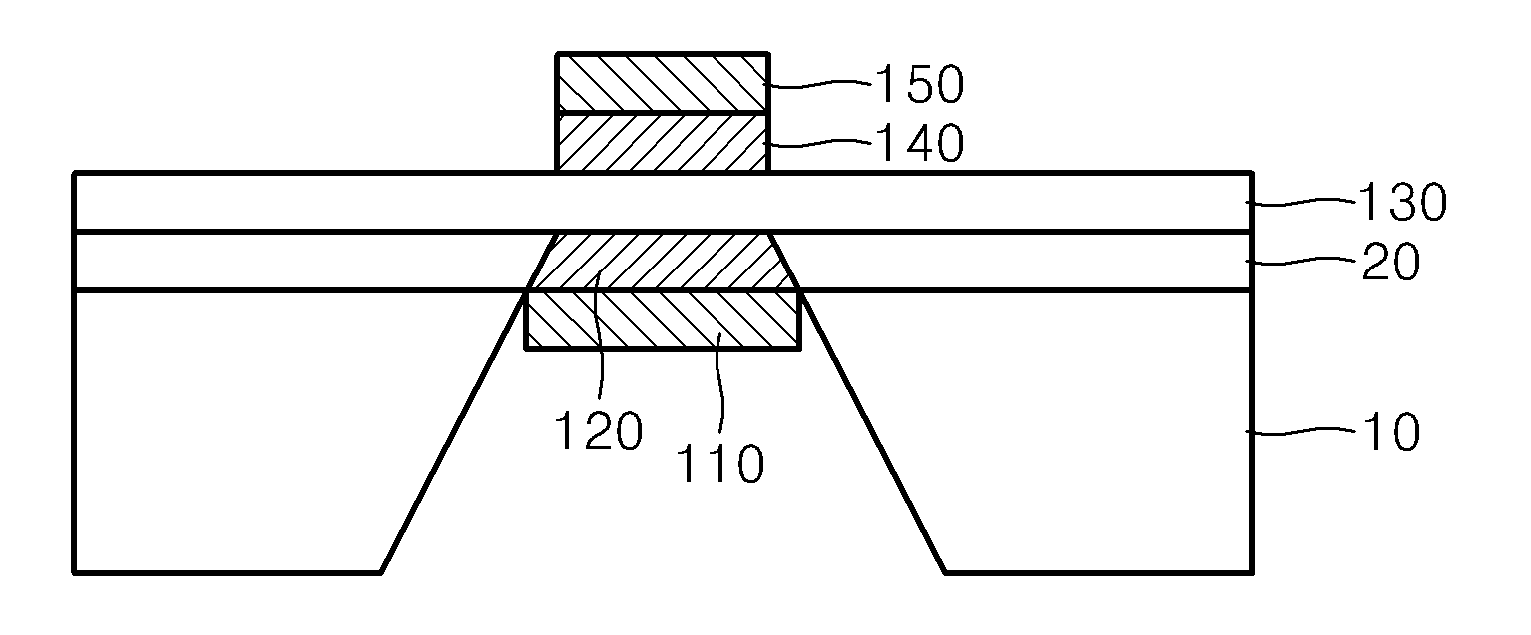
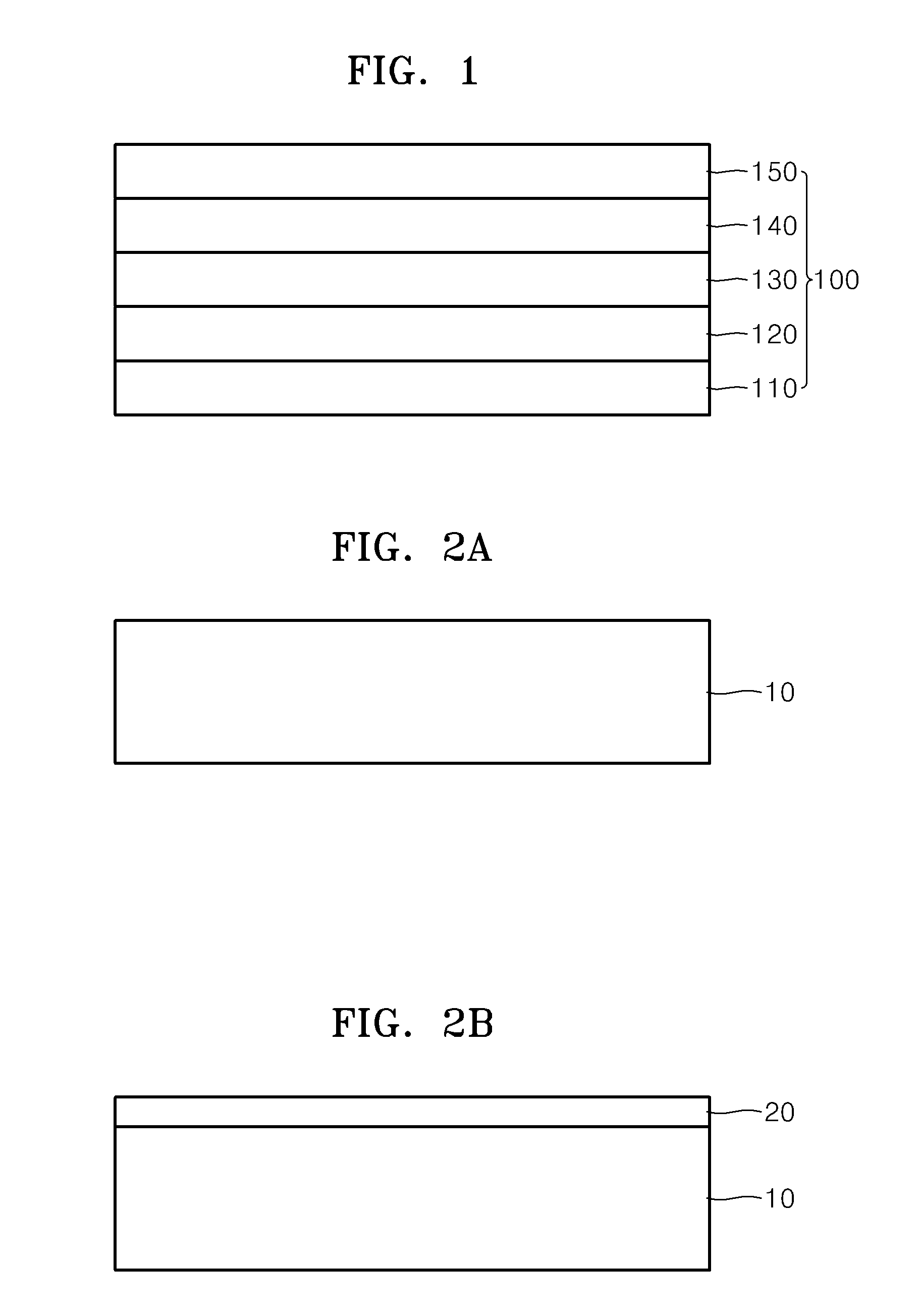
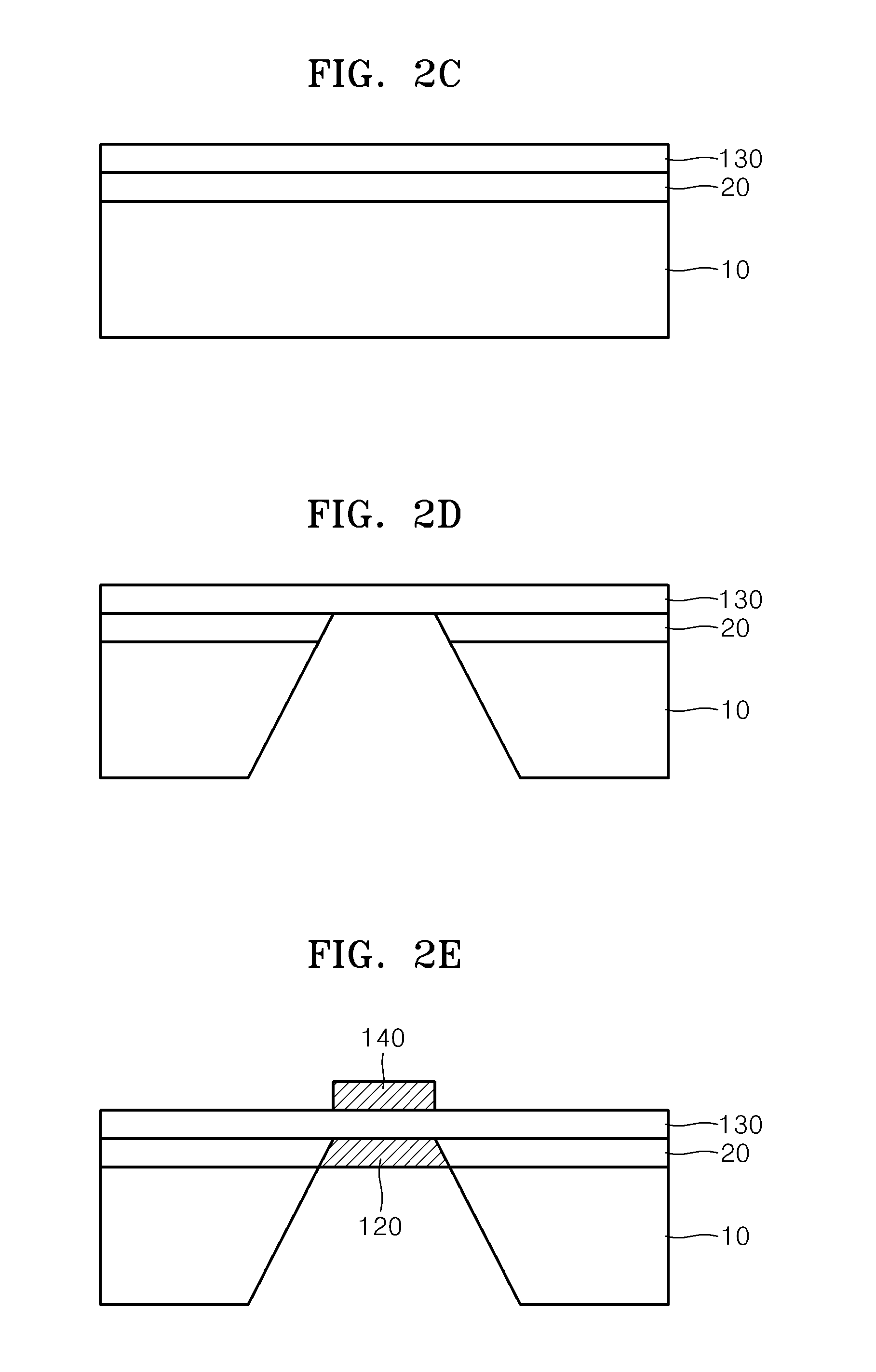
01 Composite proton exchange membranes with ceramic materials
Proton exchange membranes can be enhanced by incorporating ceramic materials such as metal oxides or inorganic particles to improve proton conductivity, mechanical strength, and thermal stability. These composite membranes demonstrate superior performance in fuel cell applications by reducing methanol crossover and maintaining high ionic conductivity across various operating conditions. The ceramic components act as both reinforcing agents and proton conducting pathways.- Composite proton exchange membranes with ceramic materials: Proton exchange membranes can be enhanced by incorporating ceramic materials such as metal oxides or inorganic particles to improve proton conductivity, mechanical strength, and thermal stability. These composite membranes demonstrate superior performance in fuel cell applications by reducing methanol crossover and maintaining high ionic conductivity across various operating conditions. The ceramic components act as reinforcing agents while providing additional proton transport pathways.
- Solid oxide electrolytes with enhanced ionic conductivity: Ceramic electrolytes based on solid oxide materials exhibit high ionic conductivity at elevated temperatures, making them suitable for solid oxide fuel cells and electrochemical devices. These electrolytes can be optimized through doping strategies and microstructure control to achieve improved oxygen ion transport and reduced activation energy. The materials demonstrate excellent chemical stability and mechanical integrity under harsh operating conditions.
- Hybrid membrane-electrode assemblies: Integration of proton-conducting membranes with ceramic electrode materials creates hybrid assemblies that combine the advantages of both components. These assemblies optimize the interface between electrolyte and electrode, reducing interfacial resistance and improving overall electrochemical performance. The design enables better catalyst utilization and enhanced durability in energy conversion devices.
- Nanostructured ceramic electrolytes for improved performance: Nanostructured ceramic electrolytes with controlled morphology and grain boundaries exhibit enhanced ionic conductivity and reduced ohmic losses. The nanoscale architecture provides increased surface area and shortened ion transport pathways, leading to superior electrochemical properties. These materials can be fabricated through various synthesis methods to achieve optimal particle size distribution and porosity.
- Protective coatings and interfacial layers for electrolyte stability: Application of protective ceramic coatings or interfacial layers on proton exchange membranes enhances chemical stability, prevents degradation, and extends operational lifetime. These layers act as barriers against contaminants while maintaining ionic conductivity. The coating strategies include thin film deposition and surface modification techniques that improve compatibility between different electrolyte components.
02 Solid oxide electrolytes for high-temperature applications
Ceramic electrolytes based on solid oxide materials exhibit excellent ionic conductivity at elevated temperatures, making them suitable for solid oxide fuel cells and electrochemical devices. These electrolytes typically feature stabilized zirconia or ceria-based compositions that provide high oxygen ion conductivity while maintaining chemical and structural stability. The materials demonstrate enhanced performance through optimized sintering processes and dopant selection.Expand Specific Solutions03 Hybrid organic-inorganic proton conducting membranes
Hybrid membranes combining organic polymers with inorganic ceramic phases offer balanced properties of flexibility and conductivity. These materials leverage the processability of polymers while incorporating ceramic nanoparticles or layers to enhance proton transport mechanisms and reduce fuel permeability. The synergistic effect between organic and inorganic components results in improved durability and electrochemical performance.Expand Specific Solutions04 Nanostructured ceramic electrolytes with enhanced ionic conductivity
Nanostructured ceramic electrolytes utilize controlled morphology and grain boundary engineering to achieve superior ionic transport properties. These materials feature reduced grain sizes, optimized porosity, and tailored interfaces that facilitate ion migration pathways. Advanced fabrication techniques enable the production of thin-film electrolytes with minimized resistance and improved power density for energy storage and conversion devices.Expand Specific Solutions05 Doped ceramic electrolytes for improved stability and performance
Strategic doping of ceramic electrolytes with specific elements enhances their electrochemical properties and operational stability. The introduction of dopants modifies the crystal structure, creates oxygen vacancies, and improves ionic conductivity while maintaining mechanical integrity. These modified electrolytes demonstrate extended operational lifetimes and better resistance to degradation under harsh operating conditions.Expand Specific Solutions
Key Players in Electrolyte Technology Industry
The proton membrane versus ceramic electrolytes technology landscape represents a rapidly evolving sector within the broader hydrogen economy and energy storage markets. The industry is currently in a transitional phase, moving from laboratory research to commercial deployment, with significant investments driving market expansion estimated to reach billions globally by 2030. Technology maturity varies considerably across players, with established companies like Toyota Motor Corp., Samsung SDI, and LG Chem leading in proton exchange membrane development, while research institutions such as Shanghai Institute of Ceramics and Tokyo Metropolitan University advance ceramic electrolyte innovations. European entities including CEA and Greenerity GmbH focus on PEM electrolysis applications, whereas emerging companies like Power To Hydrogen develop hybrid solutions combining both technologies, indicating a competitive landscape where traditional automotive and electronics manufacturers compete alongside specialized startups and academic institutions.
Toray Industries, Inc.
Technical Solution: Toray Industries specializes in advanced proton exchange membrane technology, developing perfluorosulfonic acid membranes with enhanced durability and performance characteristics. Their membrane products achieve proton conductivities exceeding 0.15 S/cm under optimal hydration conditions while maintaining mechanical integrity over extended operating periods. Toray has also explored ceramic-polymer composite electrolytes, incorporating ceramic nanoparticles into polymer matrices to enhance thermal stability and reduce methanol crossover in direct methanol fuel cells. The company's membrane technology features optimized equivalent weight and thickness parameters, typically ranging from 15-50 μm, to balance conductivity with gas barrier properties. Their manufacturing process utilizes solution casting and thermal treatment to achieve uniform membrane structure with controlled water uptake characteristics and dimensional stability.
Strengths: Excellent membrane durability, proven manufacturing scalability, superior chemical resistance. Weaknesses: Requires humidification for optimal performance, limited high-temperature operation, relatively high material costs.
Toyota Motor Corp.
Technical Solution: Toyota has developed advanced proton exchange membrane fuel cell technology with their MIRAI vehicle platform, achieving high proton conductivity through perfluorosulfonic acid membranes operating at temperatures up to 80°C. Their membrane technology demonstrates excellent chemical stability and durability under automotive operating conditions. The company has also invested in ceramic electrolyte research for solid oxide fuel cells, exploring yttria-stabilized zirconia (YSZ) and gadolinium-doped ceria (GDC) materials for high-temperature applications. Toyota's approach focuses on optimizing membrane thickness to balance proton conductivity with mechanical strength, achieving power densities exceeding 3.1 kW/L in their latest fuel cell systems.
Strengths: Proven commercial deployment experience, excellent durability and reliability in automotive applications. Weaknesses: Limited high-temperature operation capability, dependency on humidification systems for optimal performance.
Core Innovations in Electrolyte Efficacy Enhancement
Proton conducting electrolyte membranes having nano-grain YSZ as protective layers, and membrane electrode assemblies and ceramic fuel cells comprising same
PatentInactiveUS20110262839A1
Innovation
- A proton conducting electrolyte membrane with a ceramic protective layer of nano-grain yttrium-stabilized zirconia (YSZ) is used, which has proton conductivity and prevents CO2 penetration, enhancing the compatibility and stability of the ceramic electrolyte layer, and is formed using techniques like atomic layer deposition (ALD) or pulsed laser deposition (PLD).
Electrochemical device comprising a proton-conducting ceramic electrolyte
PatentWO2008043943A1
Innovation
- A proton-conducting ceramic electrolyte based on barium and indium, with a formula (Ba2(I-X)M2xIn2(I-Y)M'2yO4+δ(OH)δ', exhibiting high proton conductivity between 200°C and 600°C, is used in electrochemical devices, offering a compromise between proton mobility and hydroxyl group density, reducing the need for expensive catalysts and enhancing thermal stability.
Safety Standards for Solid-State Battery Technologies
The development of comprehensive safety standards for solid-state battery technologies represents a critical regulatory framework that must address the unique characteristics of both proton membrane and ceramic electrolyte systems. Current international standards, including IEC 62133 and UN 38.3, primarily focus on conventional lithium-ion batteries and require substantial modifications to accommodate solid-state architectures.
Proton membrane electrolytes present distinct safety considerations due to their sensitivity to moisture and temperature variations. These systems require specialized testing protocols that evaluate membrane integrity under thermal stress, humidity exposure, and mechanical deformation. The standards must establish threshold values for proton conductivity degradation and define acceptable limits for water vapor permeation that could compromise battery performance or safety.
Ceramic electrolyte systems demand different safety evaluation criteria, particularly regarding mechanical stress testing and thermal shock resistance. The brittle nature of ceramic materials necessitates rigorous fracture testing protocols and impact resistance assessments. Standards must define minimum mechanical strength requirements and establish testing methodologies for detecting microscopic cracks that could lead to dendrite formation or internal short circuits.
Thermal management standards for solid-state batteries require temperature gradient specifications that differ significantly from liquid electrolyte systems. The standards must address thermal runaway characteristics unique to each electrolyte type, establishing maximum operating temperatures and defining cooling requirements for different application scenarios.
Electrochemical safety protocols must encompass overcharge protection, deep discharge limits, and cycling stability requirements specific to solid-state architectures. These standards should define acceptable capacity fade rates and establish testing procedures for long-term stability assessment under various environmental conditions.
Manufacturing quality control standards must address the unique production challenges of solid-state batteries, including interface quality between electrodes and electrolytes, void detection methodologies, and contamination control procedures. The standards should establish acceptance criteria for manufacturing defects and define quality assurance protocols for both proton membrane and ceramic electrolyte production processes.
Proton membrane electrolytes present distinct safety considerations due to their sensitivity to moisture and temperature variations. These systems require specialized testing protocols that evaluate membrane integrity under thermal stress, humidity exposure, and mechanical deformation. The standards must establish threshold values for proton conductivity degradation and define acceptable limits for water vapor permeation that could compromise battery performance or safety.
Ceramic electrolyte systems demand different safety evaluation criteria, particularly regarding mechanical stress testing and thermal shock resistance. The brittle nature of ceramic materials necessitates rigorous fracture testing protocols and impact resistance assessments. Standards must define minimum mechanical strength requirements and establish testing methodologies for detecting microscopic cracks that could lead to dendrite formation or internal short circuits.
Thermal management standards for solid-state batteries require temperature gradient specifications that differ significantly from liquid electrolyte systems. The standards must address thermal runaway characteristics unique to each electrolyte type, establishing maximum operating temperatures and defining cooling requirements for different application scenarios.
Electrochemical safety protocols must encompass overcharge protection, deep discharge limits, and cycling stability requirements specific to solid-state architectures. These standards should define acceptable capacity fade rates and establish testing procedures for long-term stability assessment under various environmental conditions.
Manufacturing quality control standards must address the unique production challenges of solid-state batteries, including interface quality between electrodes and electrolytes, void detection methodologies, and contamination control procedures. The standards should establish acceptance criteria for manufacturing defects and define quality assurance protocols for both proton membrane and ceramic electrolyte production processes.
Manufacturing Scalability of Advanced Electrolytes
The manufacturing scalability of advanced electrolytes represents a critical bottleneck in the widespread adoption of next-generation energy storage systems. Both proton exchange membranes and ceramic electrolytes face distinct manufacturing challenges that significantly impact their commercial viability and production economics.
Proton exchange membrane manufacturing has achieved considerable maturity through decades of fuel cell development, with established production processes including solution casting, extrusion, and roll-to-roll processing. Current manufacturing capabilities can produce membranes at industrial scales, with companies like DuPont and 3M operating facilities capable of producing thousands of square meters annually. However, the high-temperature processing requirements and specialized fluoropolymer chemistry limit cost reduction potential and require significant capital investment for facility upgrades.
Ceramic electrolyte manufacturing presents more complex scalability challenges due to the precision required in solid-state processing. Traditional ceramic manufacturing techniques such as tape casting, screen printing, and sintering require careful control of temperature profiles, atmosphere conditions, and dimensional tolerances. The brittleness of ceramic materials introduces yield concerns during handling and assembly processes, potentially impacting overall production economics.
Recent advances in manufacturing technologies are addressing these scalability limitations. For proton membranes, continuous processing methods and improved catalyst deposition techniques are reducing production costs while maintaining performance standards. Automated quality control systems enable real-time monitoring of membrane thickness, conductivity, and defect detection across large production runs.
Ceramic electrolyte manufacturing is benefiting from advanced powder processing techniques, including spray drying, sol-gel methods, and additive manufacturing approaches. These technologies enable better control over microstructure and reduce sintering temperatures, potentially improving production throughput. Co-firing techniques allow simultaneous processing of multiple layers, reducing manufacturing steps and associated costs.
The scalability comparison reveals that proton membranes currently hold advantages in established manufacturing infrastructure and process maturity. However, ceramic electrolytes show promising potential for cost reduction through emerging manufacturing technologies and simplified assembly processes that eliminate liquid electrolyte handling requirements.
Proton exchange membrane manufacturing has achieved considerable maturity through decades of fuel cell development, with established production processes including solution casting, extrusion, and roll-to-roll processing. Current manufacturing capabilities can produce membranes at industrial scales, with companies like DuPont and 3M operating facilities capable of producing thousands of square meters annually. However, the high-temperature processing requirements and specialized fluoropolymer chemistry limit cost reduction potential and require significant capital investment for facility upgrades.
Ceramic electrolyte manufacturing presents more complex scalability challenges due to the precision required in solid-state processing. Traditional ceramic manufacturing techniques such as tape casting, screen printing, and sintering require careful control of temperature profiles, atmosphere conditions, and dimensional tolerances. The brittleness of ceramic materials introduces yield concerns during handling and assembly processes, potentially impacting overall production economics.
Recent advances in manufacturing technologies are addressing these scalability limitations. For proton membranes, continuous processing methods and improved catalyst deposition techniques are reducing production costs while maintaining performance standards. Automated quality control systems enable real-time monitoring of membrane thickness, conductivity, and defect detection across large production runs.
Ceramic electrolyte manufacturing is benefiting from advanced powder processing techniques, including spray drying, sol-gel methods, and additive manufacturing approaches. These technologies enable better control over microstructure and reduce sintering temperatures, potentially improving production throughput. Co-firing techniques allow simultaneous processing of multiple layers, reducing manufacturing steps and associated costs.
The scalability comparison reveals that proton membranes currently hold advantages in established manufacturing infrastructure and process maturity. However, ceramic electrolytes show promising potential for cost reduction through emerging manufacturing technologies and simplified assembly processes that eliminate liquid electrolyte handling requirements.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!