Quantify Formation Kinetics in Layered Double Hydroxides during Carbonation
APR 13, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
LDH Carbonation Formation Kinetics Background and Objectives
Layered Double Hydroxides (LDHs) represent a significant class of anionic clay materials that have garnered substantial attention in materials science and environmental engineering over the past three decades. These synthetic clay minerals, characterized by their unique layered structure and anion exchange capabilities, have demonstrated remarkable potential in carbon capture and storage applications. The carbonation process of LDHs involves the intercalation of carbonate ions into the interlayer spaces, fundamentally altering their structural and chemical properties.
The historical development of LDH research began in the 1960s with basic structural characterization studies, evolving through the 1980s and 1990s with enhanced synthesis methodologies. The 2000s marked a pivotal period when researchers began focusing on LDH interactions with atmospheric CO2, recognizing their potential for carbon sequestration applications. Recent advances in analytical techniques have enabled more sophisticated investigations into the kinetic mechanisms governing LDH carbonation processes.
Current technological trends indicate a growing emphasis on understanding the temporal dynamics of LDH structural transformations during carbonation. The field has progressed from static structural analysis to dynamic kinetic studies, driven by the need for optimized carbon capture systems. Advanced characterization techniques including in-situ X-ray diffraction, infrared spectroscopy, and electron microscopy have revolutionized our ability to monitor real-time carbonation processes.
The primary objective of quantifying formation kinetics in LDH carbonation centers on developing comprehensive mathematical models that accurately predict reaction rates under varying environmental conditions. This involves establishing correlations between structural parameters, environmental factors such as temperature and CO2 concentration, and reaction kinetics. Understanding these relationships is crucial for designing efficient carbon capture systems and predicting long-term storage stability.
Secondary objectives encompass the identification of rate-limiting steps in the carbonation mechanism, characterization of intermediate phases formed during the process, and development of predictive models for industrial applications. These goals align with broader environmental objectives of enhancing carbon sequestration technologies and developing sustainable materials for climate change mitigation strategies.
The historical development of LDH research began in the 1960s with basic structural characterization studies, evolving through the 1980s and 1990s with enhanced synthesis methodologies. The 2000s marked a pivotal period when researchers began focusing on LDH interactions with atmospheric CO2, recognizing their potential for carbon sequestration applications. Recent advances in analytical techniques have enabled more sophisticated investigations into the kinetic mechanisms governing LDH carbonation processes.
Current technological trends indicate a growing emphasis on understanding the temporal dynamics of LDH structural transformations during carbonation. The field has progressed from static structural analysis to dynamic kinetic studies, driven by the need for optimized carbon capture systems. Advanced characterization techniques including in-situ X-ray diffraction, infrared spectroscopy, and electron microscopy have revolutionized our ability to monitor real-time carbonation processes.
The primary objective of quantifying formation kinetics in LDH carbonation centers on developing comprehensive mathematical models that accurately predict reaction rates under varying environmental conditions. This involves establishing correlations between structural parameters, environmental factors such as temperature and CO2 concentration, and reaction kinetics. Understanding these relationships is crucial for designing efficient carbon capture systems and predicting long-term storage stability.
Secondary objectives encompass the identification of rate-limiting steps in the carbonation mechanism, characterization of intermediate phases formed during the process, and development of predictive models for industrial applications. These goals align with broader environmental objectives of enhancing carbon sequestration technologies and developing sustainable materials for climate change mitigation strategies.
Market Demand for Advanced LDH Carbonation Applications
The global market for advanced layered double hydroxide (LDH) carbonation applications is experiencing significant growth driven by increasing environmental regulations and industrial demand for sustainable materials. Carbon capture and storage technologies represent the largest market segment, where LDH materials serve as efficient CO2 sorbents in industrial processes. The ability to quantify formation kinetics during carbonation has become crucial for optimizing these applications and meeting stringent performance requirements.
Water treatment applications constitute another major market driver, particularly in developing regions where industrial wastewater management is becoming increasingly regulated. LDH materials modified through controlled carbonation processes demonstrate enhanced adsorption capabilities for heavy metals and organic pollutants. The pharmaceutical and cosmetics industries are also emerging as significant consumers, utilizing carbonated LDH materials as drug delivery systems and stabilizing agents.
The construction industry presents substantial growth potential, with carbonated LDH materials being incorporated into cement formulations and concrete additives. These applications benefit from precise kinetic control during carbonation, which directly impacts material performance and durability. Green building certifications and sustainability mandates are accelerating adoption rates across major construction markets.
Energy storage applications, particularly in battery technologies and supercapacitors, represent a rapidly expanding market segment. The electrochemical properties of LDH materials can be fine-tuned through controlled carbonation processes, making kinetic quantification essential for product development. Electric vehicle manufacturers and renewable energy storage providers are driving demand for high-performance LDH materials.
Regional market dynamics show strong growth in Asia-Pacific regions, driven by industrial expansion and environmental compliance requirements. European markets emphasize high-value applications in pharmaceuticals and advanced materials, while North American demand focuses on environmental remediation and energy storage applications. The market trend toward customized LDH materials with specific carbonation profiles is creating opportunities for specialized manufacturers who can precisely control formation kinetics.
Water treatment applications constitute another major market driver, particularly in developing regions where industrial wastewater management is becoming increasingly regulated. LDH materials modified through controlled carbonation processes demonstrate enhanced adsorption capabilities for heavy metals and organic pollutants. The pharmaceutical and cosmetics industries are also emerging as significant consumers, utilizing carbonated LDH materials as drug delivery systems and stabilizing agents.
The construction industry presents substantial growth potential, with carbonated LDH materials being incorporated into cement formulations and concrete additives. These applications benefit from precise kinetic control during carbonation, which directly impacts material performance and durability. Green building certifications and sustainability mandates are accelerating adoption rates across major construction markets.
Energy storage applications, particularly in battery technologies and supercapacitors, represent a rapidly expanding market segment. The electrochemical properties of LDH materials can be fine-tuned through controlled carbonation processes, making kinetic quantification essential for product development. Electric vehicle manufacturers and renewable energy storage providers are driving demand for high-performance LDH materials.
Regional market dynamics show strong growth in Asia-Pacific regions, driven by industrial expansion and environmental compliance requirements. European markets emphasize high-value applications in pharmaceuticals and advanced materials, while North American demand focuses on environmental remediation and energy storage applications. The market trend toward customized LDH materials with specific carbonation profiles is creating opportunities for specialized manufacturers who can precisely control formation kinetics.
Current State and Challenges in LDH Carbonation Kinetics
The quantification of formation kinetics in layered double hydroxides during carbonation represents a critical yet incompletely understood aspect of LDH chemistry. Current research efforts have made significant progress in characterizing the structural and compositional changes that occur during CO2 uptake, but substantial gaps remain in developing comprehensive kinetic models that can accurately predict reaction rates under varying conditions.
Existing analytical approaches primarily rely on ex-situ characterization techniques such as X-ray diffraction, thermogravimetric analysis, and infrared spectroscopy to monitor carbonation progress. While these methods provide valuable insights into the final products and overall conversion rates, they offer limited temporal resolution for capturing the dynamic aspects of the carbonation process. The lack of real-time monitoring capabilities significantly hampers efforts to establish precise kinetic parameters and understand the mechanistic pathways governing LDH carbonation.
One of the most significant challenges lies in the complexity of the carbonation mechanism itself. The process involves multiple simultaneous phenomena including CO2 diffusion through the interlayer space, anion exchange reactions, structural reorganization, and potential phase transformations. Current kinetic models often oversimplify these interactions by treating carbonation as a single-step process, failing to account for the multi-stage nature of the reaction and the interdependencies between different mechanistic steps.
Temperature and humidity effects on carbonation kinetics remain poorly quantified across different LDH compositions. While qualitative trends have been established showing accelerated carbonation at elevated temperatures and humidity levels, precise mathematical relationships describing these dependencies are lacking. This limitation severely restricts the ability to predict LDH performance in real-world applications where environmental conditions vary significantly.
The heterogeneity of LDH materials presents another fundamental challenge. Variations in particle size, crystallinity, interlayer spacing, and surface area can dramatically influence carbonation rates, yet current kinetic models rarely incorporate these structural parameters as explicit variables. This oversight leads to significant discrepancies between predicted and observed carbonation behavior, particularly when comparing different synthesis methods or LDH compositions.
Furthermore, the influence of competing anions in practical applications remains inadequately characterized. Most kinetic studies focus on pure CO2 environments, but real-world scenarios often involve complex gas mixtures containing water vapor, sulfur compounds, and other potential intercalating species that can compete with carbonate formation and alter reaction pathways.
Existing analytical approaches primarily rely on ex-situ characterization techniques such as X-ray diffraction, thermogravimetric analysis, and infrared spectroscopy to monitor carbonation progress. While these methods provide valuable insights into the final products and overall conversion rates, they offer limited temporal resolution for capturing the dynamic aspects of the carbonation process. The lack of real-time monitoring capabilities significantly hampers efforts to establish precise kinetic parameters and understand the mechanistic pathways governing LDH carbonation.
One of the most significant challenges lies in the complexity of the carbonation mechanism itself. The process involves multiple simultaneous phenomena including CO2 diffusion through the interlayer space, anion exchange reactions, structural reorganization, and potential phase transformations. Current kinetic models often oversimplify these interactions by treating carbonation as a single-step process, failing to account for the multi-stage nature of the reaction and the interdependencies between different mechanistic steps.
Temperature and humidity effects on carbonation kinetics remain poorly quantified across different LDH compositions. While qualitative trends have been established showing accelerated carbonation at elevated temperatures and humidity levels, precise mathematical relationships describing these dependencies are lacking. This limitation severely restricts the ability to predict LDH performance in real-world applications where environmental conditions vary significantly.
The heterogeneity of LDH materials presents another fundamental challenge. Variations in particle size, crystallinity, interlayer spacing, and surface area can dramatically influence carbonation rates, yet current kinetic models rarely incorporate these structural parameters as explicit variables. This oversight leads to significant discrepancies between predicted and observed carbonation behavior, particularly when comparing different synthesis methods or LDH compositions.
Furthermore, the influence of competing anions in practical applications remains inadequately characterized. Most kinetic studies focus on pure CO2 environments, but real-world scenarios often involve complex gas mixtures containing water vapor, sulfur compounds, and other potential intercalating species that can compete with carbonate formation and alter reaction pathways.
Existing Solutions for LDH Carbonation Kinetics Analysis
01 Synthesis methods for layered double hydroxides using coprecipitation
Coprecipitation is a widely used method for synthesizing layered double hydroxides (LDHs). This method involves the simultaneous precipitation of metal cations from a solution under controlled pH conditions. The kinetics of formation can be influenced by factors such as pH, temperature, concentration of reactants, and aging time. The coprecipitation method allows for control over particle size, crystallinity, and composition of the resulting LDH materials.- Synthesis methods for layered double hydroxides using coprecipitation: Coprecipitation is a widely used method for synthesizing layered double hydroxides (LDHs). This method involves the simultaneous precipitation of metal cations from a solution under controlled pH conditions. The kinetics of formation can be influenced by factors such as pH, temperature, concentration of reactants, and stirring rate. The coprecipitation method allows for control over particle size, crystallinity, and composition of the resulting LDH materials.
- Hydrothermal synthesis and crystallization kinetics of layered double hydroxides: Hydrothermal synthesis involves the formation of LDH structures under elevated temperature and pressure conditions in aqueous solutions. This method promotes better crystallization and can produce LDH materials with enhanced structural properties. The kinetics of crystallization under hydrothermal conditions are affected by reaction time, temperature, and the presence of organic or inorganic additives that can modify the growth rate and morphology of the crystals.
- Influence of reaction parameters on LDH formation rate: The formation kinetics of layered double hydroxides are significantly influenced by various reaction parameters including temperature, pH, metal ion ratios, and aging time. Higher temperatures generally accelerate the formation process, while pH control is critical for maintaining the proper precipitation conditions. The molar ratio of divalent to trivalent metal ions affects both the rate of formation and the structural properties of the final product. Understanding these parameters is essential for optimizing synthesis protocols.
- Mechanochemical synthesis and rapid formation of layered double hydroxides: Mechanochemical synthesis represents an alternative approach for LDH formation that involves mechanical energy input through grinding or milling. This method can significantly reduce reaction times compared to conventional wet chemical methods. The kinetics of mechanochemical formation are governed by factors such as milling intensity, duration, and the presence of liquid additives. This approach offers advantages in terms of reduced solvent use and the ability to produce LDH materials with unique properties.
- In-situ monitoring and characterization of LDH formation kinetics: Advanced analytical techniques enable real-time monitoring of LDH formation processes, providing insights into nucleation and growth mechanisms. Methods such as time-resolved X-ray diffraction, spectroscopy, and particle size analysis can track the evolution of LDH structures during synthesis. Understanding the kinetic pathways through in-situ characterization allows for better control over the synthesis process and optimization of material properties for specific applications.
02 Hydrothermal synthesis and crystallization kinetics of layered double hydroxides
Hydrothermal synthesis involves the formation of LDH structures under elevated temperature and pressure conditions in aqueous solutions. This method promotes better crystallization and can control the morphology and particle size distribution of LDHs. The kinetics of crystallization under hydrothermal conditions are affected by reaction temperature, time, and the presence of structure-directing agents. This approach often results in materials with improved structural ordering and enhanced properties.Expand Specific Solutions03 Mechanochemical synthesis and formation mechanisms
Mechanochemical methods involve the use of mechanical energy to induce chemical reactions for LDH formation. This solvent-free or low-solvent approach can significantly reduce reaction times and energy consumption. The kinetics of LDH formation through mechanochemical routes depend on milling parameters such as rotation speed, milling time, and ball-to-powder ratio. This method offers advantages in terms of environmental sustainability and scalability.Expand Specific Solutions04 In-situ formation and growth kinetics in polymer matrices
In-situ formation of LDHs within polymer matrices involves the nucleation and growth of LDH structures during polymer processing or curing. The kinetics of this process are influenced by the polymer-metal ion interactions, diffusion rates, and processing conditions. This approach enables the creation of nanocomposite materials with uniformly dispersed LDH particles, leading to enhanced mechanical, thermal, and barrier properties. The formation kinetics can be monitored through various analytical techniques to optimize material properties.Expand Specific Solutions05 Calcination and reconstruction kinetics of layered double hydroxides
The calcination-reconstruction process involves heating LDHs to remove interlayer anions and water, followed by rehydration to restore the layered structure. The kinetics of both decomposition during calcination and reconstruction upon rehydration are critical for applications such as catalysis and adsorption. Factors affecting these kinetics include calcination temperature, heating rate, atmosphere, and the nature of the interlayer anions. Understanding these kinetic processes enables the design of LDH materials with tailored properties and enhanced performance in various applications.Expand Specific Solutions
Key Players in LDH and Carbonation Technology Industry
The quantification of formation kinetics in layered double hydroxides during carbonation represents an emerging research area in the early development stage, with significant potential for carbon capture and storage applications. The market is currently nascent but shows promise for substantial growth as carbon management technologies gain importance globally. Technology maturity varies significantly across different player types, with academic institutions like Beijing University of Chemical Technology, University of Valencia, and Centre National de la Recherche Scientifique leading fundamental research, while industrial players such as Saudi Arabian Oil Co., NGK Corp., and Nippon Steel Corp. focus on practical applications. Chemical companies including Kuraray Co., Akzo Nobel NV, and Dow Global Technologies LLC are developing materials and processes, indicating a transition from laboratory research toward commercial viability, though widespread industrial implementation remains several years away.
Beijing University of Chemical Technology
Technical Solution: Developed advanced in-situ characterization techniques for monitoring LDH formation kinetics during carbonation processes. Their approach combines X-ray diffraction with thermogravimetric analysis to quantify the transformation rates of Mg-Al LDHs under various CO2 concentrations and temperatures. The research focuses on understanding the nucleation and growth mechanisms of carbonate-intercalated LDH phases, with particular emphasis on the role of pH buffering and ionic strength in controlling formation rates. Their methodology enables real-time tracking of structural changes and provides quantitative models for predicting LDH stability in carbonation environments.
Strengths: Strong fundamental research capabilities and advanced analytical techniques. Weaknesses: Limited industrial-scale validation and commercialization experience.
NGK Corp.
Technical Solution: Implemented industrial-scale LDH synthesis processes with integrated carbonation kinetics monitoring for ceramic and catalyst applications. Their technology utilizes continuous flow reactors with precise CO2 injection control to achieve uniform LDH formation with predictable carbonation rates. The company has developed proprietary measurement systems that combine online pH monitoring with particle size analysis to quantify formation kinetics in real-time. Their approach focuses on optimizing reaction conditions to control the degree of carbonation and maintain desired LDH properties for specific industrial applications, particularly in automotive emission control systems.
Strengths: Industrial expertise and scalable manufacturing processes. Weaknesses: Limited research publications and proprietary nature of detailed methodologies.
Core Innovations in LDH Formation Kinetics Quantification
Preparation method for anion-exchangeable, layered double hydroxides
PatentInactiveUS20100279848A1
Innovation
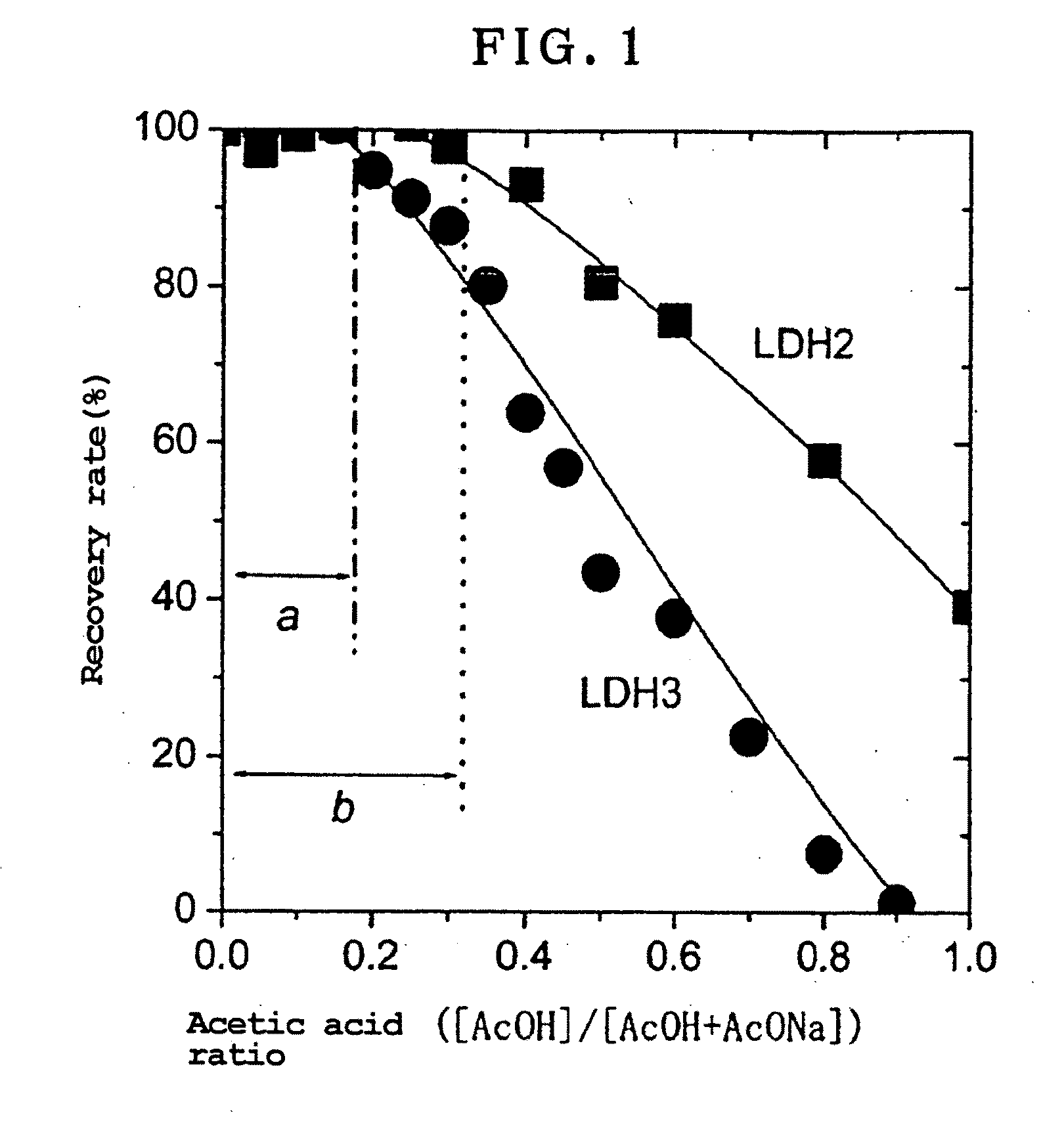
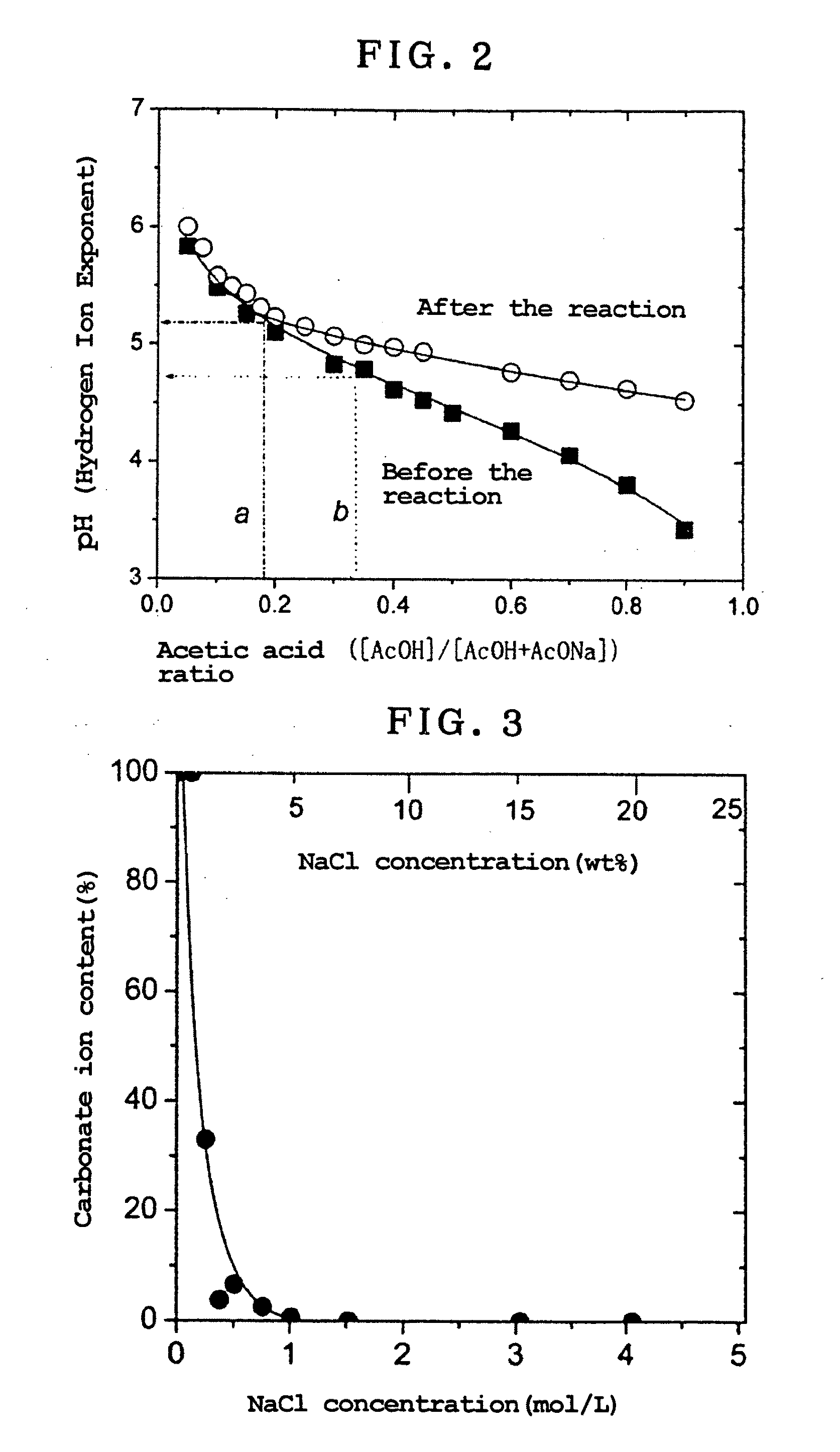
- A method involving dispersion of carbonate ion type LDH in an aqueous solution with a salt containing monovalent anions at a pH between 4 and 7, using an acetic acid buffer and NaCl, allowing for substitution of carbonate ions with chlorine ions at room temperature without significant dissolution or loss of crystallinity.
High surface area layered double hydroxides
PatentWO2015144778A1
Innovation
- A method involving rapid co-precipitation, aging, and dispersion in an aqueous miscible organic solvent to produce LDHs with specific surface areas of at least 125 m2/g, utilizing a process that includes precipitation, aging, washing, dispersion in an organic solvent, and drying, which can be scaled up cost-effectively.
Environmental Impact Assessment of LDH Carbonation
The carbonation process of layered double hydroxides presents significant environmental implications that warrant comprehensive assessment across multiple dimensions. The reaction mechanism involves CO2 sequestration from atmospheric or industrial sources, potentially contributing to carbon capture and storage strategies. During carbonation, LDH structures undergo transformation as carbonate anions intercalate between hydroxide layers, effectively immobilizing carbon dioxide in stable mineral forms.
Life cycle assessment of LDH carbonation reveals both positive and negative environmental impacts. The primary environmental benefit stems from permanent CO2 sequestration, with studies indicating capture capacities ranging from 200-400 kg CO2 per ton of LDH material. This sequestration potential positions LDH carbonation as a viable carbon mitigation technology, particularly when integrated with industrial CO2 emission sources such as cement plants or power generation facilities.
Energy consumption during LDH synthesis and carbonation processes represents a critical environmental consideration. Traditional hydrothermal synthesis methods require temperatures between 80-200°C and extended reaction times, resulting in substantial energy inputs. However, recent advances in mechanochemical synthesis and ambient temperature carbonation pathways have demonstrated potential for reducing overall energy footprints by up to 60% compared to conventional approaches.
Water resource utilization constitutes another environmental factor, as aqueous synthesis routes typically require significant water volumes for LDH preparation and subsequent carbonation reactions. The environmental burden can be mitigated through process optimization, including water recycling systems and the development of solvent-free synthesis methods that eliminate liquid waste streams entirely.
Waste generation and material sustainability aspects reveal mixed environmental outcomes. While LDH carbonation produces stable, non-toxic carbonate minerals suitable for construction applications, the synthesis process may generate alkaline wastewater requiring neutralization treatment. Additionally, the sourcing of metal precursors for LDH synthesis, particularly aluminum and magnesium compounds, introduces upstream environmental impacts related to mining and refining operations.
The scalability of LDH carbonation technology presents both opportunities and challenges for environmental impact mitigation. Large-scale deployment could significantly contribute to industrial CO2 reduction targets, yet increased production volumes may amplify resource consumption and waste generation. Regional environmental impacts vary considerably based on local energy grids, water availability, and waste management infrastructure, necessitating site-specific environmental assessments for optimal implementation strategies.
Life cycle assessment of LDH carbonation reveals both positive and negative environmental impacts. The primary environmental benefit stems from permanent CO2 sequestration, with studies indicating capture capacities ranging from 200-400 kg CO2 per ton of LDH material. This sequestration potential positions LDH carbonation as a viable carbon mitigation technology, particularly when integrated with industrial CO2 emission sources such as cement plants or power generation facilities.
Energy consumption during LDH synthesis and carbonation processes represents a critical environmental consideration. Traditional hydrothermal synthesis methods require temperatures between 80-200°C and extended reaction times, resulting in substantial energy inputs. However, recent advances in mechanochemical synthesis and ambient temperature carbonation pathways have demonstrated potential for reducing overall energy footprints by up to 60% compared to conventional approaches.
Water resource utilization constitutes another environmental factor, as aqueous synthesis routes typically require significant water volumes for LDH preparation and subsequent carbonation reactions. The environmental burden can be mitigated through process optimization, including water recycling systems and the development of solvent-free synthesis methods that eliminate liquid waste streams entirely.
Waste generation and material sustainability aspects reveal mixed environmental outcomes. While LDH carbonation produces stable, non-toxic carbonate minerals suitable for construction applications, the synthesis process may generate alkaline wastewater requiring neutralization treatment. Additionally, the sourcing of metal precursors for LDH synthesis, particularly aluminum and magnesium compounds, introduces upstream environmental impacts related to mining and refining operations.
The scalability of LDH carbonation technology presents both opportunities and challenges for environmental impact mitigation. Large-scale deployment could significantly contribute to industrial CO2 reduction targets, yet increased production volumes may amplify resource consumption and waste generation. Regional environmental impacts vary considerably based on local energy grids, water availability, and waste management infrastructure, necessitating site-specific environmental assessments for optimal implementation strategies.
Industrial Scale-up Considerations for LDH Processes
The transition from laboratory-scale LDH synthesis to industrial production presents significant engineering challenges that must be carefully addressed to maintain product quality while achieving economic viability. Scaling up LDH carbonation processes requires comprehensive consideration of reactor design, mass transfer limitations, and process control systems that can handle the complex kinetics involved in formation reactions.
Reactor design represents the most critical aspect of industrial scale-up for LDH processes. Traditional batch reactors used in laboratory settings must be replaced with continuous or semi-continuous systems capable of handling large volumes while maintaining uniform mixing and temperature control. The choice between stirred tank reactors, fluidized bed reactors, or specialized precipitation reactors depends on the specific LDH composition and desired particle characteristics. Heat management becomes increasingly challenging at industrial scale, as carbonation reactions are typically exothermic and require precise temperature control to optimize formation kinetics.
Mass transfer limitations significantly impact the scalability of LDH carbonation processes. At industrial scale, achieving uniform CO2 distribution throughout large reaction volumes becomes problematic, potentially leading to non-uniform product quality and reduced reaction efficiency. Advanced gas sparging systems, enhanced mixing technologies, and optimized reactor geometries are essential to overcome these limitations. The design must ensure adequate gas-liquid contact time while preventing excessive energy consumption that would compromise economic feasibility.
Process control and monitoring systems must be substantially more sophisticated than laboratory equivalents to handle the complexity of industrial LDH production. Real-time monitoring of pH, temperature, CO2 concentration, and particle size distribution becomes crucial for maintaining consistent product quality. Advanced process analytical technology (PAT) tools, including in-line spectroscopy and particle size analyzers, enable continuous monitoring and feedback control of formation kinetics.
Economic considerations play a decisive role in determining the viability of industrial LDH processes. Raw material costs, energy consumption, waste treatment, and equipment maintenance must be carefully balanced against product value. The development of cost-effective purification and separation techniques becomes critical, as industrial-scale processes generate larger volumes of by-products that require efficient handling.
Environmental compliance and sustainability factors add additional complexity to industrial scale-up considerations. Large-scale LDH production must meet stringent environmental regulations regarding emissions, waste disposal, and energy efficiency. Integration with carbon capture technologies or utilization of industrial CO2 waste streams can enhance the environmental profile while potentially reducing raw material costs.
Reactor design represents the most critical aspect of industrial scale-up for LDH processes. Traditional batch reactors used in laboratory settings must be replaced with continuous or semi-continuous systems capable of handling large volumes while maintaining uniform mixing and temperature control. The choice between stirred tank reactors, fluidized bed reactors, or specialized precipitation reactors depends on the specific LDH composition and desired particle characteristics. Heat management becomes increasingly challenging at industrial scale, as carbonation reactions are typically exothermic and require precise temperature control to optimize formation kinetics.
Mass transfer limitations significantly impact the scalability of LDH carbonation processes. At industrial scale, achieving uniform CO2 distribution throughout large reaction volumes becomes problematic, potentially leading to non-uniform product quality and reduced reaction efficiency. Advanced gas sparging systems, enhanced mixing technologies, and optimized reactor geometries are essential to overcome these limitations. The design must ensure adequate gas-liquid contact time while preventing excessive energy consumption that would compromise economic feasibility.
Process control and monitoring systems must be substantially more sophisticated than laboratory equivalents to handle the complexity of industrial LDH production. Real-time monitoring of pH, temperature, CO2 concentration, and particle size distribution becomes crucial for maintaining consistent product quality. Advanced process analytical technology (PAT) tools, including in-line spectroscopy and particle size analyzers, enable continuous monitoring and feedback control of formation kinetics.
Economic considerations play a decisive role in determining the viability of industrial LDH processes. Raw material costs, energy consumption, waste treatment, and equipment maintenance must be carefully balanced against product value. The development of cost-effective purification and separation techniques becomes critical, as industrial-scale processes generate larger volumes of by-products that require efficient handling.
Environmental compliance and sustainability factors add additional complexity to industrial scale-up considerations. Large-scale LDH production must meet stringent environmental regulations regarding emissions, waste disposal, and energy efficiency. Integration with carbon capture technologies or utilization of industrial CO2 waste streams can enhance the environmental profile while potentially reducing raw material costs.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!