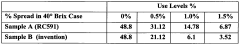
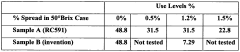
Quantify Sodium CMC's Effect on Suspension Evenness
MAR 19, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Sodium CMC Suspension Technology Background and Objectives
Sodium Carboxymethyl Cellulose (CMC) has emerged as a critical additive in suspension formulations across pharmaceutical, food, cosmetic, and industrial applications since its commercial introduction in the 1940s. This water-soluble cellulose derivative functions primarily as a rheology modifier, thickening agent, and stabilizer, fundamentally altering the physical properties of liquid systems to maintain uniform particle distribution.
The evolution of suspension technology has been driven by increasing demands for product consistency, shelf stability, and performance reliability. Traditional suspension systems often suffer from sedimentation, phase separation, and non-uniform particle distribution, leading to compromised product efficacy and consumer dissatisfaction. The introduction of hydrocolloids like sodium CMC revolutionized suspension formulation by providing effective means to control particle settling and maintain homogeneity.
Current market trends indicate growing emphasis on quantitative characterization of additive performance, moving beyond empirical formulation approaches toward data-driven optimization. Regulatory requirements across industries increasingly demand comprehensive documentation of ingredient functionality and performance metrics, particularly in pharmaceutical and food applications where product uniformity directly impacts safety and efficacy.
The primary objective of quantifying sodium CMC's effect on suspension evenness centers on establishing measurable relationships between CMC concentration, molecular weight, degree of substitution, and resulting suspension stability parameters. This quantification enables formulators to optimize CMC usage for maximum efficiency while minimizing costs and potential adverse effects on other product attributes.
Key technical objectives include developing standardized measurement protocols for suspension evenness, establishing predictive models correlating CMC properties with suspension performance, and identifying optimal CMC specifications for different suspension types. Additionally, understanding the temporal aspects of CMC's stabilizing effect allows for accurate shelf-life predictions and storage condition recommendations.
The strategic importance of this quantification extends beyond immediate formulation benefits. Precise understanding of CMC's mechanism enables development of next-generation suspension systems with enhanced performance characteristics, reduced environmental impact through optimized usage levels, and improved cost-effectiveness through targeted selection of CMC grades based on specific application requirements.
The evolution of suspension technology has been driven by increasing demands for product consistency, shelf stability, and performance reliability. Traditional suspension systems often suffer from sedimentation, phase separation, and non-uniform particle distribution, leading to compromised product efficacy and consumer dissatisfaction. The introduction of hydrocolloids like sodium CMC revolutionized suspension formulation by providing effective means to control particle settling and maintain homogeneity.
Current market trends indicate growing emphasis on quantitative characterization of additive performance, moving beyond empirical formulation approaches toward data-driven optimization. Regulatory requirements across industries increasingly demand comprehensive documentation of ingredient functionality and performance metrics, particularly in pharmaceutical and food applications where product uniformity directly impacts safety and efficacy.
The primary objective of quantifying sodium CMC's effect on suspension evenness centers on establishing measurable relationships between CMC concentration, molecular weight, degree of substitution, and resulting suspension stability parameters. This quantification enables formulators to optimize CMC usage for maximum efficiency while minimizing costs and potential adverse effects on other product attributes.
Key technical objectives include developing standardized measurement protocols for suspension evenness, establishing predictive models correlating CMC properties with suspension performance, and identifying optimal CMC specifications for different suspension types. Additionally, understanding the temporal aspects of CMC's stabilizing effect allows for accurate shelf-life predictions and storage condition recommendations.
The strategic importance of this quantification extends beyond immediate formulation benefits. Precise understanding of CMC's mechanism enables development of next-generation suspension systems with enhanced performance characteristics, reduced environmental impact through optimized usage levels, and improved cost-effectiveness through targeted selection of CMC grades based on specific application requirements.
Market Demand for Enhanced Suspension Uniformity Solutions
The pharmaceutical industry demonstrates substantial demand for enhanced suspension uniformity solutions, driven by stringent regulatory requirements and quality control standards. Pharmaceutical manufacturers face increasing pressure to ensure consistent drug distribution throughout liquid formulations, particularly for oral suspensions and injectable products where dosing accuracy directly impacts patient safety and therapeutic efficacy.
Food and beverage manufacturers represent another significant market segment seeking improved suspension technologies. The industry requires uniform distribution of particles, flavoring agents, and nutritional supplements in products ranging from protein shakes to fruit juices. Consumer expectations for consistent taste, texture, and nutritional content across batches have intensified the need for reliable suspension uniformity solutions.
The cosmetics and personal care sector exhibits growing demand for enhanced suspension formulations, particularly in products containing active ingredients, pigments, or exfoliating particles. Premium skincare brands increasingly emphasize uniform distribution of active compounds to ensure consistent product performance and consumer satisfaction. This trend has accelerated with the rise of specialized formulations targeting specific skin concerns.
Industrial applications present expanding opportunities for suspension uniformity technologies. Paint and coating manufacturers require consistent pigment distribution to achieve uniform color and coverage properties. The ceramics industry demands precise particle distribution in slurries to ensure consistent firing characteristics and final product quality.
Agricultural formulations constitute an emerging market segment where suspension uniformity directly impacts application effectiveness. Pesticide and fertilizer suspensions require uniform active ingredient distribution to ensure consistent field performance and regulatory compliance. Environmental concerns and precision agriculture trends further drive demand for optimized suspension formulations.
Market growth is accelerated by increasing automation in manufacturing processes, which demands more predictable and controllable suspension behaviors. Quality management systems and real-time monitoring technologies create additional requirements for quantifiable suspension performance metrics, supporting the adoption of advanced suspension uniformity solutions across multiple industries.
Food and beverage manufacturers represent another significant market segment seeking improved suspension technologies. The industry requires uniform distribution of particles, flavoring agents, and nutritional supplements in products ranging from protein shakes to fruit juices. Consumer expectations for consistent taste, texture, and nutritional content across batches have intensified the need for reliable suspension uniformity solutions.
The cosmetics and personal care sector exhibits growing demand for enhanced suspension formulations, particularly in products containing active ingredients, pigments, or exfoliating particles. Premium skincare brands increasingly emphasize uniform distribution of active compounds to ensure consistent product performance and consumer satisfaction. This trend has accelerated with the rise of specialized formulations targeting specific skin concerns.
Industrial applications present expanding opportunities for suspension uniformity technologies. Paint and coating manufacturers require consistent pigment distribution to achieve uniform color and coverage properties. The ceramics industry demands precise particle distribution in slurries to ensure consistent firing characteristics and final product quality.
Agricultural formulations constitute an emerging market segment where suspension uniformity directly impacts application effectiveness. Pesticide and fertilizer suspensions require uniform active ingredient distribution to ensure consistent field performance and regulatory compliance. Environmental concerns and precision agriculture trends further drive demand for optimized suspension formulations.
Market growth is accelerated by increasing automation in manufacturing processes, which demands more predictable and controllable suspension behaviors. Quality management systems and real-time monitoring technologies create additional requirements for quantifiable suspension performance metrics, supporting the adoption of advanced suspension uniformity solutions across multiple industries.
Current State and Challenges in CMC Suspension Quantification
The quantification of sodium carboxymethyl cellulose (CMC) effects on suspension evenness represents a critical challenge in modern formulation science, particularly within pharmaceutical, food, and cosmetic industries. Current analytical methodologies predominantly rely on traditional rheological measurements and visual assessment techniques, which often fail to capture the complex microstructural dynamics that govern suspension stability and uniformity.
Existing measurement approaches face significant limitations in providing real-time, quantitative data on particle distribution and settling behavior. Conventional methods such as sedimentation analysis and microscopic evaluation offer only snapshot assessments, lacking the temporal resolution necessary to understand dynamic suspension behavior. These techniques frequently struggle with reproducibility issues and operator-dependent variability, making standardized quantification extremely challenging.
The heterogeneous nature of CMC-stabilized suspensions presents additional analytical complexities. Particle size distribution, concentration gradients, and localized viscosity variations create measurement artifacts that current instrumentation cannot adequately address. Standard rheometers, while useful for bulk property assessment, fail to detect microscale heterogeneities that significantly impact overall suspension performance and stability.
Technological gaps persist in correlating CMC molecular characteristics with macroscopic suspension properties. Current analytical frameworks inadequately link polymer chain length, degree of substitution, and ionic strength effects to quantifiable suspension evenness parameters. This disconnect between molecular-level interactions and system-wide performance metrics hampers the development of predictive models for formulation optimization.
Regulatory and standardization challenges further complicate the quantification landscape. Industry lacks universally accepted protocols for defining and measuring suspension evenness, leading to inconsistent evaluation criteria across different applications. The absence of standardized metrics creates barriers for comparative analysis and quality control implementation.
Emerging measurement technologies, including dynamic light scattering and acoustic spectroscopy, show promise but remain limited by sample preparation requirements and environmental sensitivity. These advanced techniques often require specialized expertise and significant capital investment, limiting their widespread adoption in routine quality control applications.
The integration of multiple analytical techniques presents both opportunities and challenges. While multi-modal approaches can provide comprehensive suspension characterization, data fusion and interpretation complexities often outweigh the benefits, particularly in time-sensitive production environments where rapid decision-making is essential.
Existing measurement approaches face significant limitations in providing real-time, quantitative data on particle distribution and settling behavior. Conventional methods such as sedimentation analysis and microscopic evaluation offer only snapshot assessments, lacking the temporal resolution necessary to understand dynamic suspension behavior. These techniques frequently struggle with reproducibility issues and operator-dependent variability, making standardized quantification extremely challenging.
The heterogeneous nature of CMC-stabilized suspensions presents additional analytical complexities. Particle size distribution, concentration gradients, and localized viscosity variations create measurement artifacts that current instrumentation cannot adequately address. Standard rheometers, while useful for bulk property assessment, fail to detect microscale heterogeneities that significantly impact overall suspension performance and stability.
Technological gaps persist in correlating CMC molecular characteristics with macroscopic suspension properties. Current analytical frameworks inadequately link polymer chain length, degree of substitution, and ionic strength effects to quantifiable suspension evenness parameters. This disconnect between molecular-level interactions and system-wide performance metrics hampers the development of predictive models for formulation optimization.
Regulatory and standardization challenges further complicate the quantification landscape. Industry lacks universally accepted protocols for defining and measuring suspension evenness, leading to inconsistent evaluation criteria across different applications. The absence of standardized metrics creates barriers for comparative analysis and quality control implementation.
Emerging measurement technologies, including dynamic light scattering and acoustic spectroscopy, show promise but remain limited by sample preparation requirements and environmental sensitivity. These advanced techniques often require specialized expertise and significant capital investment, limiting their widespread adoption in routine quality control applications.
The integration of multiple analytical techniques presents both opportunities and challenges. While multi-modal approaches can provide comprehensive suspension characterization, data fusion and interpretation complexities often outweigh the benefits, particularly in time-sensitive production environments where rapid decision-making is essential.
Existing Methods for Measuring Suspension Evenness
01 Use of dispersing agents to improve CMC suspension evenness
Dispersing agents can be incorporated into sodium carboxymethyl cellulose (CMC) suspensions to enhance the uniformity and stability of the dispersion. These agents help prevent agglomeration and settling of CMC particles, ensuring a more homogeneous suspension. The dispersing agents work by reducing particle-particle interactions and improving the wetting properties of the CMC powder in aqueous media.- Use of dispersing agents to improve CMC suspension evenness: Dispersing agents can be incorporated into sodium carboxymethyl cellulose (CMC) suspensions to enhance the uniformity and stability of the dispersion. These agents help prevent agglomeration and settling of CMC particles, ensuring a more homogeneous suspension. The dispersing agents work by reducing particle-particle interactions and improving wetting properties, which leads to better distribution of CMC throughout the liquid medium.
- Optimization of mixing and homogenization processes: The evenness of sodium CMC suspensions can be significantly improved through optimized mixing and homogenization techniques. High-shear mixing, ultrasonic treatment, or specialized homogenization equipment can be employed to break down CMC aggregates and achieve uniform particle distribution. Process parameters such as mixing speed, duration, and temperature control are critical factors in achieving consistent suspension quality.
- Control of particle size and molecular weight distribution: The evenness of CMC suspensions is influenced by the particle size and molecular weight distribution of the sodium CMC used. Selection of appropriate CMC grades with controlled particle size ranges and narrow molecular weight distributions can enhance suspension uniformity. Manufacturing processes that ensure consistent particle characteristics contribute to improved dispersion stability and reduced sedimentation.
- Addition of stabilizers and rheology modifiers: Stabilizers and rheology modifiers can be added to sodium CMC suspensions to maintain evenness over time. These additives help control the viscosity profile and prevent phase separation or settling during storage and application. They work by modifying the suspension's flow properties and creating a stable three-dimensional network that keeps CMC particles uniformly distributed throughout the system.
- pH and ionic strength adjustment for suspension stability: The evenness of sodium CMC suspensions can be optimized by controlling pH and ionic strength of the medium. Proper adjustment of these parameters affects the degree of ionization of CMC molecules and their electrostatic interactions, which in turn influences dispersion stability. Maintaining optimal pH ranges and controlling salt concentration helps prevent flocculation and ensures uniform particle distribution throughout the suspension.
02 Optimization of mixing and homogenization processes
The evenness of sodium CMC suspensions can be significantly improved through optimized mixing and homogenization techniques. High-shear mixing, controlled agitation speeds, and specific mixing sequences help achieve better dispersion of CMC particles throughout the suspension. These process parameters ensure that the CMC is fully hydrated and evenly distributed, preventing the formation of lumps or gel particles that can affect suspension uniformity.Expand Specific Solutions03 Control of particle size distribution of CMC
The particle size and particle size distribution of sodium CMC powder directly impact the evenness of the resulting suspension. Using CMC with controlled particle size ranges or pre-treating the CMC powder through grinding or sieving can improve dispersion characteristics. Finer and more uniform particle sizes facilitate better wetting and hydration, leading to more even suspensions with reduced settling and improved stability.Expand Specific Solutions04 Addition of co-solvents and wetting agents
Co-solvents and wetting agents can be added to improve the initial dispersion and hydration of sodium CMC particles in suspension. These additives reduce the surface tension of the aqueous medium and enhance the penetration of water into CMC particles, promoting faster and more complete hydration. This approach helps eliminate dry spots and undispersed particles, resulting in a more uniform suspension with consistent viscosity throughout.Expand Specific Solutions05 Temperature and pH control during suspension preparation
The temperature and pH conditions during the preparation of sodium CMC suspensions play crucial roles in achieving optimal evenness. Controlled temperature conditions affect the hydration rate and solubility of CMC, while pH adjustment can influence the degree of ionization and swelling behavior of the polymer chains. Proper control of these parameters ensures complete dissolution, prevents premature gelation, and promotes uniform distribution of CMC throughout the suspension.Expand Specific Solutions
Key Players in CMC and Suspension Technology Industry
The sodium CMC suspension evenness technology market represents a mature industrial segment with established applications across pharmaceuticals, food processing, and manufacturing sectors. The competitive landscape is characterized by a diverse ecosystem spanning specialized chemical manufacturers like Chongqing Lihong Fine Chemicals and Shanghai Ever Bright Enterprise Development, major pharmaceutical companies including Novartis AG and Astellas Pharma, and technology-driven firms such as DKS Co. Ltd. and Merck Patent GmbH. The market demonstrates high technical maturity with companies like Dow Global Technologies and Air Liquide SA providing advanced formulation solutions, while emerging players from Asia, particularly Chinese manufacturers, are expanding production capabilities. The industry benefits from cross-sector applications, with automotive companies like Hyundai Motor and Kia Corp. utilizing CMC technologies in manufacturing processes, indicating strong market diversification and stable demand patterns across multiple end-use industries.
Novartis AG
Technical Solution: Novartis has developed pharmaceutical-grade sodium CMC formulations specifically designed for oral suspension uniformity. Their technology focuses on particle size distribution control and CMC concentration optimization to achieve consistent drug delivery. The company employs advanced analytical methods including laser diffraction and rheometry to quantify suspension evenness parameters. Their approach includes specialized CMC grades with controlled molecular weight distributions that provide optimal thickening and suspending properties for pharmaceutical applications.
Strengths: Pharmaceutical regulatory compliance and proven clinical applications. Weaknesses: Limited to pharmaceutical applications and high regulatory constraints.
Dow Global Technologies LLC
Technical Solution: Dow has developed comprehensive rheological modification technologies for sodium CMC in suspension systems. Their approach involves molecular weight optimization and degree of substitution control to achieve uniform particle distribution. The company utilizes advanced polymer chemistry to create CMC variants with specific viscosity profiles that enhance suspension stability. Their technology includes cross-linking methodologies and surface modification techniques that improve particle-matrix interactions, resulting in more homogeneous suspensions with reduced sedimentation rates.
Strengths: Extensive polymer expertise and industrial scale production capabilities. Weaknesses: High cost solutions and complex manufacturing processes.
Core Innovations in CMC Effect Quantification Techniques
Stabilizer composition of microcrystalline cellulose and carboxymethylcellulose, method for making, and uses
PatentWO2013052114A1
Innovation
- A stabilizer composition is created by blending microcrystalline cellulose with a first carboxymethyl cellulose of 0.45-0.85 degree of substitution and a second carboxymethyl cellulose of 0.9-1.5 degree of substitution, followed by extrusion and drying, to achieve improved rheological properties and dispersion stability.
Aqueous personal care compositions comprising carboxymethyl cellulose (CMC) having an optimized degree of substitution
PatentPendingUS20240216256A1
Innovation
- An aqueous personal care composition incorporating low-substituted carboxymethyl cellulose (CMC) with a degree of substitution between 0.5 and 0.7, combined with nature-derived or fossil-derived polymers and personal care additives, to enhance solubility and stability while maintaining biodegradability.
Food Safety Regulations for CMC Usage in Suspensions
The regulatory landscape for sodium carboxymethyl cellulose (CMC) usage in food suspensions is governed by comprehensive frameworks established by major food safety authorities worldwide. The U.S. Food and Drug Administration (FDA) classifies sodium CMC as Generally Recognized as Safe (GRAS) under 21 CFR 182.1745, permitting its use as a stabilizer, thickener, and emulsifier in various food applications. The European Food Safety Authority (EFSA) has approved CMC as food additive E466, with specific purity criteria and usage limitations outlined in Commission Regulation (EU) No 231/2012.
Maximum permitted levels vary significantly across different suspension categories. For beverage suspensions, regulatory limits typically range from 0.1% to 0.5% by weight, while pharmaceutical suspensions may allow higher concentrations up to 2% under specific conditions. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) has established an Acceptable Daily Intake (ADI) of 0-25 mg/kg body weight, which directly influences formulation constraints for suspension products.
Purity specifications mandate that food-grade sodium CMC must contain minimum 99.5% pure cellulose derivative, with strict limits on heavy metals including lead (≤2 mg/kg), mercury (≤1 mg/kg), and arsenic (≤3 mg/kg). Microbiological standards require total plate count below 1000 CFU/g and absence of pathogenic organisms such as Salmonella and E. coli.
Labeling requirements across jurisdictions mandate clear declaration of CMC presence, with some regions requiring specific allergen warnings despite CMC's non-allergenic nature. Recent regulatory trends indicate increasing scrutiny on nanoparticle formation in CMC-stabilized suspensions, with emerging guidelines addressing particle size distribution monitoring and potential bioavailability implications.
Compliance documentation must include comprehensive analytical certificates, stability studies demonstrating suspension uniformity over shelf life, and migration testing for packaging interactions. These regulatory frameworks directly impact the quantification methodologies for suspension evenness, as testing protocols must align with official analytical methods recognized by respective food safety authorities.
Maximum permitted levels vary significantly across different suspension categories. For beverage suspensions, regulatory limits typically range from 0.1% to 0.5% by weight, while pharmaceutical suspensions may allow higher concentrations up to 2% under specific conditions. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) has established an Acceptable Daily Intake (ADI) of 0-25 mg/kg body weight, which directly influences formulation constraints for suspension products.
Purity specifications mandate that food-grade sodium CMC must contain minimum 99.5% pure cellulose derivative, with strict limits on heavy metals including lead (≤2 mg/kg), mercury (≤1 mg/kg), and arsenic (≤3 mg/kg). Microbiological standards require total plate count below 1000 CFU/g and absence of pathogenic organisms such as Salmonella and E. coli.
Labeling requirements across jurisdictions mandate clear declaration of CMC presence, with some regions requiring specific allergen warnings despite CMC's non-allergenic nature. Recent regulatory trends indicate increasing scrutiny on nanoparticle formation in CMC-stabilized suspensions, with emerging guidelines addressing particle size distribution monitoring and potential bioavailability implications.
Compliance documentation must include comprehensive analytical certificates, stability studies demonstrating suspension uniformity over shelf life, and migration testing for packaging interactions. These regulatory frameworks directly impact the quantification methodologies for suspension evenness, as testing protocols must align with official analytical methods recognized by respective food safety authorities.
Quality Control Standards for Industrial Suspension Applications
Establishing comprehensive quality control standards for industrial suspension applications requires a systematic approach to measuring and maintaining suspension evenness when sodium carboxymethyl cellulose (CMC) is utilized as a stabilizing agent. These standards must encompass both quantitative metrics and standardized testing protocols to ensure consistent product performance across different manufacturing environments.
The foundation of quality control lies in defining measurable parameters for suspension uniformity. Particle size distribution analysis serves as a primary metric, typically measured using laser diffraction or dynamic light scattering techniques. The coefficient of variation for particle distribution should not exceed 15% for most industrial applications, while critical applications may require tighter tolerances below 10%. Sedimentation rate measurements provide another crucial parameter, with acceptable rates varying from 0.1 to 2.0 mm/hour depending on the specific application requirements.
Viscosity profiling represents a critical quality control dimension, as sodium CMC concentration directly influences rheological properties. Standard testing protocols should include measurements at multiple shear rates ranging from 0.1 to 1000 s⁻¹ using rotational rheometers. The viscosity-shear rate relationship must demonstrate consistent pseudoplastic behavior, with viscosity values falling within predetermined ranges at specific CMC concentrations.
Temporal stability assessment forms an integral component of quality standards. Suspension samples must undergo accelerated aging tests at elevated temperatures (40-60°C) for periods ranging from 7 to 30 days. During these tests, particle size distribution, viscosity, and visual appearance should remain within acceptable limits. Phase separation, if any, should not exceed 2% by volume after the specified aging period.
Standardized sampling procedures ensure representative quality assessment across production batches. Multiple sampling points throughout the suspension volume, combined with proper mixing protocols before testing, minimize measurement variability. Statistical process control charts tracking key parameters enable real-time quality monitoring and early detection of process deviations.
Documentation requirements include detailed test procedures, acceptance criteria, and corrective action protocols. Regular calibration of testing equipment and validation of analytical methods maintain measurement accuracy and traceability, ensuring consistent quality standards implementation across different production facilities and time periods.
The foundation of quality control lies in defining measurable parameters for suspension uniformity. Particle size distribution analysis serves as a primary metric, typically measured using laser diffraction or dynamic light scattering techniques. The coefficient of variation for particle distribution should not exceed 15% for most industrial applications, while critical applications may require tighter tolerances below 10%. Sedimentation rate measurements provide another crucial parameter, with acceptable rates varying from 0.1 to 2.0 mm/hour depending on the specific application requirements.
Viscosity profiling represents a critical quality control dimension, as sodium CMC concentration directly influences rheological properties. Standard testing protocols should include measurements at multiple shear rates ranging from 0.1 to 1000 s⁻¹ using rotational rheometers. The viscosity-shear rate relationship must demonstrate consistent pseudoplastic behavior, with viscosity values falling within predetermined ranges at specific CMC concentrations.
Temporal stability assessment forms an integral component of quality standards. Suspension samples must undergo accelerated aging tests at elevated temperatures (40-60°C) for periods ranging from 7 to 30 days. During these tests, particle size distribution, viscosity, and visual appearance should remain within acceptable limits. Phase separation, if any, should not exceed 2% by volume after the specified aging period.
Standardized sampling procedures ensure representative quality assessment across production batches. Multiple sampling points throughout the suspension volume, combined with proper mixing protocols before testing, minimize measurement variability. Statistical process control charts tracking key parameters enable real-time quality monitoring and early detection of process deviations.
Documentation requirements include detailed test procedures, acceptance criteria, and corrective action protocols. Regular calibration of testing equipment and validation of analytical methods maintain measurement accuracy and traceability, ensuring consistent quality standards implementation across different production facilities and time periods.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!