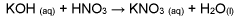Sodium Nitrate vs Ammonium Nitrate: Efficiency Analysis
MAR 3, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Nitrate Fertilizer Technology Background and Objectives
Nitrate-based fertilizers have been fundamental to agricultural productivity since the early 20th century, with sodium nitrate and ammonium nitrate emerging as two primary nitrogen sources for crop nutrition. The development of these compounds traces back to the Chilean saltpeter industry for sodium nitrate and the Haber-Bosch process advancement for ammonium nitrate synthesis. Both fertilizers have evolved through decades of agricultural application, with their usage patterns shaped by regional availability, crop requirements, and economic considerations.
The historical trajectory of nitrate fertilizer technology demonstrates a continuous evolution from natural mineral extraction to sophisticated synthetic production methods. Sodium nitrate, initially harvested from natural deposits in Chile, represented the first large-scale commercial nitrogen fertilizer. Subsequently, the industrial synthesis of ammonium nitrate through ammonia oxidation processes revolutionized nitrogen fertilizer production, enabling mass manufacturing and global distribution.
Current technological trends in nitrate fertilizer development focus on optimizing nutrient delivery efficiency, minimizing environmental impact, and enhancing crop-specific formulations. The industry has witnessed significant advancements in controlled-release mechanisms, precision application technologies, and integrated nutrient management systems. These developments reflect growing demands for sustainable agricultural practices and improved fertilizer utilization rates.
The primary objective of comparative efficiency analysis between sodium nitrate and ammonium nitrate centers on determining optimal nitrogen delivery performance across diverse agricultural applications. This evaluation encompasses nutrient availability kinetics, soil interaction mechanisms, crop uptake efficiency, and environmental fate characteristics. Understanding these comparative advantages enables informed decision-making for specific cropping systems and environmental conditions.
Secondary objectives include assessing economic viability, storage stability, application convenience, and compatibility with modern precision agriculture technologies. The analysis aims to establish clear performance benchmarks that guide fertilizer selection based on crop type, soil characteristics, climatic conditions, and farming practices. These insights support the development of targeted fertilizer recommendations and improved nutrient management strategies.
The technological framework for this efficiency analysis integrates agronomic performance metrics, environmental impact assessments, and economic feasibility studies to provide comprehensive evaluation criteria for nitrate fertilizer selection and optimization.
The historical trajectory of nitrate fertilizer technology demonstrates a continuous evolution from natural mineral extraction to sophisticated synthetic production methods. Sodium nitrate, initially harvested from natural deposits in Chile, represented the first large-scale commercial nitrogen fertilizer. Subsequently, the industrial synthesis of ammonium nitrate through ammonia oxidation processes revolutionized nitrogen fertilizer production, enabling mass manufacturing and global distribution.
Current technological trends in nitrate fertilizer development focus on optimizing nutrient delivery efficiency, minimizing environmental impact, and enhancing crop-specific formulations. The industry has witnessed significant advancements in controlled-release mechanisms, precision application technologies, and integrated nutrient management systems. These developments reflect growing demands for sustainable agricultural practices and improved fertilizer utilization rates.
The primary objective of comparative efficiency analysis between sodium nitrate and ammonium nitrate centers on determining optimal nitrogen delivery performance across diverse agricultural applications. This evaluation encompasses nutrient availability kinetics, soil interaction mechanisms, crop uptake efficiency, and environmental fate characteristics. Understanding these comparative advantages enables informed decision-making for specific cropping systems and environmental conditions.
Secondary objectives include assessing economic viability, storage stability, application convenience, and compatibility with modern precision agriculture technologies. The analysis aims to establish clear performance benchmarks that guide fertilizer selection based on crop type, soil characteristics, climatic conditions, and farming practices. These insights support the development of targeted fertilizer recommendations and improved nutrient management strategies.
The technological framework for this efficiency analysis integrates agronomic performance metrics, environmental impact assessments, and economic feasibility studies to provide comprehensive evaluation criteria for nitrate fertilizer selection and optimization.
Agricultural Market Demand for Nitrate-Based Fertilizers
The global agricultural fertilizer market demonstrates substantial demand for nitrate-based fertilizers, driven by increasing food production requirements and the need for enhanced crop yields. Nitrate fertilizers, including both sodium nitrate and ammonium nitrate, occupy a significant portion of the nitrogen fertilizer segment, which represents the largest category within the global fertilizer market. The demand is particularly pronounced in regions with intensive agricultural practices, including North America, Europe, and Asia-Pacific.
Market dynamics reveal distinct preferences for different nitrate formulations based on regional agricultural practices and crop types. Ammonium nitrate maintains strong demand in temperate regions where cereal crops dominate, particularly for wheat, corn, and barley production. Its dual nitrogen source provides both immediate and sustained nutrient release, making it highly valued among commercial farmers seeking optimal yield outcomes.
Sodium nitrate experiences growing demand in specialty crop markets, including vegetable production, fruit cultivation, and greenhouse operations. The increasing trend toward high-value crop production and precision agriculture has expanded the market for sodium nitrate, particularly in regions focusing on export-oriented agriculture and organic farming transitions.
Regional demand patterns show significant variation based on agricultural infrastructure and regulatory environments. European markets demonstrate sophisticated demand for both nitrate types, with strict quality standards driving premium product segments. Asian markets, particularly China and India, show rapidly expanding demand driven by agricultural modernization and food security initiatives.
The horticultural sector represents an emerging high-growth segment for nitrate-based fertilizers. Greenhouse cultivation, urban farming, and controlled environment agriculture create specialized demand for precise nutrient formulations. This sector particularly values the immediate availability and predictable performance characteristics of nitrate fertilizers.
Market demand is increasingly influenced by sustainability considerations and environmental regulations. Farmers seek fertilizer solutions that maximize nutrient use efficiency while minimizing environmental impact. This trend drives demand for enhanced-efficiency nitrate formulations and precision application technologies.
The specialty fertilizer segment within the nitrate market shows robust growth potential, driven by increasing adoption of fertigation systems and liquid fertilizer applications. Both sodium nitrate and ammonium nitrate serve important roles in these advanced delivery systems, with market demand reflecting the specific solubility and compatibility requirements of modern irrigation infrastructure.
Market dynamics reveal distinct preferences for different nitrate formulations based on regional agricultural practices and crop types. Ammonium nitrate maintains strong demand in temperate regions where cereal crops dominate, particularly for wheat, corn, and barley production. Its dual nitrogen source provides both immediate and sustained nutrient release, making it highly valued among commercial farmers seeking optimal yield outcomes.
Sodium nitrate experiences growing demand in specialty crop markets, including vegetable production, fruit cultivation, and greenhouse operations. The increasing trend toward high-value crop production and precision agriculture has expanded the market for sodium nitrate, particularly in regions focusing on export-oriented agriculture and organic farming transitions.
Regional demand patterns show significant variation based on agricultural infrastructure and regulatory environments. European markets demonstrate sophisticated demand for both nitrate types, with strict quality standards driving premium product segments. Asian markets, particularly China and India, show rapidly expanding demand driven by agricultural modernization and food security initiatives.
The horticultural sector represents an emerging high-growth segment for nitrate-based fertilizers. Greenhouse cultivation, urban farming, and controlled environment agriculture create specialized demand for precise nutrient formulations. This sector particularly values the immediate availability and predictable performance characteristics of nitrate fertilizers.
Market demand is increasingly influenced by sustainability considerations and environmental regulations. Farmers seek fertilizer solutions that maximize nutrient use efficiency while minimizing environmental impact. This trend drives demand for enhanced-efficiency nitrate formulations and precision application technologies.
The specialty fertilizer segment within the nitrate market shows robust growth potential, driven by increasing adoption of fertigation systems and liquid fertilizer applications. Both sodium nitrate and ammonium nitrate serve important roles in these advanced delivery systems, with market demand reflecting the specific solubility and compatibility requirements of modern irrigation infrastructure.
Current Status and Challenges in Nitrate Fertilizer Production
The global nitrate fertilizer industry currently operates within a complex landscape characterized by significant production capacity and technological diversity. Sodium nitrate and ammonium nitrate represent two primary nitrogen sources, each with distinct manufacturing processes and efficiency profiles. Current global production capacity for nitrate fertilizers exceeds 180 million metric tons annually, with ammonium nitrate accounting for approximately 60% of total nitrate fertilizer production, while sodium nitrate maintains a smaller but strategically important market share of roughly 15%.
Manufacturing efficiency varies considerably between these two compounds due to fundamental differences in production methodologies. Ammonium nitrate production relies heavily on the Haber-Bosch process for ammonia synthesis, followed by nitric acid production and neutralization reactions. This process typically achieves energy efficiency rates of 28-32 GJ per metric ton, though modern plants with advanced heat recovery systems can reduce this to 26-28 GJ per metric ton. Conversely, sodium nitrate production primarily depends on natural deposits in Chile and synthetic production through sodium hydroxide neutralization with nitric acid, presenting different efficiency parameters.
Several critical challenges currently constrain optimal efficiency in nitrate fertilizer production. Energy consumption remains the most significant bottleneck, particularly for ammonium nitrate synthesis, where natural gas costs can represent 70-80% of total production expenses. Price volatility in natural gas markets directly impacts production economics, creating uncertainty in long-term efficiency optimization investments. Additionally, environmental regulations increasingly demand lower carbon footprints, pushing manufacturers toward more energy-efficient processes and alternative feedstock sources.
Technical limitations in existing production infrastructure present another major challenge. Many facilities operate with legacy equipment that cannot achieve optimal energy integration or process intensification. Heat recovery systems in older plants typically capture only 60-70% of available waste heat, compared to 85-90% in state-of-the-art facilities. This efficiency gap represents substantial improvement potential but requires significant capital investment for retrofitting or replacement.
Geographical distribution of production capacity creates additional complexity in efficiency analysis. Ammonium nitrate production is globally distributed with major facilities in China, Russia, India, and North America, while sodium nitrate production remains concentrated in Chile's Atacama Desert, creating different logistical and supply chain efficiency considerations. Transportation costs and storage requirements vary significantly between these compounds, affecting overall system efficiency beyond manufacturing processes.
Regulatory constraints, particularly safety regulations governing ammonium nitrate handling and storage, impose additional operational costs and efficiency limitations. These requirements necessitate specialized equipment, enhanced safety systems, and specific storage protocols that can reduce overall operational efficiency while ensuring compliance with international safety standards.
Manufacturing efficiency varies considerably between these two compounds due to fundamental differences in production methodologies. Ammonium nitrate production relies heavily on the Haber-Bosch process for ammonia synthesis, followed by nitric acid production and neutralization reactions. This process typically achieves energy efficiency rates of 28-32 GJ per metric ton, though modern plants with advanced heat recovery systems can reduce this to 26-28 GJ per metric ton. Conversely, sodium nitrate production primarily depends on natural deposits in Chile and synthetic production through sodium hydroxide neutralization with nitric acid, presenting different efficiency parameters.
Several critical challenges currently constrain optimal efficiency in nitrate fertilizer production. Energy consumption remains the most significant bottleneck, particularly for ammonium nitrate synthesis, where natural gas costs can represent 70-80% of total production expenses. Price volatility in natural gas markets directly impacts production economics, creating uncertainty in long-term efficiency optimization investments. Additionally, environmental regulations increasingly demand lower carbon footprints, pushing manufacturers toward more energy-efficient processes and alternative feedstock sources.
Technical limitations in existing production infrastructure present another major challenge. Many facilities operate with legacy equipment that cannot achieve optimal energy integration or process intensification. Heat recovery systems in older plants typically capture only 60-70% of available waste heat, compared to 85-90% in state-of-the-art facilities. This efficiency gap represents substantial improvement potential but requires significant capital investment for retrofitting or replacement.
Geographical distribution of production capacity creates additional complexity in efficiency analysis. Ammonium nitrate production is globally distributed with major facilities in China, Russia, India, and North America, while sodium nitrate production remains concentrated in Chile's Atacama Desert, creating different logistical and supply chain efficiency considerations. Transportation costs and storage requirements vary significantly between these compounds, affecting overall system efficiency beyond manufacturing processes.
Regulatory constraints, particularly safety regulations governing ammonium nitrate handling and storage, impose additional operational costs and efficiency limitations. These requirements necessitate specialized equipment, enhanced safety systems, and specific storage protocols that can reduce overall operational efficiency while ensuring compliance with international safety standards.
Current Technical Solutions for Nitrate Fertilizer Efficiency
01 Comparative efficiency in fertilizer applications
Studies comparing sodium nitrate and ammonium nitrate as fertilizers demonstrate differences in nitrogen availability and uptake efficiency by plants. Ammonium nitrate typically shows faster nitrogen release and better absorption rates in various soil conditions, while sodium nitrate provides more stable nitrogen delivery over extended periods. The choice between these compounds depends on soil pH, crop type, and environmental conditions.- Comparative efficiency in fertilizer applications: Studies comparing sodium nitrate and ammonium nitrate as fertilizers demonstrate differences in nitrogen availability and uptake efficiency by plants. Ammonium nitrate typically shows faster absorption rates due to its dual nitrogen forms, while sodium nitrate provides immediate nitrate availability but may affect soil sodium levels. The efficiency varies based on soil type, crop requirements, and environmental conditions.
- Manufacturing and production efficiency: The production processes for these two nitrate compounds differ significantly in terms of energy requirements, raw material costs, and yield efficiency. Manufacturing methods impact the overall economic viability and industrial-scale production capabilities. Process optimization techniques have been developed to improve conversion rates and reduce production costs for both compounds.
- Explosive and industrial applications: Both compounds serve as oxidizing agents in various industrial applications, with distinct performance characteristics. Their detonation properties, stability, and energy release rates differ, making each suitable for specific applications. Safety considerations and handling requirements vary between the two compounds based on their chemical properties and reactivity profiles.
- Storage stability and hygroscopic properties: The two nitrates exhibit different moisture absorption characteristics and storage stability profiles. Ammonium nitrate is more hygroscopic and requires specific storage conditions to prevent caking and degradation. Sodium nitrate demonstrates better storage stability under various humidity conditions. These properties affect handling procedures, packaging requirements, and shelf life in commercial applications.
- Environmental impact and soil chemistry effects: The environmental implications of using these nitrates differ in terms of soil pH modification, groundwater contamination potential, and long-term soil health effects. Sodium nitrate can lead to sodium accumulation in soil, affecting soil structure and salinity levels. Ammonium nitrate influences soil acidity and microbial activity differently. Understanding these effects is crucial for sustainable agricultural practices and environmental management.
02 Explosive properties and industrial applications
The two nitrates exhibit distinct characteristics in explosive compositions and industrial uses. Ammonium nitrate demonstrates higher energy density and sensitivity in explosive formulations, making it more suitable for certain blasting applications. Sodium nitrate offers greater stability and is often used in combination with other compounds to achieve desired detonation properties. Manufacturing processes and safety considerations differ significantly between the two compounds.Expand Specific Solutions03 Solubility and hygroscopic behavior
Sodium nitrate and ammonium nitrate display different solubility characteristics and moisture absorption properties. Ammonium nitrate exhibits higher hygroscopicity, requiring special storage conditions to prevent caking and degradation. Sodium nitrate shows better stability in humid environments and maintains its physical properties over longer storage periods. These differences impact handling, storage requirements, and application methods in various industries.Expand Specific Solutions04 Production methods and cost efficiency
Manufacturing processes for these nitrates involve different raw materials and production techniques, affecting overall cost efficiency. Ammonium nitrate production typically utilizes ammonia and nitric acid through neutralization reactions, while sodium nitrate can be synthesized from sodium compounds or extracted from natural deposits. Energy requirements, yield rates, and production scale economics vary between the two processes, influencing their commercial viability for different applications.Expand Specific Solutions05 Environmental impact and decomposition characteristics
The environmental behavior and decomposition patterns of sodium nitrate and ammonium nitrate differ significantly. Ammonium nitrate undergoes thermal decomposition at lower temperatures and can contribute to soil acidification through nitrification processes. Sodium nitrate shows greater thermal stability and different leaching characteristics in soil systems. Their respective impacts on groundwater contamination, atmospheric emissions, and long-term soil health vary based on application rates and environmental conditions.Expand Specific Solutions
Major Players in Nitrate Fertilizer Manufacturing Industry
The sodium nitrate versus ammonium nitrate efficiency analysis represents a mature market segment within the broader fertilizer and chemical industry, currently valued at several billion dollars globally. The industry is in a consolidation phase, with established players like Yara International ASA and thyssenkrupp AG dominating production capabilities alongside regional manufacturers such as Deepak Nitrite Ltd. and Qinghai Salt Lake Industry Co., Ltd. Technology maturity varies significantly across the competitive landscape - while traditional production methods are well-established among companies like Tessenderlo Group NV and Kingenta Ecological Engineering Group Co., Ltd., emerging players like Atmonia ehf are developing innovative electrolyser technologies for sustainable ammonia production. Industrial giants including Hitachi Ltd., Resonac Holdings Corp., and AGC Inc. contribute advanced process optimization and materials science expertise, while specialized chemical manufacturers such as AdvanSix Resins & Chemicals LLC and Hubei Sanning Chemical Co., Ltd. focus on application-specific formulations and efficiency improvements in nitrate production processes.
AdvanSix Resins & Chemicals LLC
Technical Solution: AdvanSix has developed integrated production processes for ammonium nitrate focusing on industrial applications and fertilizer markets. Their technology emphasizes process optimization through advanced reactor design and heat recovery systems, achieving energy efficiency improvements of approximately 12-15% compared to conventional methods. The company's ammonium nitrate production features enhanced safety protocols and automated control systems that maintain consistent product quality with nitrogen content exceeding 34%. Their comparative analysis shows ammonium nitrate provides superior cost-effectiveness in neutral to acidic soils, while acknowledging sodium nitrate's advantages in specific crop applications requiring immediate nitrogen availability and reduced soil acidification risks.
Strengths: Integrated production capabilities, strong safety record, cost-effective manufacturing processes. Weaknesses: Limited global presence compared to major competitors, dependency on North American markets.
Honeywell International Technologies Ltd.
Technical Solution: Honeywell has developed advanced process control and safety technologies for nitrate production facilities, focusing on optimization of both sodium nitrate and ammonium nitrate manufacturing processes. Their integrated control systems utilize predictive analytics and real-time monitoring to optimize reaction conditions, achieving production efficiency improvements of 8-12%. For ammonium nitrate production, their safety systems include advanced temperature monitoring and automated emergency response protocols, addressing the compound's thermal sensitivity. Honeywell's process optimization studies demonstrate that ammonium nitrate production can achieve higher throughput rates with lower energy consumption per unit of nitrogen output. Their technology also addresses sodium nitrate production challenges, including crystallization control and impurity management, ensuring consistent product quality for both agricultural and industrial applications while maintaining strict safety standards.
Strengths: Advanced automation and control systems, strong safety technology portfolio, proven industrial implementation. Weaknesses: Limited direct production experience, higher technology licensing costs.
Core Patents in Nitrate Fertilizer Optimization Technologies
A Method for the Manufacture of Pure Ammonium Nitrate from Sodium Nitrate and Ammonium Sulphate.
PatentInactiveGB190619465A
Innovation
- The process involves creating a mixed solution with excess sodium nitrate, evaporating to saturation with ammonium nitrate, and then cooling to precipitate out sodium sulphate and sodium nitrate, followed by dilution to keep these salts in solution, allowing for the separation of pure ammonium nitrate.
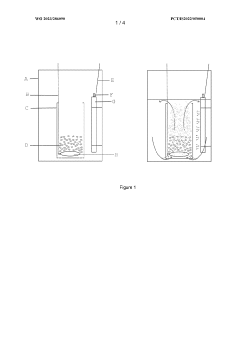
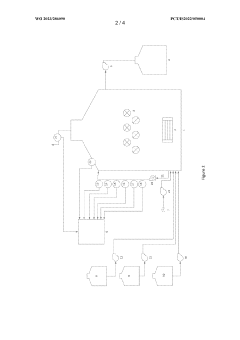
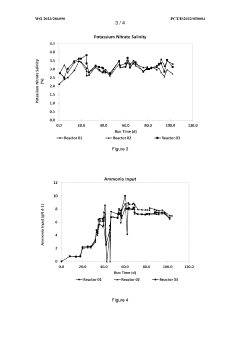
Bioreactor system and process for nitrate production
PatentWO2023286090A1
Innovation
- A bioreactor system and process that uses halophilic nitrifier microorganisms to continuously convert ammonia to nitrate, producing high-concentration nitrate solutions with reduced greenhouse emissions, suitable for both small-scale and large-scale operations, including on-site use at individual farms, utilizing a well-defined aqueous reactor medium and immobilized nitrifier organisms in various reactor types.
Environmental Regulations for Nitrate Fertilizer Production
The regulatory landscape governing nitrate fertilizer production has evolved significantly over the past two decades, driven by growing environmental concerns and scientific understanding of nitrogen's impact on ecosystems. Both sodium nitrate and ammonium nitrate production facilities must navigate an increasingly complex web of environmental regulations that vary by jurisdiction but share common objectives of minimizing air emissions, water contamination, and soil degradation.
Air quality regulations represent the most stringent aspect of nitrate fertilizer production oversight. The manufacturing process for both compounds generates nitrogen oxides (NOx), particulate matter, and ammonia emissions that are subject to strict limits under frameworks such as the Clean Air Act in the United States and the Industrial Emissions Directive in Europe. Ammonium nitrate production typically faces more rigorous scrutiny due to higher NOx emission rates during the neutralization process, requiring advanced selective catalytic reduction systems and continuous emission monitoring.
Water discharge regulations impose comprehensive restrictions on effluent quality from nitrate fertilizer facilities. The primary concerns include nitrate and nitrite concentrations in wastewater, which can contribute to eutrophication in receiving water bodies. Sodium nitrate production often generates higher volumes of process wastewater due to purification requirements, necessitating advanced treatment systems including biological nutrient removal and reverse osmosis technologies to meet discharge standards.
Waste management regulations address the handling and disposal of production byproducts and contaminated materials. Both sodium nitrate and ammonium nitrate facilities must comply with hazardous waste classification systems, with particular attention to materials containing heavy metals or persistent organic compounds. The regulatory framework requires comprehensive waste characterization, approved disposal methods, and long-term monitoring of disposal sites.
Recent regulatory trends indicate increasing emphasis on lifecycle environmental impact assessment and circular economy principles. New regulations are emerging that require fertilizer manufacturers to demonstrate sustainable sourcing of raw materials, energy efficiency improvements, and integration of waste-to-resource conversion technologies. These developments are reshaping production strategies and driving innovation in cleaner production technologies for both sodium nitrate and ammonium nitrate manufacturing processes.
Air quality regulations represent the most stringent aspect of nitrate fertilizer production oversight. The manufacturing process for both compounds generates nitrogen oxides (NOx), particulate matter, and ammonia emissions that are subject to strict limits under frameworks such as the Clean Air Act in the United States and the Industrial Emissions Directive in Europe. Ammonium nitrate production typically faces more rigorous scrutiny due to higher NOx emission rates during the neutralization process, requiring advanced selective catalytic reduction systems and continuous emission monitoring.
Water discharge regulations impose comprehensive restrictions on effluent quality from nitrate fertilizer facilities. The primary concerns include nitrate and nitrite concentrations in wastewater, which can contribute to eutrophication in receiving water bodies. Sodium nitrate production often generates higher volumes of process wastewater due to purification requirements, necessitating advanced treatment systems including biological nutrient removal and reverse osmosis technologies to meet discharge standards.
Waste management regulations address the handling and disposal of production byproducts and contaminated materials. Both sodium nitrate and ammonium nitrate facilities must comply with hazardous waste classification systems, with particular attention to materials containing heavy metals or persistent organic compounds. The regulatory framework requires comprehensive waste characterization, approved disposal methods, and long-term monitoring of disposal sites.
Recent regulatory trends indicate increasing emphasis on lifecycle environmental impact assessment and circular economy principles. New regulations are emerging that require fertilizer manufacturers to demonstrate sustainable sourcing of raw materials, energy efficiency improvements, and integration of waste-to-resource conversion technologies. These developments are reshaping production strategies and driving innovation in cleaner production technologies for both sodium nitrate and ammonium nitrate manufacturing processes.
Safety Standards and Risk Management in Nitrate Handling
The handling of sodium nitrate and ammonium nitrate requires adherence to stringent safety standards due to their inherent chemical properties and potential hazards. Both compounds are classified as oxidizing agents under international chemical classification systems, with ammonium nitrate presenting higher explosion risks due to its thermal instability and sensitivity to contamination. Regulatory frameworks such as OSHA standards in the United States and REACH regulations in Europe mandate specific storage, transportation, and handling protocols for these materials.
Storage facility requirements differ significantly between the two nitrates. Sodium nitrate storage facilities must maintain temperatures below 380°C to prevent decomposition, while ammonium nitrate requires more stringent controls with temperature limits below 210°C. Fire suppression systems must be specifically designed for oxidizer fires, typically employing water-based systems rather than traditional chemical suppressants that may react adversely with nitrate compounds.
Personnel safety protocols encompass comprehensive training programs covering chemical properties, emergency response procedures, and proper use of personal protective equipment. Workers handling these materials must be equipped with appropriate respiratory protection, chemical-resistant clothing, and eye protection. Regular health monitoring is essential, particularly for employees with prolonged exposure to nitrate dust or vapors.
Transportation safety standards follow UN Model Regulations for dangerous goods, with both compounds classified under Class 5.1 oxidizing substances. Ammonium nitrate faces additional restrictions under various national security regulations due to its potential misuse in explosive devices. Specialized transport containers, route planning, and driver certification requirements ensure safe movement of these materials through supply chains.
Emergency response planning constitutes a critical component of risk management frameworks. Facilities must maintain detailed emergency action plans addressing potential scenarios including fire, explosion, chemical spills, and exposure incidents. Coordination with local emergency services, establishment of evacuation procedures, and maintenance of specialized firefighting equipment designed for oxidizer incidents are mandatory requirements.
Risk assessment methodologies employ quantitative analysis techniques to evaluate potential failure modes and their consequences. Hazard and operability studies, fault tree analysis, and consequence modeling help identify critical control points and establish appropriate safety margins. Regular safety audits and compliance monitoring ensure continuous improvement of safety management systems and adherence to evolving regulatory requirements.
Storage facility requirements differ significantly between the two nitrates. Sodium nitrate storage facilities must maintain temperatures below 380°C to prevent decomposition, while ammonium nitrate requires more stringent controls with temperature limits below 210°C. Fire suppression systems must be specifically designed for oxidizer fires, typically employing water-based systems rather than traditional chemical suppressants that may react adversely with nitrate compounds.
Personnel safety protocols encompass comprehensive training programs covering chemical properties, emergency response procedures, and proper use of personal protective equipment. Workers handling these materials must be equipped with appropriate respiratory protection, chemical-resistant clothing, and eye protection. Regular health monitoring is essential, particularly for employees with prolonged exposure to nitrate dust or vapors.
Transportation safety standards follow UN Model Regulations for dangerous goods, with both compounds classified under Class 5.1 oxidizing substances. Ammonium nitrate faces additional restrictions under various national security regulations due to its potential misuse in explosive devices. Specialized transport containers, route planning, and driver certification requirements ensure safe movement of these materials through supply chains.
Emergency response planning constitutes a critical component of risk management frameworks. Facilities must maintain detailed emergency action plans addressing potential scenarios including fire, explosion, chemical spills, and exposure incidents. Coordination with local emergency services, establishment of evacuation procedures, and maintenance of specialized firefighting equipment designed for oxidizer incidents are mandatory requirements.
Risk assessment methodologies employ quantitative analysis techniques to evaluate potential failure modes and their consequences. Hazard and operability studies, fault tree analysis, and consequence modeling help identify critical control points and establish appropriate safety margins. Regular safety audits and compliance monitoring ensure continuous improvement of safety management systems and adherence to evolving regulatory requirements.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!