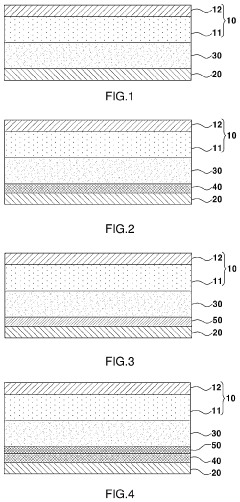
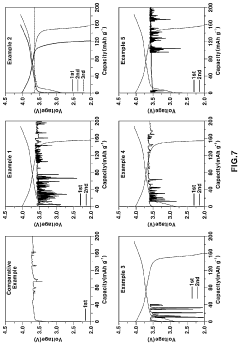
Solid Electrolyte Choices Benchmark For Anode-Free Solid-State
SEP 1, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Solid Electrolyte Technology Background and Objectives
Solid electrolyte technology has evolved significantly over the past few decades, transitioning from a theoretical concept to a promising solution for next-generation energy storage systems. The development trajectory began in the 1970s with the discovery of fast ion conductors, followed by incremental improvements in ionic conductivity and mechanical properties throughout the 1990s and 2000s. Recent breakthroughs in material science and manufacturing techniques have accelerated the advancement of solid electrolytes, positioning them as viable alternatives to conventional liquid electrolytes in battery applications.
The technological evolution has been driven by the inherent limitations of traditional lithium-ion batteries, particularly safety concerns related to flammable liquid electrolytes and the performance ceiling imposed by existing electrode-electrolyte combinations. Solid electrolytes offer potential solutions to these challenges by enabling higher energy density, enhanced safety, and extended cycle life, which align with the growing demands for improved energy storage systems across various sectors.
Current research focuses on three primary categories of solid electrolytes: oxide-based, sulfide-based, and polymer-based materials. Each category presents distinct advantages and challenges regarding ionic conductivity, electrochemical stability, and mechanical properties. The oxide-based electrolytes, such as LLZO (Li7La3Zr2O12), demonstrate excellent chemical stability but face challenges with grain boundary resistance. Sulfide-based electrolytes, including LGPS (Li10GeP2S12), offer superior ionic conductivity but are sensitive to moisture and air. Polymer-based electrolytes provide flexibility and processability advantages but struggle with lower conductivity at room temperature.
The anode-free solid-state battery configuration represents a particularly promising direction, as it eliminates the need for pre-lithiated anodes, potentially increasing energy density by 30-50% compared to conventional designs. This approach relies heavily on the selection of appropriate solid electrolytes that can withstand lithium plating and stripping while maintaining stable interfaces.
The primary objectives of solid electrolyte technology development for anode-free solid-state batteries include achieving room temperature ionic conductivity exceeding 10^-3 S/cm, establishing electrochemical stability windows wider than 4.5V, developing manufacturing processes compatible with existing production infrastructure, and ensuring long-term cycling stability beyond 1000 cycles with minimal capacity degradation.
Additionally, research aims to address the critical challenges of interfacial resistance between electrolytes and electrodes, mechanical stability during volume changes, and scalable production methods that maintain material performance at commercially viable costs. These objectives collectively support the overarching goal of creating solid-state batteries that outperform current lithium-ion technology while enhancing safety and sustainability.
The technological evolution has been driven by the inherent limitations of traditional lithium-ion batteries, particularly safety concerns related to flammable liquid electrolytes and the performance ceiling imposed by existing electrode-electrolyte combinations. Solid electrolytes offer potential solutions to these challenges by enabling higher energy density, enhanced safety, and extended cycle life, which align with the growing demands for improved energy storage systems across various sectors.
Current research focuses on three primary categories of solid electrolytes: oxide-based, sulfide-based, and polymer-based materials. Each category presents distinct advantages and challenges regarding ionic conductivity, electrochemical stability, and mechanical properties. The oxide-based electrolytes, such as LLZO (Li7La3Zr2O12), demonstrate excellent chemical stability but face challenges with grain boundary resistance. Sulfide-based electrolytes, including LGPS (Li10GeP2S12), offer superior ionic conductivity but are sensitive to moisture and air. Polymer-based electrolytes provide flexibility and processability advantages but struggle with lower conductivity at room temperature.
The anode-free solid-state battery configuration represents a particularly promising direction, as it eliminates the need for pre-lithiated anodes, potentially increasing energy density by 30-50% compared to conventional designs. This approach relies heavily on the selection of appropriate solid electrolytes that can withstand lithium plating and stripping while maintaining stable interfaces.
The primary objectives of solid electrolyte technology development for anode-free solid-state batteries include achieving room temperature ionic conductivity exceeding 10^-3 S/cm, establishing electrochemical stability windows wider than 4.5V, developing manufacturing processes compatible with existing production infrastructure, and ensuring long-term cycling stability beyond 1000 cycles with minimal capacity degradation.
Additionally, research aims to address the critical challenges of interfacial resistance between electrolytes and electrodes, mechanical stability during volume changes, and scalable production methods that maintain material performance at commercially viable costs. These objectives collectively support the overarching goal of creating solid-state batteries that outperform current lithium-ion technology while enhancing safety and sustainability.
Market Analysis for Anode-Free Solid-State Batteries
The anode-free solid-state battery market is experiencing significant growth potential, driven by the increasing demand for high-energy-density energy storage solutions across multiple sectors. Current market projections indicate that the global solid-state battery market could reach $8 billion by 2030, with anode-free configurations potentially capturing 15-20% of this emerging segment.
The automotive industry represents the primary demand driver, as manufacturers seek battery technologies that can deliver greater range, faster charging capabilities, and enhanced safety profiles. Major automotive OEMs including Toyota, Volkswagen, and BMW have made substantial investments in solid-state battery technology, with particular interest in anode-free designs that promise energy densities exceeding 400 Wh/kg.
Consumer electronics constitutes the second-largest market segment, where the compact form factor and improved safety characteristics of anode-free solid-state batteries address critical pain points in portable device design. Market research indicates that premium smartphone and wearable device manufacturers are actively exploring integration pathways for this technology.
Stationary energy storage applications represent an emerging opportunity, particularly for grid-scale implementations where energy density, cycle life, and safety are paramount considerations. Though currently a smaller segment, industry analysts project 25-30% annual growth in this application area through 2028.
Regional market analysis reveals Asia-Pacific as the dominant manufacturing hub, with Japan and South Korea leading in intellectual property development for solid electrolytes suitable for anode-free configurations. North America demonstrates strength in materials innovation and system integration, while European markets show accelerating adoption rates driven by stringent safety regulations and sustainability initiatives.
Customer demand analysis indicates three primary value propositions driving market interest: safety improvements compared to conventional lithium-ion batteries, potential for higher energy density, and extended operational lifetimes. Market surveys show that 70% of potential industrial customers rank safety as their primary consideration when evaluating next-generation battery technologies.
Supply chain constraints represent a significant market challenge, particularly regarding the availability of high-purity solid electrolyte materials at commercial scale. Current production capacity for ceramic and sulfide-based electrolytes suitable for anode-free designs remains limited, creating potential bottlenecks as demand accelerates.
Pricing trends suggest that anode-free solid-state batteries currently command a significant premium over conventional lithium-ion technologies, with cost-per-kWh estimates 3-4 times higher than established technologies. However, learning curve projections indicate potential for 60-70% cost reduction by 2030 as manufacturing processes mature and economies of scale are realized.
The automotive industry represents the primary demand driver, as manufacturers seek battery technologies that can deliver greater range, faster charging capabilities, and enhanced safety profiles. Major automotive OEMs including Toyota, Volkswagen, and BMW have made substantial investments in solid-state battery technology, with particular interest in anode-free designs that promise energy densities exceeding 400 Wh/kg.
Consumer electronics constitutes the second-largest market segment, where the compact form factor and improved safety characteristics of anode-free solid-state batteries address critical pain points in portable device design. Market research indicates that premium smartphone and wearable device manufacturers are actively exploring integration pathways for this technology.
Stationary energy storage applications represent an emerging opportunity, particularly for grid-scale implementations where energy density, cycle life, and safety are paramount considerations. Though currently a smaller segment, industry analysts project 25-30% annual growth in this application area through 2028.
Regional market analysis reveals Asia-Pacific as the dominant manufacturing hub, with Japan and South Korea leading in intellectual property development for solid electrolytes suitable for anode-free configurations. North America demonstrates strength in materials innovation and system integration, while European markets show accelerating adoption rates driven by stringent safety regulations and sustainability initiatives.
Customer demand analysis indicates three primary value propositions driving market interest: safety improvements compared to conventional lithium-ion batteries, potential for higher energy density, and extended operational lifetimes. Market surveys show that 70% of potential industrial customers rank safety as their primary consideration when evaluating next-generation battery technologies.
Supply chain constraints represent a significant market challenge, particularly regarding the availability of high-purity solid electrolyte materials at commercial scale. Current production capacity for ceramic and sulfide-based electrolytes suitable for anode-free designs remains limited, creating potential bottlenecks as demand accelerates.
Pricing trends suggest that anode-free solid-state batteries currently command a significant premium over conventional lithium-ion technologies, with cost-per-kWh estimates 3-4 times higher than established technologies. However, learning curve projections indicate potential for 60-70% cost reduction by 2030 as manufacturing processes mature and economies of scale are realized.
Current Status and Technical Challenges of Solid Electrolytes
Solid electrolytes represent a critical component in the development of next-generation solid-state batteries, particularly for anode-free configurations. Currently, three major categories dominate the solid electrolyte landscape: oxide-based, sulfide-based, and polymer-based materials, each with distinct advantages and limitations.
Oxide-based solid electrolytes, such as LLZO (Li7La3Zr2O12) and LATP (Li1.3Al0.3Ti1.7(PO4)3), offer excellent thermal and chemical stability with high oxidation resistance. However, they suffer from relatively low ionic conductivity at room temperature (typically 10^-4 to 10^-3 S/cm) and high grain boundary resistance, requiring high-temperature sintering processes that complicate manufacturing.
Sulfide-based electrolytes, including Li10GeP2S12 (LGPS) and Li6PS5Cl, demonstrate superior ionic conductivity (10^-2 to 10^-3 S/cm at room temperature), approaching that of liquid electrolytes. Their mechanical properties allow for better interfacial contact with electrodes. Nevertheless, these materials are highly sensitive to moisture and air, necessitating stringent manufacturing environments. Additionally, they exhibit limited electrochemical stability windows, particularly at high voltages.
Polymer-based electrolytes, such as PEO (polyethylene oxide) complexes, offer excellent flexibility and processability, enabling simpler battery designs. Their major drawback is significantly lower ionic conductivity at room temperature, often requiring operation at elevated temperatures (>60°C) to achieve practical performance levels.
The primary technical challenges facing solid electrolytes for anode-free configurations include interfacial stability issues, particularly at the electrolyte-cathode interface where high-voltage operation can trigger decomposition reactions. Mechanical stability during cycling represents another significant hurdle, as volume changes in the cathode and the formation of lithium at the anode side create mechanical stresses that can lead to contact loss or electrolyte fracture.
Manufacturing scalability remains problematic, with many high-performance solid electrolytes requiring complex synthesis routes or extreme processing conditions. The trade-off between ionic conductivity and electrochemical stability window continues to challenge researchers, as materials with excellent conductivity often suffer from narrow stability windows.
For anode-free configurations specifically, the solid electrolyte must withstand lithium plating/stripping processes while preventing dendrite penetration—a particularly demanding requirement that few current materials can fully satisfy. The development of composite electrolytes that combine the advantages of different material classes shows promise but introduces additional interface management challenges.
Recent research has focused on interface engineering strategies, including the use of interlayers and gradient compositions to mitigate these challenges, though no universal solution has yet emerged for commercial-scale implementation.
Oxide-based solid electrolytes, such as LLZO (Li7La3Zr2O12) and LATP (Li1.3Al0.3Ti1.7(PO4)3), offer excellent thermal and chemical stability with high oxidation resistance. However, they suffer from relatively low ionic conductivity at room temperature (typically 10^-4 to 10^-3 S/cm) and high grain boundary resistance, requiring high-temperature sintering processes that complicate manufacturing.
Sulfide-based electrolytes, including Li10GeP2S12 (LGPS) and Li6PS5Cl, demonstrate superior ionic conductivity (10^-2 to 10^-3 S/cm at room temperature), approaching that of liquid electrolytes. Their mechanical properties allow for better interfacial contact with electrodes. Nevertheless, these materials are highly sensitive to moisture and air, necessitating stringent manufacturing environments. Additionally, they exhibit limited electrochemical stability windows, particularly at high voltages.
Polymer-based electrolytes, such as PEO (polyethylene oxide) complexes, offer excellent flexibility and processability, enabling simpler battery designs. Their major drawback is significantly lower ionic conductivity at room temperature, often requiring operation at elevated temperatures (>60°C) to achieve practical performance levels.
The primary technical challenges facing solid electrolytes for anode-free configurations include interfacial stability issues, particularly at the electrolyte-cathode interface where high-voltage operation can trigger decomposition reactions. Mechanical stability during cycling represents another significant hurdle, as volume changes in the cathode and the formation of lithium at the anode side create mechanical stresses that can lead to contact loss or electrolyte fracture.
Manufacturing scalability remains problematic, with many high-performance solid electrolytes requiring complex synthesis routes or extreme processing conditions. The trade-off between ionic conductivity and electrochemical stability window continues to challenge researchers, as materials with excellent conductivity often suffer from narrow stability windows.
For anode-free configurations specifically, the solid electrolyte must withstand lithium plating/stripping processes while preventing dendrite penetration—a particularly demanding requirement that few current materials can fully satisfy. The development of composite electrolytes that combine the advantages of different material classes shows promise but introduces additional interface management challenges.
Recent research has focused on interface engineering strategies, including the use of interlayers and gradient compositions to mitigate these challenges, though no universal solution has yet emerged for commercial-scale implementation.
Benchmark Methodology for Solid Electrolyte Materials
01 Ionic conductivity benchmarks for solid electrolytes
Ionic conductivity is a critical performance metric for solid electrolytes in battery applications. Various testing methodologies are employed to measure and benchmark the ionic conductivity of different solid electrolyte materials. These measurements typically involve impedance spectroscopy techniques under controlled temperature conditions. Higher ionic conductivity values generally indicate better performance for solid electrolyte materials, with some advanced materials approaching the conductivity levels of liquid electrolytes.- Performance evaluation methods for solid electrolytes: Various methods and systems are used to evaluate the performance of solid electrolytes in batteries. These include benchmarking techniques that measure key parameters such as ionic conductivity, electrochemical stability, and mechanical properties. Advanced testing protocols help in standardizing the evaluation process, allowing for consistent comparison between different solid electrolyte materials and formulations.
- Solid-state battery electrolyte composition benchmarks: The composition of solid electrolytes significantly impacts their performance in batteries. Benchmarks have been established for various compositions including ceramic, polymer, and composite electrolytes. These benchmarks focus on the chemical stability, thermal properties, and compatibility with electrode materials. Different formulations are evaluated against these standards to determine their suitability for specific battery applications.
- Computational modeling and simulation for solid electrolyte performance: Computational methods are employed to predict and analyze the performance of solid electrolytes before physical testing. These include molecular dynamics simulations, density functional theory calculations, and machine learning approaches that help identify promising materials and optimize their properties. Such computational benchmarks accelerate the development process by narrowing down candidate materials for experimental validation.
- Interface engineering and stability benchmarks: The interfaces between solid electrolytes and electrodes are critical for battery performance. Benchmarks have been developed to evaluate interface stability, resistance, and degradation mechanisms. These standards help in designing solid electrolytes with improved interfacial properties, reducing impedance and enhancing long-term cycling stability of solid-state batteries.
- Manufacturing process benchmarks for solid electrolytes: The manufacturing processes for solid electrolytes significantly impact their performance characteristics. Benchmarks have been established for various fabrication methods including sintering, solution casting, and vapor deposition techniques. These standards evaluate the scalability, reproducibility, and cost-effectiveness of production methods while ensuring the resulting electrolytes meet performance requirements for commercial applications.
02 Mechanical stability and interface performance metrics
The mechanical properties and interface characteristics of solid electrolytes are crucial performance benchmarks. These include measurements of Young's modulus, fracture toughness, and interfacial resistance with electrode materials. Solid electrolytes must maintain structural integrity during battery cycling while forming stable interfaces with electrodes. Testing protocols evaluate the ability of solid electrolytes to withstand volume changes during cycling and prevent dendrite formation, which are essential for long-term battery reliability and safety.Expand Specific Solutions03 Electrochemical stability window assessment
The electrochemical stability window is a key performance benchmark for solid electrolytes, defining the voltage range within which the electrolyte remains stable without decomposition. Testing methodologies include cyclic voltammetry and galvanostatic cycling to determine the upper and lower voltage limits. Wider electrochemical windows enable higher energy density batteries by allowing the use of high-voltage cathode materials. Various solid electrolyte compositions are evaluated based on their ability to maintain stability across broader voltage ranges.Expand Specific Solutions04 Temperature performance and thermal stability benchmarks
Temperature-dependent performance is a critical benchmark for solid electrolytes, evaluating their conductivity and stability across a wide temperature range. Testing protocols measure ionic conductivity at various temperatures to determine activation energy and identify phase transitions. Thermal stability tests assess the material's resistance to decomposition at elevated temperatures. Superior solid electrolytes maintain consistent performance across broad temperature ranges, enabling battery operation in extreme environments and improving safety by eliminating thermal runaway risks associated with liquid electrolytes.Expand Specific Solutions05 Standardized testing protocols and performance comparison frameworks
Standardized testing methodologies and benchmarking frameworks are essential for comparing different solid electrolyte materials. These protocols establish consistent measurement conditions for properties like ionic conductivity, interfacial resistance, and cycle life. Automated testing systems enable high-throughput screening of multiple electrolyte compositions. Benchmark databases compile performance metrics across various material classes, allowing researchers to identify promising candidates and performance trends. These standardized approaches facilitate meaningful comparisons between different research efforts and accelerate the development of improved solid electrolyte materials.Expand Specific Solutions
Leading Companies and Research Institutions in Solid Electrolytes
The solid electrolyte market for anode-free solid-state batteries is in an early growth phase, characterized by intensive R&D and strategic positioning. The market is projected to expand significantly as electric vehicle adoption accelerates, with estimates suggesting multi-billion dollar potential by 2030. Technologically, the field remains in development with varying maturity levels across different electrolyte chemistries. Key players demonstrate diverse approaches: established automotive manufacturers (Toyota, BMW, Hyundai) are investing heavily in proprietary technologies; specialized battery developers (QuantumScape, Blue Current) are advancing innovative electrolyte solutions; while major electronics companies (Samsung SDI, LG Energy Solution) leverage their manufacturing expertise. Academic-industrial partnerships, particularly involving institutions like University of Maryland and Chinese Academy of Sciences, are accelerating commercialization timelines through fundamental research breakthroughs.
GM Global Technology Operations LLC
Technical Solution: GM has developed a hybrid solid electrolyte system for anode-free solid-state batteries that combines a polymer matrix with ceramic fillers to achieve balanced performance characteristics. Their approach utilizes a proprietary polymer blend that provides mechanical flexibility while ceramic additives enhance ionic conductivity. GM's solid electrolyte design incorporates gradient structures where composition varies across the electrolyte thickness to optimize performance at both cathode and anode interfaces. The manufacturing process employs solution-based techniques compatible with existing production infrastructure, potentially enabling more rapid scaling. GM has demonstrated prototype cells achieving energy densities exceeding 350 Wh/kg while maintaining stable cycling at automotive-relevant temperatures (-20°C to 60°C). Their technology roadmap focuses on progressive improvements in electrolyte formulation to enhance rate capability while maintaining the stability advantages of the solid-state system.
Strengths: Integrated vehicle-level optimization approach; manufacturing process compatible with existing production lines; good low-temperature performance; balanced mechanical and electrochemical properties. Weaknesses: Lower ionic conductivity compared to pure ceramic systems; potential for polymer degradation at elevated temperatures; higher interfacial resistance than liquid electrolyte systems.
QuantumScape Corp.
Technical Solution: QuantumScape has developed a proprietary ceramic solid electrolyte separator for anode-free solid-state batteries. Their technology employs a lithium-metal anode that forms in situ during the first charge cycle, eliminating the need for a pre-deposited lithium metal layer. The solid electrolyte is a lithium-conducting ceramic material that enables fast charging capabilities (reportedly 80% charge in less than 15 minutes) while maintaining high energy density. QuantumScape's solid electrolyte design addresses dendrite formation issues through a proprietary interface engineering approach that prevents lithium penetration even at high current densities. Their multi-layer cell architecture has demonstrated over 800 cycles with greater than 80% capacity retention under automotive test conditions. The company has secured partnerships with Volkswagen and other automotive manufacturers to commercialize this technology.
Strengths: Superior energy density (reportedly >400 Wh/kg at cell level); excellent fast-charging capability; demonstrated cycle life exceeding industry benchmarks; strong automotive partnerships. Weaknesses: Manufacturing scalability remains challenging; high production costs; technology still in pre-commercial phase; limited public disclosure of specific material composition.
Critical Patents and Scientific Literature on Solid Electrolytes
Anode-free all-solid-state battery including solid electrolyte having high ion conductivity and surface-roughened anode current collector
PatentPendingUS20220393180A1
Innovation
- An anode-free all-solid-state battery design featuring an anode current collector layer with surface roughness of 100 nm to 1,000 nm, directly contacting a solid electrolyte layer with ionic conductivity of 1 mS/cm to 20 mS/cm, and optionally a coating layer containing carbon and lithium-forming metals, which enhances lithium deposition uniformity and stability without additional layers.
Anode-free solid-state battery and use thereof
PatentWO2025103689A1
Innovation
- Incorporating an additional solid-state electrolyte layer between the solid-state electrolyte separator and the conductor improves deformability and maintains contact between the components during charging cycles.
Manufacturing Scalability Assessment
The manufacturing scalability of solid electrolytes represents a critical factor in the commercial viability of anode-free solid-state batteries. Current production methods for oxide-based solid electrolytes typically involve energy-intensive high-temperature sintering processes, which present significant challenges for large-scale manufacturing. These processes often require temperatures exceeding 1000°C, resulting in substantial energy consumption and specialized equipment requirements that increase production costs.
Sulfide-based electrolytes offer more promising manufacturing pathways, as they can be processed at lower temperatures (typically 200-400°C) and exhibit better mechanical properties for cold pressing techniques. However, their extreme sensitivity to moisture necessitates stringent environmental controls during production, adding complexity to manufacturing infrastructure. Companies like Solid Power have made notable progress in developing scalable manufacturing processes for sulfide electrolytes, though significant challenges remain in maintaining consistent quality at industrial scales.
Polymer-based solid electrolytes present perhaps the most scalable option from a manufacturing perspective. Their compatibility with established roll-to-roll processing techniques allows for adaptation of existing lithium-ion battery manufacturing infrastructure. This compatibility significantly reduces capital expenditure requirements for battery manufacturers looking to transition to solid-state technology. However, the trade-off comes in the form of lower ionic conductivity compared to inorganic alternatives, particularly at room temperature.
Composite electrolytes that combine inorganic fillers with polymer matrices represent a promising middle ground, potentially offering improved manufacturability while maintaining acceptable performance. Recent advancements in co-extrusion and solution processing methods have demonstrated potential for scaling these hybrid systems, though optimization of filler distribution remains challenging.
From an equipment perspective, the transition to solid electrolytes requires significant modifications to existing battery production lines. The absence of liquid components eliminates certain manufacturing steps but introduces new challenges in layer uniformity and interfacial contact. Specialized equipment for dry room processing, precision layer deposition, and high-pressure assembly represents substantial capital investments that must be considered in scalability assessments.
Material supply chains also impact manufacturing scalability, with certain solid electrolyte compositions requiring rare or strategically important elements. For instance, NASICON-type electrolytes containing zirconium and LLZO electrolytes containing lanthanum may face supply constraints as production scales. Developing alternative formulations with more abundant materials represents an important direction for improving long-term manufacturing scalability.
Sulfide-based electrolytes offer more promising manufacturing pathways, as they can be processed at lower temperatures (typically 200-400°C) and exhibit better mechanical properties for cold pressing techniques. However, their extreme sensitivity to moisture necessitates stringent environmental controls during production, adding complexity to manufacturing infrastructure. Companies like Solid Power have made notable progress in developing scalable manufacturing processes for sulfide electrolytes, though significant challenges remain in maintaining consistent quality at industrial scales.
Polymer-based solid electrolytes present perhaps the most scalable option from a manufacturing perspective. Their compatibility with established roll-to-roll processing techniques allows for adaptation of existing lithium-ion battery manufacturing infrastructure. This compatibility significantly reduces capital expenditure requirements for battery manufacturers looking to transition to solid-state technology. However, the trade-off comes in the form of lower ionic conductivity compared to inorganic alternatives, particularly at room temperature.
Composite electrolytes that combine inorganic fillers with polymer matrices represent a promising middle ground, potentially offering improved manufacturability while maintaining acceptable performance. Recent advancements in co-extrusion and solution processing methods have demonstrated potential for scaling these hybrid systems, though optimization of filler distribution remains challenging.
From an equipment perspective, the transition to solid electrolytes requires significant modifications to existing battery production lines. The absence of liquid components eliminates certain manufacturing steps but introduces new challenges in layer uniformity and interfacial contact. Specialized equipment for dry room processing, precision layer deposition, and high-pressure assembly represents substantial capital investments that must be considered in scalability assessments.
Material supply chains also impact manufacturing scalability, with certain solid electrolyte compositions requiring rare or strategically important elements. For instance, NASICON-type electrolytes containing zirconium and LLZO electrolytes containing lanthanum may face supply constraints as production scales. Developing alternative formulations with more abundant materials represents an important direction for improving long-term manufacturing scalability.
Safety and Performance Comparison Framework
The safety and performance evaluation of solid electrolytes for anode-free solid-state batteries requires a comprehensive framework that addresses multiple critical parameters simultaneously. This framework must balance the inherent trade-offs between safety features and electrochemical performance metrics to provide meaningful benchmarking data for research and development teams.
A robust comparison framework begins with standardized safety testing protocols that evaluate thermal stability, chemical compatibility with electrode materials, and mechanical integrity under various operating conditions. These protocols should include thermal runaway tests, dendrite penetration resistance measurements, and accelerated aging studies to predict long-term stability profiles of different solid electrolyte candidates.
Performance metrics must be systematically evaluated across operating temperature ranges relevant to real-world applications (-20°C to 60°C). Key parameters include ionic conductivity, electronic insulation properties, electrochemical stability windows, and interfacial resistance evolution during cycling. The framework should incorporate both static measurements and dynamic testing under various current densities to reflect actual usage scenarios.
Material-specific benchmarking categories should be established for the major solid electrolyte families: oxide-based ceramics (LLZO, LATP), sulfide-based glasses and glass-ceramics (LGPS, Li3PS4), polymer-based systems (PEO-based), and composite/hybrid electrolytes. Each category requires tailored testing protocols that address their unique failure modes and performance limitations.
Cycle life projection models must be integrated into the framework, correlating initial performance metrics with long-term degradation patterns. This approach enables researchers to identify early indicators of potential failure mechanisms and estimate practical service life without conducting exhaustive long-duration testing for every candidate material.
Manufacturing scalability assessment should be included as a practical dimension of the framework, evaluating factors such as raw material availability, processing complexity, and compatibility with existing battery production infrastructure. This ensures that promising materials identified through laboratory testing can realistically transition to commercial production environments.
The framework should ultimately generate a multi-dimensional performance index that weights various parameters according to application-specific requirements, allowing for customized evaluation based on whether the priority is safety, energy density, power capability, or operational lifespan. This adaptable approach recognizes that no single solid electrolyte solution will be optimal across all possible use cases in the evolving landscape of solid-state battery technologies.
A robust comparison framework begins with standardized safety testing protocols that evaluate thermal stability, chemical compatibility with electrode materials, and mechanical integrity under various operating conditions. These protocols should include thermal runaway tests, dendrite penetration resistance measurements, and accelerated aging studies to predict long-term stability profiles of different solid electrolyte candidates.
Performance metrics must be systematically evaluated across operating temperature ranges relevant to real-world applications (-20°C to 60°C). Key parameters include ionic conductivity, electronic insulation properties, electrochemical stability windows, and interfacial resistance evolution during cycling. The framework should incorporate both static measurements and dynamic testing under various current densities to reflect actual usage scenarios.
Material-specific benchmarking categories should be established for the major solid electrolyte families: oxide-based ceramics (LLZO, LATP), sulfide-based glasses and glass-ceramics (LGPS, Li3PS4), polymer-based systems (PEO-based), and composite/hybrid electrolytes. Each category requires tailored testing protocols that address their unique failure modes and performance limitations.
Cycle life projection models must be integrated into the framework, correlating initial performance metrics with long-term degradation patterns. This approach enables researchers to identify early indicators of potential failure mechanisms and estimate practical service life without conducting exhaustive long-duration testing for every candidate material.
Manufacturing scalability assessment should be included as a practical dimension of the framework, evaluating factors such as raw material availability, processing complexity, and compatibility with existing battery production infrastructure. This ensures that promising materials identified through laboratory testing can realistically transition to commercial production environments.
The framework should ultimately generate a multi-dimensional performance index that weights various parameters according to application-specific requirements, allowing for customized evaluation based on whether the priority is safety, energy density, power capability, or operational lifespan. This adaptable approach recognizes that no single solid electrolyte solution will be optimal across all possible use cases in the evolving landscape of solid-state battery technologies.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!