Utilizing Sodium Nitrate in Catalysis for Reaction Acceleration
MAR 3, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sodium Nitrate Catalysis Background and Objectives
Sodium nitrate (NaNO3) has emerged as a significant catalyst in modern chemical synthesis, representing a paradigm shift from traditional expensive metal-based catalysts toward more sustainable and cost-effective alternatives. This inorganic salt, historically recognized primarily for its applications in fertilizers and food preservation, has gained substantial attention in the catalysis community due to its unique oxidizing properties and ability to facilitate various organic transformations under mild conditions.
The evolution of sodium nitrate catalysis can be traced back to early observations of nitrate-mediated oxidation reactions in the mid-20th century. However, systematic investigation of its catalytic potential began gaining momentum in the 1990s when researchers discovered its effectiveness in promoting C-H activation and cross-coupling reactions. The breakthrough came with the recognition that sodium nitrate could serve as both an oxidant and a catalyst, enabling previously challenging transformations while maintaining excellent selectivity profiles.
Recent developments have positioned sodium nitrate as a versatile catalyst capable of accelerating diverse reaction types, including oxidative coupling reactions, dehydrogenation processes, and heterocycle synthesis. Its mechanism typically involves the generation of nitrate radicals or nitrite intermediates, which facilitate electron transfer processes and promote bond formation or cleavage. This mechanistic versatility has opened new avenues for synthetic chemists seeking environmentally benign alternatives to traditional catalytic systems.
The primary objective of advancing sodium nitrate catalysis centers on developing more efficient and selective catalytic protocols that can operate under milder reaction conditions while achieving superior yields. Key goals include expanding the substrate scope to encompass challenging molecular frameworks, improving reaction kinetics through optimized catalyst formulations, and establishing predictable selectivity patterns for complex multi-functional substrates.
Furthermore, the integration of sodium nitrate catalysis with green chemistry principles represents a crucial objective, aiming to reduce waste generation, eliminate toxic solvents, and minimize energy consumption. The development of recyclable catalytic systems and the exploration of continuous flow processes using sodium nitrate catalysts constitute important research directions that align with sustainable manufacturing practices and industrial scalability requirements.
The evolution of sodium nitrate catalysis can be traced back to early observations of nitrate-mediated oxidation reactions in the mid-20th century. However, systematic investigation of its catalytic potential began gaining momentum in the 1990s when researchers discovered its effectiveness in promoting C-H activation and cross-coupling reactions. The breakthrough came with the recognition that sodium nitrate could serve as both an oxidant and a catalyst, enabling previously challenging transformations while maintaining excellent selectivity profiles.
Recent developments have positioned sodium nitrate as a versatile catalyst capable of accelerating diverse reaction types, including oxidative coupling reactions, dehydrogenation processes, and heterocycle synthesis. Its mechanism typically involves the generation of nitrate radicals or nitrite intermediates, which facilitate electron transfer processes and promote bond formation or cleavage. This mechanistic versatility has opened new avenues for synthetic chemists seeking environmentally benign alternatives to traditional catalytic systems.
The primary objective of advancing sodium nitrate catalysis centers on developing more efficient and selective catalytic protocols that can operate under milder reaction conditions while achieving superior yields. Key goals include expanding the substrate scope to encompass challenging molecular frameworks, improving reaction kinetics through optimized catalyst formulations, and establishing predictable selectivity patterns for complex multi-functional substrates.
Furthermore, the integration of sodium nitrate catalysis with green chemistry principles represents a crucial objective, aiming to reduce waste generation, eliminate toxic solvents, and minimize energy consumption. The development of recyclable catalytic systems and the exploration of continuous flow processes using sodium nitrate catalysts constitute important research directions that align with sustainable manufacturing practices and industrial scalability requirements.
Market Demand for Sodium Nitrate Catalytic Applications
The global chemical industry is experiencing unprecedented demand for efficient catalytic solutions, with sodium nitrate emerging as a promising catalyst for reaction acceleration across multiple sectors. The pharmaceutical industry represents one of the most significant market drivers, where sodium nitrate catalysis enables faster synthesis of active pharmaceutical ingredients and intermediates. This acceleration capability directly translates to reduced production costs and shorter time-to-market for critical medications, making it particularly valuable for generic drug manufacturers seeking competitive advantages.
Industrial chemical production constitutes another major demand segment, particularly in the synthesis of specialty chemicals and fine chemicals. Manufacturing facilities are increasingly adopting sodium nitrate-based catalytic systems to enhance reaction rates in oxidation processes, nitration reactions, and organic transformations. The ability to operate under milder conditions while achieving superior conversion rates has attracted significant interest from chemical processors focused on energy efficiency and sustainability.
The agricultural sector presents substantial growth potential for sodium nitrate catalytic applications, especially in fertilizer production and agrochemical synthesis. The dual functionality of sodium nitrate as both a nutrient source and catalytic agent creates unique value propositions for agricultural chemical manufacturers. This convergence of nutritional and catalytic properties enables streamlined production processes and cost-effective manufacturing approaches.
Environmental remediation applications are driving emerging demand patterns, where sodium nitrate catalysis facilitates the breakdown of organic pollutants and hazardous compounds. Water treatment facilities and environmental engineering companies are exploring these catalytic systems for enhanced degradation of persistent organic pollutants and industrial waste streams.
The renewable energy sector is generating new demand streams through applications in fuel synthesis and energy storage systems. Sodium nitrate catalysis shows promise in biomass conversion processes and synthetic fuel production, aligning with global sustainability initiatives and carbon reduction targets.
Regional demand patterns reveal strong growth in Asia-Pacific markets, driven by expanding chemical manufacturing capacity and increasing environmental regulations. North American and European markets demonstrate steady demand growth, primarily focused on pharmaceutical and specialty chemical applications where regulatory compliance and process efficiency are paramount considerations.
Industrial chemical production constitutes another major demand segment, particularly in the synthesis of specialty chemicals and fine chemicals. Manufacturing facilities are increasingly adopting sodium nitrate-based catalytic systems to enhance reaction rates in oxidation processes, nitration reactions, and organic transformations. The ability to operate under milder conditions while achieving superior conversion rates has attracted significant interest from chemical processors focused on energy efficiency and sustainability.
The agricultural sector presents substantial growth potential for sodium nitrate catalytic applications, especially in fertilizer production and agrochemical synthesis. The dual functionality of sodium nitrate as both a nutrient source and catalytic agent creates unique value propositions for agricultural chemical manufacturers. This convergence of nutritional and catalytic properties enables streamlined production processes and cost-effective manufacturing approaches.
Environmental remediation applications are driving emerging demand patterns, where sodium nitrate catalysis facilitates the breakdown of organic pollutants and hazardous compounds. Water treatment facilities and environmental engineering companies are exploring these catalytic systems for enhanced degradation of persistent organic pollutants and industrial waste streams.
The renewable energy sector is generating new demand streams through applications in fuel synthesis and energy storage systems. Sodium nitrate catalysis shows promise in biomass conversion processes and synthetic fuel production, aligning with global sustainability initiatives and carbon reduction targets.
Regional demand patterns reveal strong growth in Asia-Pacific markets, driven by expanding chemical manufacturing capacity and increasing environmental regulations. North American and European markets demonstrate steady demand growth, primarily focused on pharmaceutical and specialty chemical applications where regulatory compliance and process efficiency are paramount considerations.
Current Status and Challenges in Nitrate Catalysis
Sodium nitrate catalysis has emerged as a promising field within heterogeneous catalysis, demonstrating significant potential for accelerating various chemical reactions. Current research primarily focuses on utilizing sodium nitrate as both a catalyst and co-catalyst in oxidation reactions, dehydrogenation processes, and selective organic transformations. The technology has shown particular effectiveness in promoting C-H bond activation, alcohol oxidation, and alkane functionalization under relatively mild conditions.
The global distribution of nitrate catalysis research reveals concentrated efforts in developed nations, with leading research institutions in the United States, Germany, Japan, and China driving fundamental discoveries. European research centers have particularly excelled in mechanistic studies, while Asian institutions have focused more on industrial applications and process optimization. This geographical disparity has created knowledge gaps in technology transfer and standardization across different regions.
Several technical challenges currently limit the widespread adoption of sodium nitrate catalysis. The primary constraint involves catalyst stability under high-temperature conditions, where sodium nitrate tends to decompose or undergo phase transitions that reduce catalytic activity. Additionally, selectivity control remains problematic in multi-step reactions, often leading to unwanted side products that complicate purification processes.
Another significant challenge lies in understanding the precise reaction mechanisms. While empirical evidence supports sodium nitrate's catalytic effectiveness, the molecular-level interactions between the nitrate species and substrate molecules are not fully elucidated. This knowledge gap hinders rational catalyst design and optimization efforts.
Scalability issues present additional obstacles for industrial implementation. Laboratory-scale successes often fail to translate to larger production volumes due to heat and mass transfer limitations, catalyst recovery difficulties, and economic considerations related to sodium nitrate consumption and regeneration.
Environmental concerns also pose challenges, particularly regarding nitrate waste management and potential NOx emissions during high-temperature operations. Regulatory compliance requirements vary significantly across different jurisdictions, creating additional complexity for commercial deployment.
The heterogeneous nature of current research approaches has resulted in fragmented knowledge bases, with limited standardization in experimental protocols and performance metrics. This situation complicates comparative studies and slows overall progress in the field.
The global distribution of nitrate catalysis research reveals concentrated efforts in developed nations, with leading research institutions in the United States, Germany, Japan, and China driving fundamental discoveries. European research centers have particularly excelled in mechanistic studies, while Asian institutions have focused more on industrial applications and process optimization. This geographical disparity has created knowledge gaps in technology transfer and standardization across different regions.
Several technical challenges currently limit the widespread adoption of sodium nitrate catalysis. The primary constraint involves catalyst stability under high-temperature conditions, where sodium nitrate tends to decompose or undergo phase transitions that reduce catalytic activity. Additionally, selectivity control remains problematic in multi-step reactions, often leading to unwanted side products that complicate purification processes.
Another significant challenge lies in understanding the precise reaction mechanisms. While empirical evidence supports sodium nitrate's catalytic effectiveness, the molecular-level interactions between the nitrate species and substrate molecules are not fully elucidated. This knowledge gap hinders rational catalyst design and optimization efforts.
Scalability issues present additional obstacles for industrial implementation. Laboratory-scale successes often fail to translate to larger production volumes due to heat and mass transfer limitations, catalyst recovery difficulties, and economic considerations related to sodium nitrate consumption and regeneration.
Environmental concerns also pose challenges, particularly regarding nitrate waste management and potential NOx emissions during high-temperature operations. Regulatory compliance requirements vary significantly across different jurisdictions, creating additional complexity for commercial deployment.
The heterogeneous nature of current research approaches has resulted in fragmented knowledge bases, with limited standardization in experimental protocols and performance metrics. This situation complicates comparative studies and slows overall progress in the field.
Existing Sodium Nitrate Catalytic Solutions
01 Catalytic acceleration using metal compounds
The reaction rate of sodium nitrate can be enhanced through the use of metal-based catalysts or metal compound additives. These catalysts facilitate the decomposition or conversion reactions of sodium nitrate by lowering activation energy and providing active sites for the reaction. Various transition metals and their oxides have been found effective in accelerating sodium nitrate reactions under different temperature and pressure conditions.- Catalytic acceleration of sodium nitrate reactions: Various catalysts can be employed to accelerate reactions involving sodium nitrate. These catalysts work by lowering the activation energy required for the reaction to proceed, thereby increasing the reaction rate. Metal oxides, transition metal compounds, and other catalytic materials can be used to enhance the decomposition or conversion of sodium nitrate in chemical processes. The selection of appropriate catalysts depends on the specific reaction conditions and desired products.
- Temperature control for sodium nitrate reaction enhancement: Controlling the temperature is a critical factor in accelerating sodium nitrate reactions. Elevated temperatures generally increase the kinetic energy of molecules, leading to more frequent and energetic collisions between reactants. Optimal temperature ranges can be established for specific sodium nitrate reactions to maximize reaction rates while maintaining product quality and safety. Temperature management systems and heating methods can be implemented to achieve the desired acceleration effects.
- Use of additives and co-reactants: The addition of specific chemical compounds or co-reactants can significantly accelerate sodium nitrate reactions. These additives may include acids, bases, reducing agents, or other reactive species that participate in or facilitate the reaction mechanism. The additives can modify the reaction pathway, stabilize intermediate products, or create more favorable reaction conditions. Proper selection and dosing of additives are essential for achieving optimal acceleration while avoiding unwanted side reactions.
- Reactor design and mixing optimization: The design of reaction vessels and optimization of mixing conditions play important roles in accelerating sodium nitrate reactions. Enhanced mixing ensures better contact between reactants and uniform distribution of heat and catalysts throughout the reaction medium. Specialized reactor configurations, such as continuous flow reactors, stirred tank reactors, or fluidized bed reactors, can be employed to improve mass transfer and reaction efficiency. Proper agitation methods and equipment design contribute to faster reaction rates and improved yields.
- Pressure modification for reaction acceleration: Adjusting the pressure conditions can effectively accelerate certain sodium nitrate reactions. Increased pressure can enhance the concentration of gaseous reactants or products, shift equilibrium positions, and increase collision frequencies between molecules. Pressure control systems and specialized high-pressure reactors may be utilized to create optimal conditions for accelerated reactions. The pressure effects must be carefully balanced with safety considerations and equipment limitations to achieve the desired acceleration while maintaining process control.
02 Temperature and pressure optimization
Acceleration of sodium nitrate reactions can be achieved by optimizing reaction conditions, particularly temperature and pressure parameters. Elevated temperatures generally increase reaction kinetics, while controlled pressure conditions can influence the reaction pathway and rate. Process designs incorporating specific temperature ranges and pressure controls have been developed to maximize reaction efficiency and product yield.Expand Specific Solutions03 Co-reactant and additive systems
The introduction of specific co-reactants or chemical additives can significantly accelerate sodium nitrate reactions. These substances may include organic compounds, acids, bases, or other inorganic salts that interact with sodium nitrate to promote faster reaction rates. The selection of appropriate additives depends on the desired reaction products and process conditions.Expand Specific Solutions04 Reactor design and mixing enhancement
Specialized reactor configurations and improved mixing techniques can accelerate sodium nitrate reactions by enhancing mass transfer and heat distribution. Equipment designs featuring optimized flow patterns, agitation systems, and contact surfaces promote more efficient reactions. These engineering approaches ensure better reactant contact and uniform reaction conditions throughout the process.Expand Specific Solutions05 Microwave and electromagnetic field assistance
The application of microwave radiation or electromagnetic fields represents an innovative approach to accelerating sodium nitrate reactions. These energy sources provide rapid and uniform heating, selective activation of reactant molecules, and enhanced reaction kinetics compared to conventional heating methods. This technology enables faster processing times and improved energy efficiency in sodium nitrate conversion processes.Expand Specific Solutions
Major Players in Nitrate Catalysis Industry
The sodium nitrate catalysis field represents an emerging technology area in the early development stage, with significant growth potential driven by increasing demand for sustainable and efficient catalytic processes. The market remains relatively niche but shows promising expansion as industries seek cost-effective alternatives to traditional catalysts. Technology maturity varies considerably across different applications, with academic institutions like California Institute of Technology, Jilin University, and Southeast University leading fundamental research, while industrial players such as BASF Corp., China Petroleum & Chemical Corp., and Nippon Shokubai Co., Ltd. focus on commercial applications. The competitive landscape features a mix of established chemical companies, automotive manufacturers like Hyundai Motor Co. and Kia Corp. exploring catalytic applications, and research organizations including Southwest Research Institute and Council of Scientific & Industrial Research driving innovation. This diverse ecosystem indicates growing interest but suggests the technology is still transitioning from laboratory research to industrial implementation.
BASF Corp.
Technical Solution: BASF has developed advanced sodium nitrate-based catalytic systems for selective oxidation reactions, particularly in the production of fine chemicals and pharmaceuticals. Their proprietary technology utilizes sodium nitrate as both an oxidizing agent and catalyst promoter in multi-step organic synthesis processes. The company has implemented continuous flow reactor systems that optimize sodium nitrate concentration and temperature control to achieve enhanced reaction rates while maintaining product selectivity. Their research focuses on heterogeneous catalysis where sodium nitrate is supported on various metal oxide carriers to improve catalyst stability and recyclability in industrial applications.
Strengths: Extensive industrial experience and proven scalability in chemical manufacturing processes. Weaknesses: High energy requirements and potential environmental concerns with nitrate waste management.
Mitsubishi Kasei Corp.
Technical Solution: Mitsubishi Kasei has developed innovative sodium nitrate catalytic systems for environmental applications, particularly in NOx reduction and air purification processes. Their technology employs sodium nitrate as both a catalyst and reactant in selective catalytic reduction systems, achieving enhanced conversion rates for nitrogen oxide elimination. The company has created advanced formulations where sodium nitrate is combined with metal oxide supports to create highly active catalytic surfaces for automotive emission control and industrial flue gas treatment. Their approach focuses on optimizing sodium nitrate dispersion and thermal stability to maintain catalytic activity under harsh operating conditions.
Strengths: Advanced materials science capabilities and strong focus on environmental catalysis applications. Weaknesses: Relatively narrow application focus primarily in emission control rather than broader chemical synthesis.
Core Patents in Nitrate-Accelerated Reactions
Catalyst for electrochemical reaction capable of selective oxidation or reduction of active material and manufacturing method thereof
PatentPendingKR1020240070806A
Innovation
- A method involving the synthesis of a catalyst with different active materials, followed by heat treatment to provide fluidity, and then selectively oxidizing or reducing specific materials at temperatures below their respective oxidation or reduction temperatures, maintaining fluidity to achieve desired oxidation states.
Molten-salt type catalyst
PatentInactiveUS6777368B2
Innovation
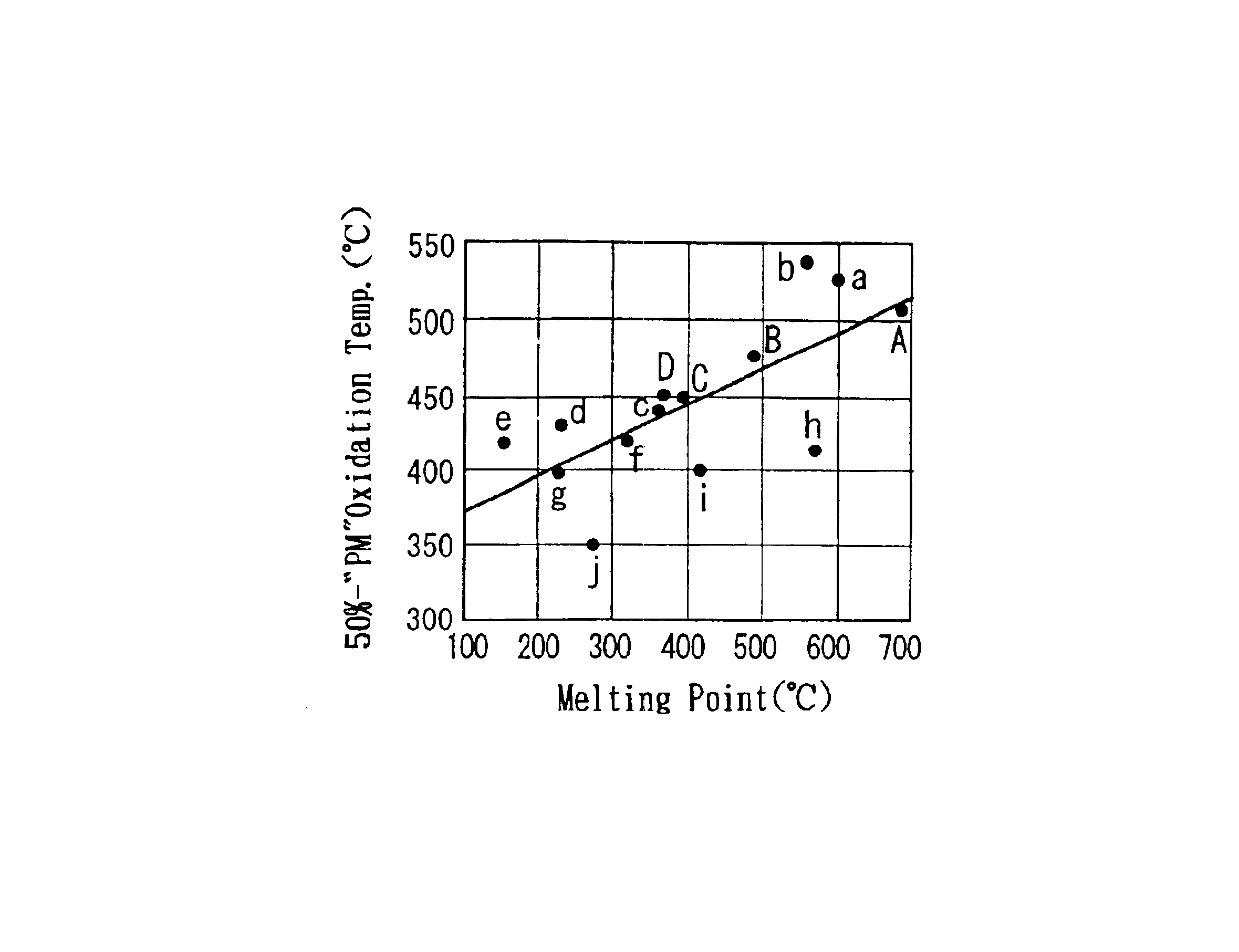
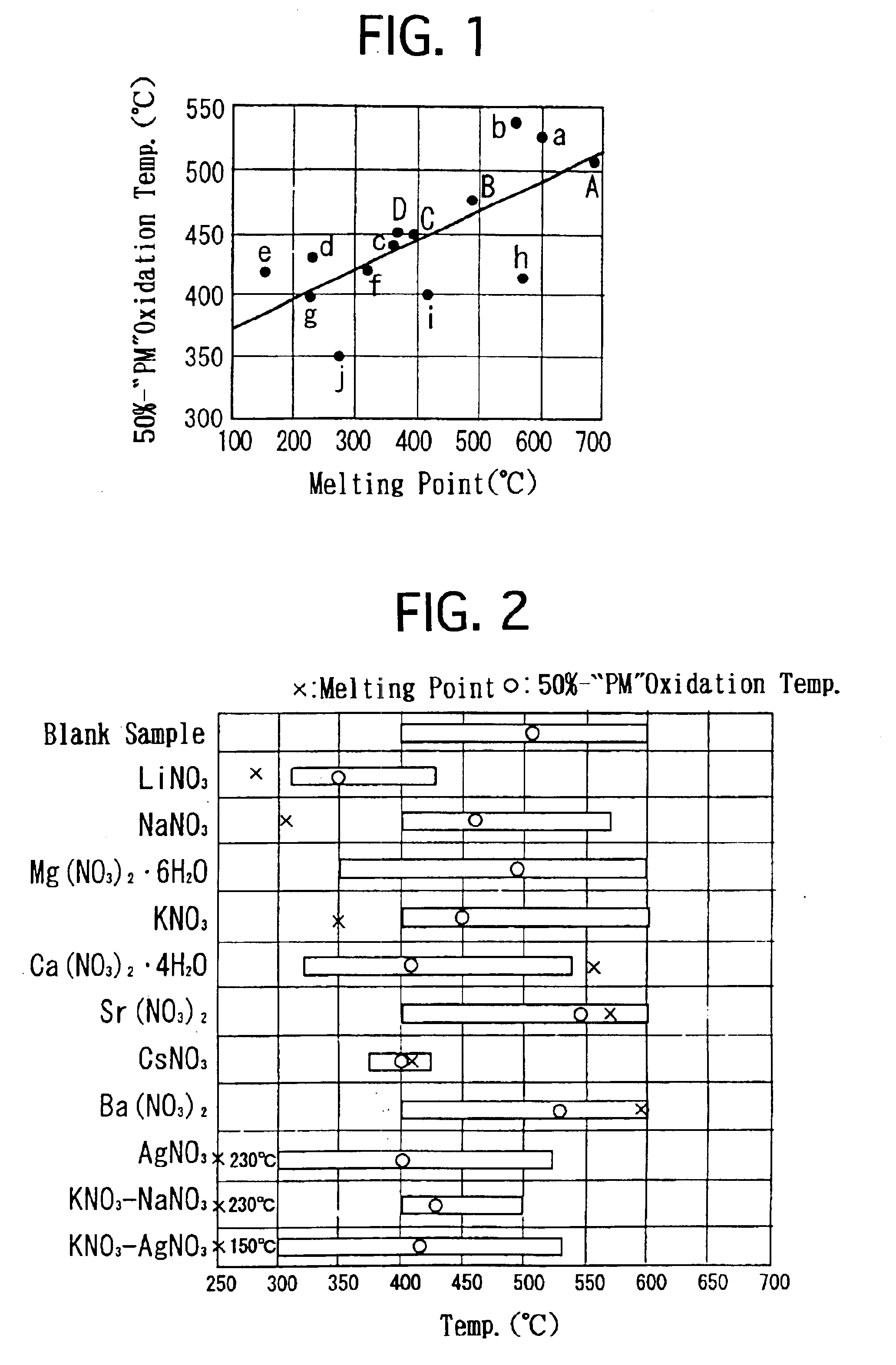
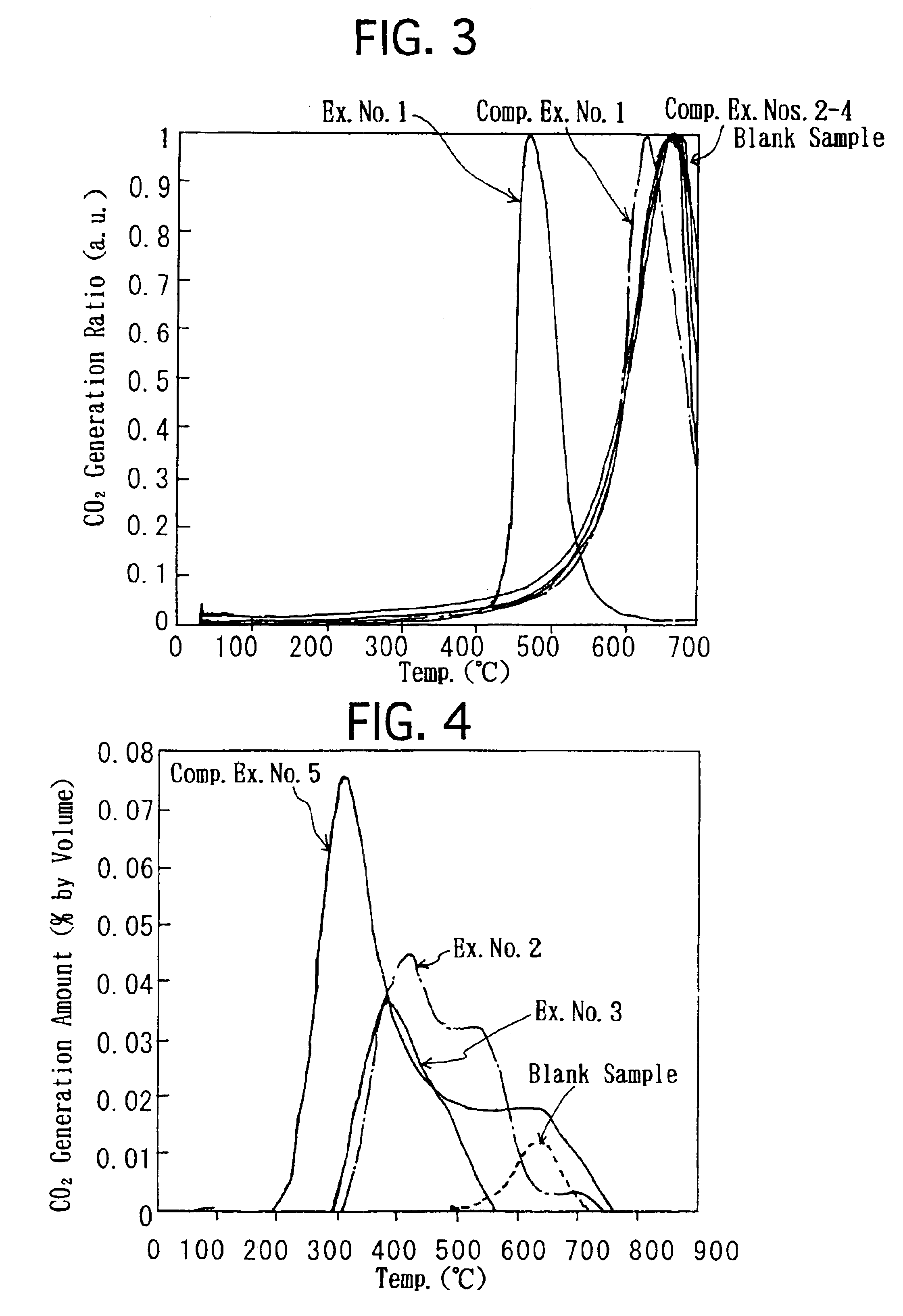
- A molten-salt type catalyst with a solid support and catalytic ingredients like silver nitrate, alkali metal nitrate, and rare-earth nitrate, which melts at low temperatures, ensuring adherence to the support and enhanced PM combustion probability, using a basic support like magnesia spinel to inhibit solid phase reactions and improve durability.
Environmental Impact of Nitrate Catalytic Processes
The environmental implications of sodium nitrate-based catalytic processes present a complex landscape of both benefits and challenges that require careful evaluation. While these catalytic systems offer significant advantages in reaction acceleration and efficiency, their environmental footprint encompasses multiple dimensions including resource consumption, waste generation, and ecosystem interactions.
Sodium nitrate catalytic processes demonstrate notable environmental advantages through enhanced reaction selectivity and reduced energy requirements. The improved catalytic efficiency typically translates to lower operating temperatures and shorter reaction times, resulting in decreased overall energy consumption compared to conventional thermal processes. This energy reduction directly correlates with reduced carbon emissions, particularly when fossil fuel-derived energy sources are involved in industrial operations.
The water solubility of sodium nitrate presents both environmental opportunities and concerns. On the positive side, this characteristic facilitates easier catalyst recovery and recycling through aqueous separation techniques, potentially reducing material waste. However, the same solubility raises concerns about potential groundwater contamination if proper containment and waste management protocols are not implemented. Nitrate leaching into water systems can contribute to eutrophication and pose risks to aquatic ecosystems.
Atmospheric emissions from nitrate catalytic processes require particular attention due to the potential formation of nitrogen oxides during high-temperature operations. These NOx compounds contribute to air quality degradation and can participate in photochemical smog formation. Advanced process design incorporating emission control systems becomes essential to mitigate these atmospheric impacts while maintaining catalytic performance.
The lifecycle assessment of sodium nitrate catalysts reveals important considerations regarding raw material sourcing and end-of-life management. Natural sodium nitrate deposits, primarily found in Chile, involve mining operations with associated environmental disturbances. Alternatively, synthetic production routes may offer more controlled environmental profiles but require careful evaluation of their own resource and energy demands.
Waste stream management in nitrate catalytic processes demands comprehensive strategies to prevent environmental contamination. Spent catalyst materials and reaction byproducts containing nitrate compounds require specialized treatment to prevent soil and water contamination. Implementation of closed-loop systems and advanced purification technologies can significantly reduce environmental discharge while improving overall process sustainability.
Sodium nitrate catalytic processes demonstrate notable environmental advantages through enhanced reaction selectivity and reduced energy requirements. The improved catalytic efficiency typically translates to lower operating temperatures and shorter reaction times, resulting in decreased overall energy consumption compared to conventional thermal processes. This energy reduction directly correlates with reduced carbon emissions, particularly when fossil fuel-derived energy sources are involved in industrial operations.
The water solubility of sodium nitrate presents both environmental opportunities and concerns. On the positive side, this characteristic facilitates easier catalyst recovery and recycling through aqueous separation techniques, potentially reducing material waste. However, the same solubility raises concerns about potential groundwater contamination if proper containment and waste management protocols are not implemented. Nitrate leaching into water systems can contribute to eutrophication and pose risks to aquatic ecosystems.
Atmospheric emissions from nitrate catalytic processes require particular attention due to the potential formation of nitrogen oxides during high-temperature operations. These NOx compounds contribute to air quality degradation and can participate in photochemical smog formation. Advanced process design incorporating emission control systems becomes essential to mitigate these atmospheric impacts while maintaining catalytic performance.
The lifecycle assessment of sodium nitrate catalysts reveals important considerations regarding raw material sourcing and end-of-life management. Natural sodium nitrate deposits, primarily found in Chile, involve mining operations with associated environmental disturbances. Alternatively, synthetic production routes may offer more controlled environmental profiles but require careful evaluation of their own resource and energy demands.
Waste stream management in nitrate catalytic processes demands comprehensive strategies to prevent environmental contamination. Spent catalyst materials and reaction byproducts containing nitrate compounds require specialized treatment to prevent soil and water contamination. Implementation of closed-loop systems and advanced purification technologies can significantly reduce environmental discharge while improving overall process sustainability.
Safety Protocols for Sodium Nitrate Handling
The handling of sodium nitrate in catalytic applications requires comprehensive safety protocols due to its oxidizing properties and potential health hazards. Sodium nitrate is classified as an oxidizing agent that can intensify fires and may cause or contribute to combustion of other materials. When used in catalytic processes for reaction acceleration, proper safety measures become critical to prevent accidents and ensure worker protection.
Personal protective equipment constitutes the first line of defense when handling sodium nitrate. Workers must wear appropriate respiratory protection, including NIOSH-approved dust masks or respirators when airborne particles are present. Eye protection through safety goggles or face shields is mandatory to prevent contact with eyes, which can cause severe irritation. Chemical-resistant gloves made of nitrile or neoprene should be worn to prevent skin contact, as sodium nitrate can cause dermatitis upon prolonged exposure.
Storage protocols require sodium nitrate to be kept in cool, dry, well-ventilated areas away from incompatible materials. The compound must be stored separately from organic materials, reducing agents, and combustible substances to prevent potential reactions. Containers should be tightly sealed and properly labeled with hazard warnings. Temperature control is essential, as elevated temperatures can increase the risk of decomposition and release of toxic nitrogen oxides.
Handling procedures during catalytic operations demand careful attention to prevent spills and minimize exposure. Work areas should be equipped with adequate ventilation systems to control airborne concentrations. Emergency eyewash stations and safety showers must be readily accessible within the immediate work area. All handling should occur in designated areas with appropriate containment measures to prevent environmental contamination.
Emergency response protocols must address potential exposure scenarios and spill situations. In case of skin or eye contact, immediate flushing with copious amounts of water for at least fifteen minutes is required, followed by medical attention. For inhalation exposure, affected individuals should be moved to fresh air immediately. Spill cleanup procedures involve using non-combustible absorbent materials while wearing full protective equipment, ensuring proper disposal according to local regulations.
Regular monitoring and maintenance of safety systems ensure continued protection throughout catalytic operations. Air quality monitoring should be conducted to verify that exposure limits are not exceeded. Safety equipment must be inspected regularly and replaced as needed. Training programs should be implemented to ensure all personnel understand proper handling procedures and emergency response protocols when working with sodium nitrate in catalytic applications.
Personal protective equipment constitutes the first line of defense when handling sodium nitrate. Workers must wear appropriate respiratory protection, including NIOSH-approved dust masks or respirators when airborne particles are present. Eye protection through safety goggles or face shields is mandatory to prevent contact with eyes, which can cause severe irritation. Chemical-resistant gloves made of nitrile or neoprene should be worn to prevent skin contact, as sodium nitrate can cause dermatitis upon prolonged exposure.
Storage protocols require sodium nitrate to be kept in cool, dry, well-ventilated areas away from incompatible materials. The compound must be stored separately from organic materials, reducing agents, and combustible substances to prevent potential reactions. Containers should be tightly sealed and properly labeled with hazard warnings. Temperature control is essential, as elevated temperatures can increase the risk of decomposition and release of toxic nitrogen oxides.
Handling procedures during catalytic operations demand careful attention to prevent spills and minimize exposure. Work areas should be equipped with adequate ventilation systems to control airborne concentrations. Emergency eyewash stations and safety showers must be readily accessible within the immediate work area. All handling should occur in designated areas with appropriate containment measures to prevent environmental contamination.
Emergency response protocols must address potential exposure scenarios and spill situations. In case of skin or eye contact, immediate flushing with copious amounts of water for at least fifteen minutes is required, followed by medical attention. For inhalation exposure, affected individuals should be moved to fresh air immediately. Spill cleanup procedures involve using non-combustible absorbent materials while wearing full protective equipment, ensuring proper disposal according to local regulations.
Regular monitoring and maintenance of safety systems ensure continued protection throughout catalytic operations. Air quality monitoring should be conducted to verify that exposure limits are not exceeded. Safety equipment must be inspected regularly and replaced as needed. Training programs should be implemented to ensure all personnel understand proper handling procedures and emergency response protocols when working with sodium nitrate in catalytic applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!