Wave Imaging vs Optical Coherence: Comparative Precision
MAR 9, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Wave Imaging and OCT Technology Background and Objectives
Wave imaging and optical coherence tomography (OCT) represent two distinct yet complementary approaches to high-resolution medical imaging, each leveraging different physical principles to achieve precise tissue visualization. Wave imaging encompasses various modalities including ultrasound, photoacoustic imaging, and elastography, which utilize mechanical or acoustic waves to penetrate tissues and generate detailed structural information. These technologies have evolved from basic ultrasound applications in the 1940s to sophisticated multi-dimensional imaging systems capable of real-time visualization with submillimeter resolution.
OCT technology emerged in the early 1990s as a revolutionary optical imaging technique that employs low-coherence interferometry to capture cross-sectional images of biological tissues. By measuring the time delay and intensity of backscattered light, OCT systems can achieve axial resolutions approaching 1-2 micrometers, significantly surpassing conventional imaging modalities. The technology has undergone substantial advancement from time-domain OCT to frequency-domain and swept-source implementations, dramatically improving imaging speed and sensitivity.
The comparative precision analysis between these technologies has become increasingly critical as medical applications demand higher accuracy for early disease detection, surgical guidance, and treatment monitoring. Wave imaging excels in deep tissue penetration, typically reaching depths of several centimeters with resolution ranging from 50-200 micrometers, making it invaluable for cardiac, abdominal, and vascular imaging applications.
OCT demonstrates superior resolution capabilities in superficial tissues, achieving cellular-level detail within the first 1-2 millimeters of tissue depth. This precision advantage has established OCT as the gold standard for retinal imaging, dermatological assessment, and intravascular plaque characterization. The technology's ability to perform functional imaging, including Doppler OCT and OCT angiography, further enhances its diagnostic capabilities.
The primary objective of this comparative analysis focuses on establishing quantitative precision benchmarks for both technologies across various clinical applications. Key performance metrics include spatial resolution, penetration depth, imaging speed, contrast sensitivity, and motion artifact susceptibility. Understanding these parameters enables optimal technology selection for specific diagnostic requirements and identifies potential areas for technological convergence or hybrid system development.
OCT technology emerged in the early 1990s as a revolutionary optical imaging technique that employs low-coherence interferometry to capture cross-sectional images of biological tissues. By measuring the time delay and intensity of backscattered light, OCT systems can achieve axial resolutions approaching 1-2 micrometers, significantly surpassing conventional imaging modalities. The technology has undergone substantial advancement from time-domain OCT to frequency-domain and swept-source implementations, dramatically improving imaging speed and sensitivity.
The comparative precision analysis between these technologies has become increasingly critical as medical applications demand higher accuracy for early disease detection, surgical guidance, and treatment monitoring. Wave imaging excels in deep tissue penetration, typically reaching depths of several centimeters with resolution ranging from 50-200 micrometers, making it invaluable for cardiac, abdominal, and vascular imaging applications.
OCT demonstrates superior resolution capabilities in superficial tissues, achieving cellular-level detail within the first 1-2 millimeters of tissue depth. This precision advantage has established OCT as the gold standard for retinal imaging, dermatological assessment, and intravascular plaque characterization. The technology's ability to perform functional imaging, including Doppler OCT and OCT angiography, further enhances its diagnostic capabilities.
The primary objective of this comparative analysis focuses on establishing quantitative precision benchmarks for both technologies across various clinical applications. Key performance metrics include spatial resolution, penetration depth, imaging speed, contrast sensitivity, and motion artifact susceptibility. Understanding these parameters enables optimal technology selection for specific diagnostic requirements and identifies potential areas for technological convergence or hybrid system development.
Market Demand for High-Precision Medical Imaging Solutions
The global medical imaging market is experiencing unprecedented growth driven by an aging population, increasing prevalence of chronic diseases, and rising demand for early disease detection. Healthcare providers worldwide are seeking advanced imaging solutions that can deliver superior diagnostic accuracy while reducing patient examination times and improving clinical workflows.
High-precision imaging technologies have become critical in multiple medical specialties, particularly in ophthalmology, cardiology, dermatology, and oncology. The demand for non-invasive diagnostic tools that can provide real-time, high-resolution imaging continues to escalate as healthcare systems prioritize preventive care and early intervention strategies.
Wave imaging and optical coherence technologies represent two distinct approaches to addressing precision imaging requirements. Wave imaging techniques, including advanced ultrasound and photoacoustic imaging, offer deep tissue penetration capabilities and real-time visualization of dynamic processes. These technologies are particularly valued in cardiovascular imaging, where precise measurement of blood flow and tissue elasticity is essential for accurate diagnosis.
Optical coherence tomography has established strong market presence in ophthalmology, where micron-level resolution is crucial for detecting retinal pathologies and monitoring treatment responses. The technology's ability to provide cross-sectional tissue imaging without contrast agents has driven adoption across multiple clinical applications, extending beyond traditional ophthalmic uses into gastroenterology, dermatology, and interventional cardiology.
Market demand patterns reveal distinct preferences based on clinical requirements. Healthcare facilities prioritizing deep tissue imaging and functional assessment tend to favor wave-based technologies, while institutions requiring ultra-high resolution surface and near-surface imaging gravitate toward optical coherence solutions. Cost considerations also influence adoption, as wave imaging systems often provide broader application versatility within single platforms.
Emerging markets demonstrate particularly strong growth potential, driven by expanding healthcare infrastructure and increasing access to advanced medical technologies. Developed markets show sustained demand for precision upgrades and multi-modal imaging capabilities that combine different technological approaches to maximize diagnostic information.
The competitive landscape reflects ongoing technological convergence, with manufacturers developing hybrid systems that integrate multiple imaging modalities. This trend indicates market recognition that optimal precision often requires complementary technologies rather than single-solution approaches, suggesting future growth opportunities for integrated imaging platforms.
High-precision imaging technologies have become critical in multiple medical specialties, particularly in ophthalmology, cardiology, dermatology, and oncology. The demand for non-invasive diagnostic tools that can provide real-time, high-resolution imaging continues to escalate as healthcare systems prioritize preventive care and early intervention strategies.
Wave imaging and optical coherence technologies represent two distinct approaches to addressing precision imaging requirements. Wave imaging techniques, including advanced ultrasound and photoacoustic imaging, offer deep tissue penetration capabilities and real-time visualization of dynamic processes. These technologies are particularly valued in cardiovascular imaging, where precise measurement of blood flow and tissue elasticity is essential for accurate diagnosis.
Optical coherence tomography has established strong market presence in ophthalmology, where micron-level resolution is crucial for detecting retinal pathologies and monitoring treatment responses. The technology's ability to provide cross-sectional tissue imaging without contrast agents has driven adoption across multiple clinical applications, extending beyond traditional ophthalmic uses into gastroenterology, dermatology, and interventional cardiology.
Market demand patterns reveal distinct preferences based on clinical requirements. Healthcare facilities prioritizing deep tissue imaging and functional assessment tend to favor wave-based technologies, while institutions requiring ultra-high resolution surface and near-surface imaging gravitate toward optical coherence solutions. Cost considerations also influence adoption, as wave imaging systems often provide broader application versatility within single platforms.
Emerging markets demonstrate particularly strong growth potential, driven by expanding healthcare infrastructure and increasing access to advanced medical technologies. Developed markets show sustained demand for precision upgrades and multi-modal imaging capabilities that combine different technological approaches to maximize diagnostic information.
The competitive landscape reflects ongoing technological convergence, with manufacturers developing hybrid systems that integrate multiple imaging modalities. This trend indicates market recognition that optimal precision often requires complementary technologies rather than single-solution approaches, suggesting future growth opportunities for integrated imaging platforms.
Current Status and Challenges in Wave vs OCT Imaging
Wave imaging and optical coherence tomography (OCT) represent two distinct yet complementary approaches to high-resolution biological imaging, each facing unique technical challenges in their pursuit of enhanced precision. Wave imaging technologies, encompassing ultrasound, photoacoustic, and elastography modalities, currently achieve spatial resolutions ranging from 50-200 micrometers depending on frequency and tissue depth. These systems struggle with fundamental trade-offs between penetration depth and resolution, as higher frequencies necessary for improved precision suffer from increased attenuation in biological tissues.
OCT systems have established themselves as the gold standard for superficial tissue imaging, delivering axial resolutions of 1-10 micrometers and lateral resolutions of 10-20 micrometers. However, OCT faces significant limitations in imaging depth, typically restricted to 1-2 millimeters in highly scattering tissues due to multiple scattering effects and signal attenuation. Advanced OCT variants, including swept-source and spectral-domain configurations, have pushed these boundaries but remain constrained by fundamental physics of light-tissue interactions.
The current technological landscape reveals distinct geographical concentrations of expertise. North American institutions lead in OCT innovation, particularly in retinal imaging applications, while European research centers excel in industrial OCT implementations. Asian markets, especially Japan and South Korea, dominate wave imaging hardware manufacturing and piezoelectric transducer development. This distribution creates regional strengths but also technological silos that limit cross-pollination of innovations.
Signal processing represents a critical bottleneck for both modalities. Wave imaging systems face challenges in real-time beamforming algorithms, particularly for 3D volumetric imaging where computational demands increase exponentially. OCT systems struggle with speckle noise reduction and motion artifact correction, requiring sophisticated post-processing algorithms that often compromise imaging speed. Current processing capabilities limit real-time high-resolution imaging to 2D cross-sections rather than full volumetric reconstruction.
Integration challenges persist as clinical demands push toward multimodal imaging platforms. Combining wave-based and optical coherence approaches requires synchronization of disparate data acquisition rates, coordinate system alignment, and fusion of complementary contrast mechanisms. These technical hurdles, coupled with regulatory approval complexities for hybrid systems, significantly slow clinical translation despite promising research outcomes demonstrating enhanced diagnostic capabilities through combined modalities.
OCT systems have established themselves as the gold standard for superficial tissue imaging, delivering axial resolutions of 1-10 micrometers and lateral resolutions of 10-20 micrometers. However, OCT faces significant limitations in imaging depth, typically restricted to 1-2 millimeters in highly scattering tissues due to multiple scattering effects and signal attenuation. Advanced OCT variants, including swept-source and spectral-domain configurations, have pushed these boundaries but remain constrained by fundamental physics of light-tissue interactions.
The current technological landscape reveals distinct geographical concentrations of expertise. North American institutions lead in OCT innovation, particularly in retinal imaging applications, while European research centers excel in industrial OCT implementations. Asian markets, especially Japan and South Korea, dominate wave imaging hardware manufacturing and piezoelectric transducer development. This distribution creates regional strengths but also technological silos that limit cross-pollination of innovations.
Signal processing represents a critical bottleneck for both modalities. Wave imaging systems face challenges in real-time beamforming algorithms, particularly for 3D volumetric imaging where computational demands increase exponentially. OCT systems struggle with speckle noise reduction and motion artifact correction, requiring sophisticated post-processing algorithms that often compromise imaging speed. Current processing capabilities limit real-time high-resolution imaging to 2D cross-sections rather than full volumetric reconstruction.
Integration challenges persist as clinical demands push toward multimodal imaging platforms. Combining wave-based and optical coherence approaches requires synchronization of disparate data acquisition rates, coordinate system alignment, and fusion of complementary contrast mechanisms. These technical hurdles, coupled with regulatory approval complexities for hybrid systems, significantly slow clinical translation despite promising research outcomes demonstrating enhanced diagnostic capabilities through combined modalities.
Current Technical Solutions for Precision Enhancement
01 Optical coherence tomography systems with enhanced imaging precision
Advanced optical coherence tomography (OCT) systems utilize sophisticated algorithms and hardware configurations to achieve high-precision imaging. These systems employ techniques such as spectral domain analysis, swept-source technology, and adaptive optics to improve image resolution and depth penetration. The integration of real-time processing capabilities enables accurate visualization of tissue structures with minimal artifacts and enhanced signal-to-noise ratios.- Optical coherence tomography systems with enhanced imaging precision: Advanced optical coherence tomography (OCT) systems utilize sophisticated algorithms and hardware configurations to achieve high-precision imaging. These systems employ techniques such as spectral domain analysis, swept-source technology, and adaptive optics to improve image resolution and depth penetration. The integration of real-time processing capabilities enables accurate visualization of tissue structures with minimal artifacts and enhanced signal-to-noise ratios.
- Wave interference and coherence detection methods: Precision measurement techniques based on wave interference patterns and coherence detection enable accurate determination of optical path differences and phase information. These methods utilize interferometric principles to analyze coherent light sources, employing sophisticated detection schemes that can distinguish minute variations in optical signals. The technology finds applications in high-resolution imaging where coherence properties of light waves are exploited for enhanced measurement accuracy.
- Signal processing and image reconstruction algorithms: Advanced computational methods for processing optical coherence signals involve complex algorithms that reconstruct high-quality images from raw interferometric data. These techniques include Fourier domain processing, dispersion compensation, and noise reduction algorithms that enhance image clarity and diagnostic value. Machine learning and artificial intelligence approaches are increasingly integrated to optimize image quality and automate feature detection.
- Multi-modal and functional imaging integration: Integrated imaging systems combine optical coherence techniques with complementary modalities to provide comprehensive tissue characterization. These hybrid approaches merge structural imaging with functional information such as blood flow, elastography, or molecular contrast. The integration enables simultaneous acquisition of multiple tissue parameters, improving diagnostic capabilities and providing more complete anatomical and physiological information in a single examination.
- Miniaturized and catheter-based imaging probes: Compact optical coherence imaging devices designed for minimally invasive applications utilize miniaturized optical components and flexible delivery systems. These probes enable in vivo imaging in confined spaces such as blood vessels, airways, and other internal body cavities. The technology incorporates micro-optics, fiber-based light delivery, and rotational or linear scanning mechanisms to achieve high-resolution imaging in clinical interventional procedures.
02 Wave interference and coherence detection methods
Precision measurement techniques based on wave interference patterns and coherence detection enable accurate characterization of optical properties. These methods utilize interferometric principles to analyze phase relationships and temporal coherence of light waves. Advanced detection schemes incorporate balanced detection, heterodyne detection, and phase-sensitive approaches to extract high-fidelity information from interference signals, improving measurement accuracy and sensitivity.Expand Specific Solutions03 Image processing and reconstruction algorithms for coherence imaging
Sophisticated computational methods are employed to process and reconstruct images from coherence-based imaging systems. These algorithms include dispersion compensation, motion artifact correction, speckle reduction, and three-dimensional volumetric reconstruction techniques. Machine learning and artificial intelligence approaches are increasingly integrated to enhance image quality, automate analysis, and extract clinically relevant features from complex datasets.Expand Specific Solutions04 Multi-modal and functional optical coherence imaging
Advanced imaging systems combine multiple imaging modalities and functional imaging capabilities to provide comprehensive tissue characterization. These systems integrate techniques such as Doppler imaging for flow visualization, polarization-sensitive imaging for birefringence mapping, and spectroscopic analysis for molecular composition assessment. The multi-modal approach enables simultaneous acquisition of structural and functional information, enhancing diagnostic capabilities.Expand Specific Solutions05 Miniaturized and catheter-based coherence imaging devices
Compact optical coherence imaging devices designed for minimally invasive applications utilize miniaturized optical components and flexible delivery systems. These devices incorporate micro-optics, fiber-optic probes, and rotational scanning mechanisms to enable intravascular, endoscopic, and surgical imaging applications. Advanced packaging techniques and biocompatible materials ensure safe deployment while maintaining high imaging performance in confined spaces.Expand Specific Solutions
Major Players in Wave Imaging and OCT Industry
The wave imaging versus optical coherence precision comparison represents a mature technological landscape in the advanced development stage, with significant market penetration across medical diagnostics and industrial applications. The global market demonstrates substantial growth driven by increasing demand for high-precision imaging solutions in healthcare and manufacturing sectors. Technology maturity varies significantly among key players, with established leaders like Carl Zeiss Meditec, Canon, Philips, and FUJIFILM demonstrating advanced optical coherence tomography capabilities, while companies such as Nidek, Terumo, and Excelitas Technologies focus on specialized wave imaging applications. Research institutions including Northwestern University, Caltech, and National University of Singapore contribute fundamental innovations, while emerging players like Shanghai Microelectronics and Appotronics drive competitive dynamics through novel approaches, creating a diverse ecosystem spanning from fundamental research to commercial implementation.
Excelitas Technologies Corp.
Technical Solution: Excelitas Technologies develops photonic components and systems for both wave imaging and optical coherence applications, focusing on superluminescent diodes (SLDs) and swept-source lasers for OCT systems[4]. Their swept-source technology operates at 1310nm wavelength with tuning ranges exceeding 100nm, enabling enhanced tissue penetration and reduced scattering compared to 840nm systems[5]. The company's precision optics and detector arrays support wave imaging applications with quantum efficiency exceeding 90% in near-infrared ranges, while their OCT light sources provide coherence lengths optimized for 3-6mm imaging depths with phase stability better than λ/50[6].
Strengths: Comprehensive component portfolio supporting both imaging modalities with high-performance light sources. Weaknesses: Component supplier rather than complete system integrator, limiting direct market presence.
Carl Zeiss Meditec, Inc.
Technical Solution: Carl Zeiss Meditec has developed advanced optical coherence tomography (OCT) systems that achieve axial resolution of 5-7 micrometers and lateral resolution of 10-20 micrometers for retinal imaging[1]. Their Cirrus HD-OCT platform incorporates spectral-domain technology with acquisition speeds up to 27,000 A-scans per second, enabling high-precision 3D imaging of ocular structures[2]. The company's OCT systems utilize superluminescent diode light sources with center wavelengths around 840nm, providing penetration depths of 2-3mm in tissue while maintaining coherence length optimization for enhanced image quality[3].
Strengths: Market-leading OCT technology with exceptional axial resolution and established clinical validation. Weaknesses: Limited penetration depth compared to wave imaging modalities and higher system costs.
Core Patents in Wave Imaging vs OCT Precision
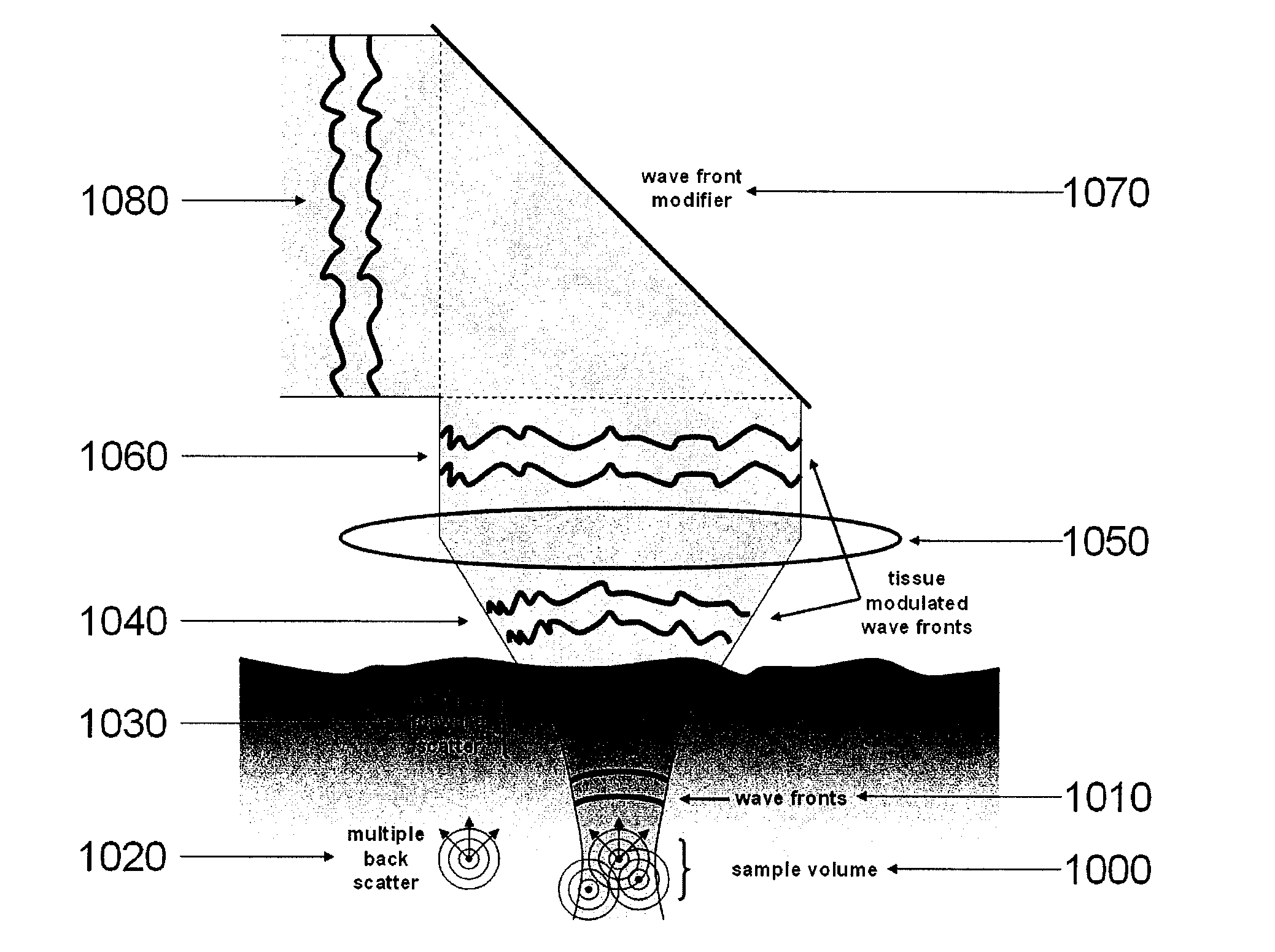
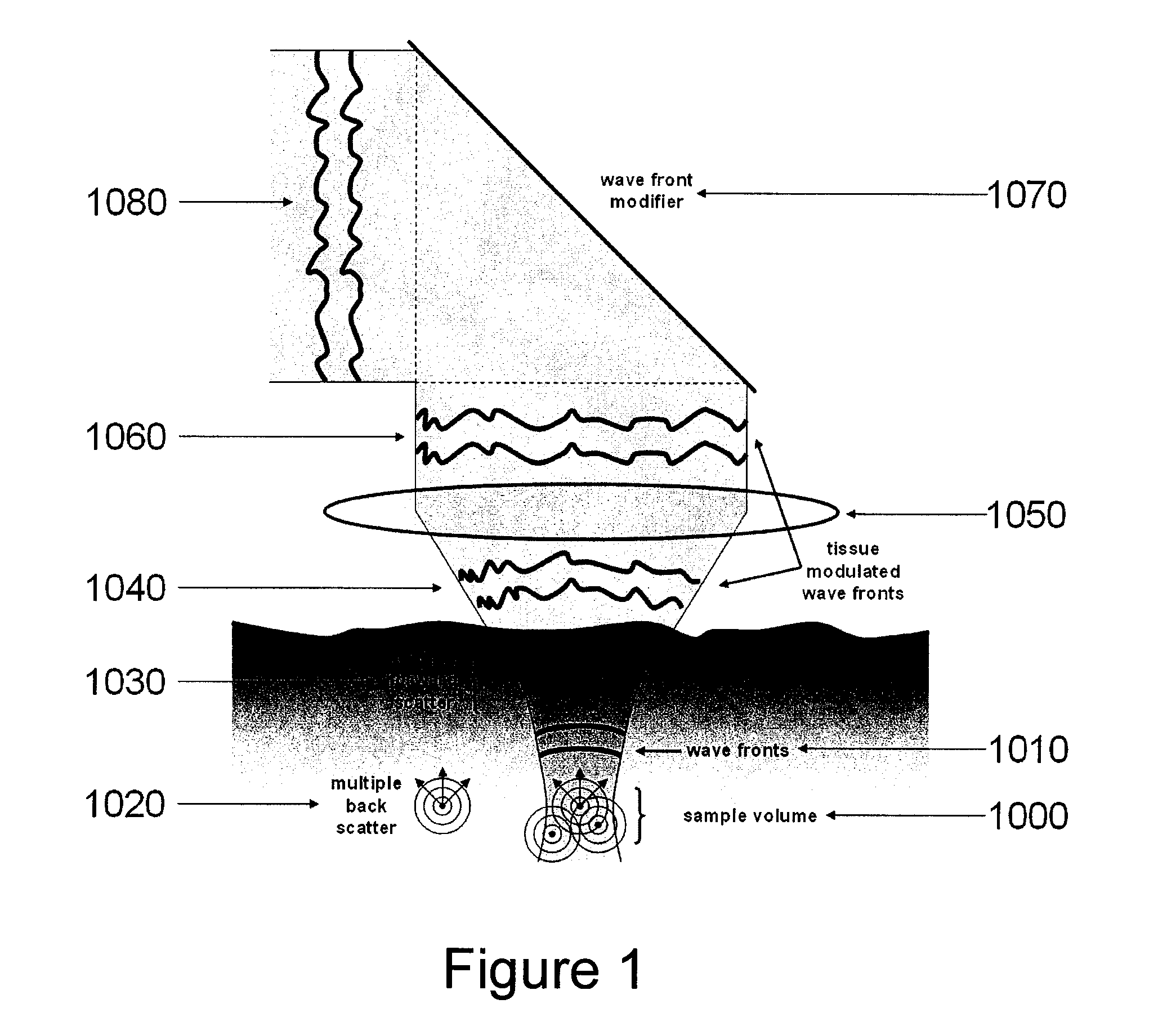
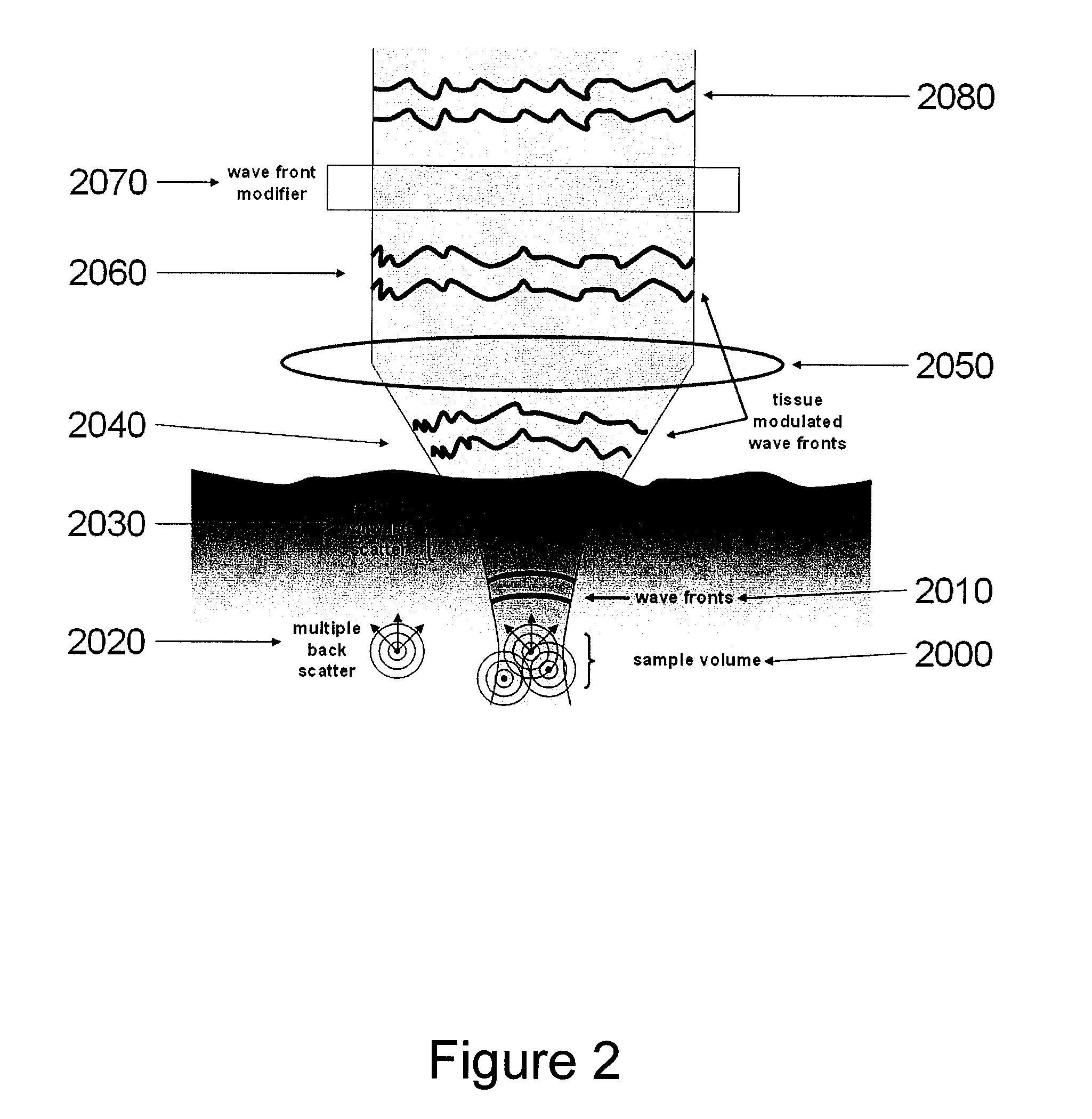
System, arrangement and process for providing speckle reductions using a wave front modulation for optical coherence tomography
PatentInactiveUS20080002211A1
Innovation
- A wave front modification technique is introduced in the sample arm beam path of OCT systems, allowing for averaging of small numbers of depth scans with different speckle patterns to reduce speckle contrast within a single image, without significant loss in axial or lateral resolution, using elements like micro-deformable mirrors to alter the wave front of light returning from the sample.
System for fourier domain optical coherence tomography
PatentInactiveUS20040239938A1
Innovation
- The implementation of a Fourier-domain OCT (FDOCT) system that eliminates the need for a mechanically scanned reference delay, utilizes array detection technologies, and acquires signals from all illuminated axial pixels simultaneously, employing multiple reference phases and orthogonal polarization channels to encode phase delays, and photodiode arrays for optimal signal-to-noise ratio.
Clinical Validation Standards for Imaging Technologies
Clinical validation of imaging technologies requires adherence to rigorous standards that ensure both safety and efficacy in medical applications. For wave imaging and optical coherence technologies, validation frameworks must address the unique characteristics and performance metrics of each modality while maintaining consistency with established regulatory guidelines.
The foundation of clinical validation begins with preclinical testing phases that evaluate fundamental imaging parameters including resolution, penetration depth, signal-to-noise ratio, and acquisition speed. Wave imaging technologies typically undergo validation through phantom studies using tissue-mimicking materials that replicate acoustic properties of human organs. These studies establish baseline performance metrics and identify optimal imaging parameters for specific clinical applications.
Optical coherence tomography validation follows similar principles but emphasizes different parameters such as axial and lateral resolution, imaging depth limitations, and tissue scattering characteristics. Standardized test protocols must account for wavelength-dependent tissue interactions and establish consistent measurement methodologies across different OCT system configurations.
Clinical trial design for imaging technology validation requires careful consideration of patient population selection, imaging protocol standardization, and outcome measurement criteria. Multi-center studies become essential for establishing technology reliability across diverse clinical environments and patient demographics. These trials must demonstrate not only technical performance but also clinical utility in improving diagnostic accuracy or patient outcomes.
Regulatory compliance frameworks vary significantly across global markets, with FDA 510(k) clearance, CE marking, and other regional approvals requiring specific documentation of clinical validation data. Each regulatory pathway demands comprehensive evidence packages that include clinical performance data, safety profiles, and comparative effectiveness studies against established imaging standards.
Quality assurance protocols form a critical component of validation standards, encompassing regular calibration procedures, performance monitoring systems, and maintenance protocols. These standards ensure consistent imaging quality throughout the technology's clinical lifecycle and provide frameworks for identifying and addressing performance degradation over time.
Comparative validation studies between wave imaging and optical coherence technologies require standardized metrics that enable meaningful performance comparisons. These studies must account for application-specific requirements and establish clear criteria for technology selection based on clinical needs rather than purely technical specifications.
The foundation of clinical validation begins with preclinical testing phases that evaluate fundamental imaging parameters including resolution, penetration depth, signal-to-noise ratio, and acquisition speed. Wave imaging technologies typically undergo validation through phantom studies using tissue-mimicking materials that replicate acoustic properties of human organs. These studies establish baseline performance metrics and identify optimal imaging parameters for specific clinical applications.
Optical coherence tomography validation follows similar principles but emphasizes different parameters such as axial and lateral resolution, imaging depth limitations, and tissue scattering characteristics. Standardized test protocols must account for wavelength-dependent tissue interactions and establish consistent measurement methodologies across different OCT system configurations.
Clinical trial design for imaging technology validation requires careful consideration of patient population selection, imaging protocol standardization, and outcome measurement criteria. Multi-center studies become essential for establishing technology reliability across diverse clinical environments and patient demographics. These trials must demonstrate not only technical performance but also clinical utility in improving diagnostic accuracy or patient outcomes.
Regulatory compliance frameworks vary significantly across global markets, with FDA 510(k) clearance, CE marking, and other regional approvals requiring specific documentation of clinical validation data. Each regulatory pathway demands comprehensive evidence packages that include clinical performance data, safety profiles, and comparative effectiveness studies against established imaging standards.
Quality assurance protocols form a critical component of validation standards, encompassing regular calibration procedures, performance monitoring systems, and maintenance protocols. These standards ensure consistent imaging quality throughout the technology's clinical lifecycle and provide frameworks for identifying and addressing performance degradation over time.
Comparative validation studies between wave imaging and optical coherence technologies require standardized metrics that enable meaningful performance comparisons. These studies must account for application-specific requirements and establish clear criteria for technology selection based on clinical needs rather than purely technical specifications.
Cost-Benefit Analysis of Wave vs OCT Systems
The economic evaluation of wave imaging and optical coherence tomography systems reveals significant disparities in initial capital investment requirements. Wave imaging systems typically demand lower upfront costs, with basic ultrasound equipment ranging from $50,000 to $200,000 for mid-range clinical units. In contrast, OCT systems require substantially higher initial investments, with advanced spectral-domain OCT units costing between $150,000 to $500,000, while swept-source OCT systems can exceed $800,000 for premium configurations.
Operational expenditure analysis demonstrates contrasting cost structures between the two technologies. Wave imaging systems benefit from lower maintenance costs due to simpler mechanical components and established service networks. Consumable costs remain minimal, primarily involving ultrasound gel and probe covers. OCT systems incur higher operational expenses through specialized maintenance contracts, frequent software updates, and replacement of sophisticated optical components including laser sources and photodetectors.
Personnel training investments vary considerably between platforms. Wave imaging leverages existing ultrasound expertise within medical facilities, requiring minimal additional training for basic applications. OCT technology demands specialized training programs costing $10,000 to $25,000 per technician, given the complexity of image interpretation and system optimization protocols.
Revenue generation potential differs substantially across clinical applications. OCT systems command premium reimbursement rates in ophthalmology and cardiology, with procedure fees ranging from $200 to $800 per examination. Wave imaging generates moderate revenue streams through high-volume applications, typically earning $100 to $300 per procedure but compensating through increased patient throughput.
Return on investment calculations favor OCT systems in specialized high-value applications despite higher initial costs. Premium OCT installations achieve payback periods of 18-24 months in busy ophthalmology practices. Wave imaging systems demonstrate consistent but modest returns, typically achieving payback within 12-18 months through volume-based utilization across diverse clinical scenarios.
Long-term economic sustainability analysis indicates OCT systems maintain competitive advantages in precision-critical applications where diagnostic accuracy justifies premium pricing. Wave imaging systems excel in cost-sensitive environments requiring broad diagnostic capabilities with acceptable precision thresholds, particularly in primary care and emergency medicine settings.
Operational expenditure analysis demonstrates contrasting cost structures between the two technologies. Wave imaging systems benefit from lower maintenance costs due to simpler mechanical components and established service networks. Consumable costs remain minimal, primarily involving ultrasound gel and probe covers. OCT systems incur higher operational expenses through specialized maintenance contracts, frequent software updates, and replacement of sophisticated optical components including laser sources and photodetectors.
Personnel training investments vary considerably between platforms. Wave imaging leverages existing ultrasound expertise within medical facilities, requiring minimal additional training for basic applications. OCT technology demands specialized training programs costing $10,000 to $25,000 per technician, given the complexity of image interpretation and system optimization protocols.
Revenue generation potential differs substantially across clinical applications. OCT systems command premium reimbursement rates in ophthalmology and cardiology, with procedure fees ranging from $200 to $800 per examination. Wave imaging generates moderate revenue streams through high-volume applications, typically earning $100 to $300 per procedure but compensating through increased patient throughput.
Return on investment calculations favor OCT systems in specialized high-value applications despite higher initial costs. Premium OCT installations achieve payback periods of 18-24 months in busy ophthalmology practices. Wave imaging systems demonstrate consistent but modest returns, typically achieving payback within 12-18 months through volume-based utilization across diverse clinical scenarios.
Long-term economic sustainability analysis indicates OCT systems maintain competitive advantages in precision-critical applications where diagnostic accuracy justifies premium pricing. Wave imaging systems excel in cost-sensitive environments requiring broad diagnostic capabilities with acceptable precision thresholds, particularly in primary care and emergency medicine settings.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!