Zinc vs Polymer-based: Recharge Reliability
APR 17, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Zinc vs Polymer Battery Tech Background and Goals
Battery technology has undergone significant evolution since the early 19th century, with zinc-based systems representing one of the oldest electrochemical energy storage approaches. Zinc batteries emerged in the 1860s with the Leclanché cell, establishing zinc as a reliable anode material due to its high theoretical capacity and natural abundance. The technology progressed through various iterations including zinc-carbon, zinc-chloride, and modern zinc-air configurations.
Polymer-based battery technologies emerged much later, gaining prominence in the 1970s with the development of conductive polymers. These systems utilize organic polymer materials as active components, offering unique advantages in terms of flexibility, lightweight design, and potentially sustainable manufacturing processes. The polymer approach represents a paradigm shift from traditional inorganic electrode materials toward organic electrochemistry.
The fundamental challenge of recharge reliability has become increasingly critical as energy storage applications demand higher cycle life and consistent performance. Traditional zinc batteries historically suffered from dendrite formation, shape change, and electrolyte degradation during repeated charge-discharge cycles. These phenomena significantly limit cycle life and create safety concerns in rechargeable applications.
Polymer-based systems face different reliability challenges, including polymer degradation, limited ionic conductivity, and structural instability during cycling. However, recent advances in polymer chemistry and nanostructuring have opened new pathways for addressing these limitations through molecular design and composite architectures.
Current technological objectives focus on achieving commercially viable rechargeable systems with extended cycle life exceeding 1000 cycles while maintaining capacity retention above 80%. For zinc-based systems, research targets include developing stable electrolyte formulations, advanced separator technologies, and novel anode architectures to suppress dendrite growth and minimize side reactions.
Polymer battery development aims to enhance ionic conductivity through advanced polymer electrolytes, improve mechanical stability via cross-linking strategies, and optimize redox-active polymer structures for reversible charge storage. The integration of nanotechnology and hybrid organic-inorganic approaches represents a promising direction for both technology platforms.
The ultimate goal involves establishing reliable, cost-effective rechargeable battery systems that can compete with lithium-ion technology in specific applications while offering unique advantages such as environmental sustainability, resource abundance, and enhanced safety profiles. Success in achieving superior recharge reliability will determine the commercial viability and market penetration potential of these alternative battery technologies.
Polymer-based battery technologies emerged much later, gaining prominence in the 1970s with the development of conductive polymers. These systems utilize organic polymer materials as active components, offering unique advantages in terms of flexibility, lightweight design, and potentially sustainable manufacturing processes. The polymer approach represents a paradigm shift from traditional inorganic electrode materials toward organic electrochemistry.
The fundamental challenge of recharge reliability has become increasingly critical as energy storage applications demand higher cycle life and consistent performance. Traditional zinc batteries historically suffered from dendrite formation, shape change, and electrolyte degradation during repeated charge-discharge cycles. These phenomena significantly limit cycle life and create safety concerns in rechargeable applications.
Polymer-based systems face different reliability challenges, including polymer degradation, limited ionic conductivity, and structural instability during cycling. However, recent advances in polymer chemistry and nanostructuring have opened new pathways for addressing these limitations through molecular design and composite architectures.
Current technological objectives focus on achieving commercially viable rechargeable systems with extended cycle life exceeding 1000 cycles while maintaining capacity retention above 80%. For zinc-based systems, research targets include developing stable electrolyte formulations, advanced separator technologies, and novel anode architectures to suppress dendrite growth and minimize side reactions.
Polymer battery development aims to enhance ionic conductivity through advanced polymer electrolytes, improve mechanical stability via cross-linking strategies, and optimize redox-active polymer structures for reversible charge storage. The integration of nanotechnology and hybrid organic-inorganic approaches represents a promising direction for both technology platforms.
The ultimate goal involves establishing reliable, cost-effective rechargeable battery systems that can compete with lithium-ion technology in specific applications while offering unique advantages such as environmental sustainability, resource abundance, and enhanced safety profiles. Success in achieving superior recharge reliability will determine the commercial viability and market penetration potential of these alternative battery technologies.
Market Demand for Rechargeable Battery Solutions
The global rechargeable battery market is experiencing unprecedented growth driven by the accelerating transition toward electrification across multiple sectors. Electric vehicles represent the largest demand driver, with automotive manufacturers worldwide committing to ambitious electrification timelines. Consumer electronics continue to demand higher energy density and longer-lasting power solutions, while grid-scale energy storage systems require reliable, cost-effective battery technologies to support renewable energy integration.
Traditional lithium-ion batteries currently dominate the rechargeable battery landscape, but emerging concerns about resource scarcity, cost volatility, and safety limitations are creating opportunities for alternative chemistries. The market is increasingly seeking battery solutions that offer improved recharge reliability, extended cycle life, and enhanced safety characteristics while maintaining competitive cost structures.
Zinc-based rechargeable batteries are gaining attention due to zinc's abundance, inherent safety properties, and potential for aqueous electrolyte systems. These characteristics address growing market demands for sustainable and safe energy storage solutions. The technology shows particular promise for stationary energy storage applications where weight is less critical than reliability and cost-effectiveness.
Polymer-based rechargeable battery systems, including solid polymer electrolytes and polymer-based active materials, are attracting significant market interest for their potential to address safety concerns associated with liquid electrolytes. The flexibility and processability of polymer materials enable innovative form factors and manufacturing approaches that could unlock new application areas in wearable electronics and flexible devices.
Market demand patterns reveal distinct preferences across application segments. Consumer electronics prioritize energy density and fast charging capabilities, while electric vehicle applications emphasize both energy density and charging reliability over extended operational lifespans. Grid storage applications focus primarily on cycle life, safety, and cost per kilowatt-hour, with less emphasis on energy density.
The reliability of recharge cycles has emerged as a critical market requirement across all segments. Battery degradation during repeated charge-discharge cycles directly impacts total cost of ownership and user experience. Market research indicates that recharge reliability, measured by capacity retention over thousands of cycles, has become a key differentiating factor influencing technology adoption decisions.
Regulatory frameworks worldwide are increasingly emphasizing battery sustainability, recyclability, and supply chain security. These regulatory trends are creating market opportunities for battery technologies that utilize abundant materials and offer simplified recycling pathways, potentially favoring zinc-based solutions over rare metal-dependent alternatives.
Traditional lithium-ion batteries currently dominate the rechargeable battery landscape, but emerging concerns about resource scarcity, cost volatility, and safety limitations are creating opportunities for alternative chemistries. The market is increasingly seeking battery solutions that offer improved recharge reliability, extended cycle life, and enhanced safety characteristics while maintaining competitive cost structures.
Zinc-based rechargeable batteries are gaining attention due to zinc's abundance, inherent safety properties, and potential for aqueous electrolyte systems. These characteristics address growing market demands for sustainable and safe energy storage solutions. The technology shows particular promise for stationary energy storage applications where weight is less critical than reliability and cost-effectiveness.
Polymer-based rechargeable battery systems, including solid polymer electrolytes and polymer-based active materials, are attracting significant market interest for their potential to address safety concerns associated with liquid electrolytes. The flexibility and processability of polymer materials enable innovative form factors and manufacturing approaches that could unlock new application areas in wearable electronics and flexible devices.
Market demand patterns reveal distinct preferences across application segments. Consumer electronics prioritize energy density and fast charging capabilities, while electric vehicle applications emphasize both energy density and charging reliability over extended operational lifespans. Grid storage applications focus primarily on cycle life, safety, and cost per kilowatt-hour, with less emphasis on energy density.
The reliability of recharge cycles has emerged as a critical market requirement across all segments. Battery degradation during repeated charge-discharge cycles directly impacts total cost of ownership and user experience. Market research indicates that recharge reliability, measured by capacity retention over thousands of cycles, has become a key differentiating factor influencing technology adoption decisions.
Regulatory frameworks worldwide are increasingly emphasizing battery sustainability, recyclability, and supply chain security. These regulatory trends are creating market opportunities for battery technologies that utilize abundant materials and offer simplified recycling pathways, potentially favoring zinc-based solutions over rare metal-dependent alternatives.
Current State of Zinc and Polymer Battery Reliability
Zinc-based batteries currently demonstrate moderate recharge reliability with significant variations across different chemistries. Zinc-air batteries face substantial challenges in rechargeability due to oxygen electrode degradation and electrolyte carbonation during cycling. Commercial zinc-air systems typically achieve 100-300 charge-discharge cycles before capacity drops below 80% of initial performance. Zinc-ion batteries show more promising results, with aqueous systems reaching 1,000-3,000 cycles, though performance varies significantly based on electrolyte composition and electrode materials.
Polymer-based battery technologies exhibit superior recharge reliability in most commercial applications. Lithium polymer batteries routinely achieve 500-1,500 cycles while maintaining 80% capacity retention under standard operating conditions. Advanced polymer electrolyte formulations have pushed cycle life beyond 2,000 cycles in laboratory settings. Solid polymer electrolytes demonstrate exceptional stability during repeated charging, with minimal dendrite formation and reduced side reactions compared to liquid electrolytes.
Key reliability factors affecting zinc systems include zinc dendrite formation, electrode corrosion, and electrolyte stability. Dendrite growth causes internal short circuits and capacity fade, particularly problematic in alkaline zinc systems. Corrosion of zinc anodes leads to hydrogen gas evolution and electrolyte pH changes, compromising long-term performance. Recent developments in zinc electrode surface treatments and electrolyte additives have shown promise in mitigating these issues.
Polymer battery reliability benefits from stable electrode-electrolyte interfaces and controlled ion transport mechanisms. Polymer matrices provide mechanical stability that prevents electrode degradation during volume changes. However, polymer systems face challenges including temperature sensitivity, ionic conductivity limitations, and potential polymer degradation under extreme conditions.
Current testing standards reveal disparate performance metrics between technologies. Zinc batteries often undergo accelerated aging tests showing rapid initial capacity loss followed by stabilization. Polymer systems typically exhibit more gradual, linear capacity decline over extended cycling periods. Temperature cycling tests particularly highlight zinc battery vulnerabilities, with performance degradation accelerating above 40°C, while polymer systems maintain more consistent behavior across broader temperature ranges.
Manufacturing quality control significantly impacts reliability outcomes for both technologies. Zinc battery consistency suffers from electrode uniformity challenges and electrolyte contamination sensitivity. Polymer battery reliability depends heavily on precise polymer film thickness control and electrode-polymer interface optimization during manufacturing processes.
Polymer-based battery technologies exhibit superior recharge reliability in most commercial applications. Lithium polymer batteries routinely achieve 500-1,500 cycles while maintaining 80% capacity retention under standard operating conditions. Advanced polymer electrolyte formulations have pushed cycle life beyond 2,000 cycles in laboratory settings. Solid polymer electrolytes demonstrate exceptional stability during repeated charging, with minimal dendrite formation and reduced side reactions compared to liquid electrolytes.
Key reliability factors affecting zinc systems include zinc dendrite formation, electrode corrosion, and electrolyte stability. Dendrite growth causes internal short circuits and capacity fade, particularly problematic in alkaline zinc systems. Corrosion of zinc anodes leads to hydrogen gas evolution and electrolyte pH changes, compromising long-term performance. Recent developments in zinc electrode surface treatments and electrolyte additives have shown promise in mitigating these issues.
Polymer battery reliability benefits from stable electrode-electrolyte interfaces and controlled ion transport mechanisms. Polymer matrices provide mechanical stability that prevents electrode degradation during volume changes. However, polymer systems face challenges including temperature sensitivity, ionic conductivity limitations, and potential polymer degradation under extreme conditions.
Current testing standards reveal disparate performance metrics between technologies. Zinc batteries often undergo accelerated aging tests showing rapid initial capacity loss followed by stabilization. Polymer systems typically exhibit more gradual, linear capacity decline over extended cycling periods. Temperature cycling tests particularly highlight zinc battery vulnerabilities, with performance degradation accelerating above 40°C, while polymer systems maintain more consistent behavior across broader temperature ranges.
Manufacturing quality control significantly impacts reliability outcomes for both technologies. Zinc battery consistency suffers from electrode uniformity challenges and electrolyte contamination sensitivity. Polymer battery reliability depends heavily on precise polymer film thickness control and electrode-polymer interface optimization during manufacturing processes.
Existing Solutions for Battery Recharge Reliability
01 Zinc-based battery recharge cycle improvements
Technologies focused on enhancing the rechargeability and cycle life of zinc-based batteries through electrode modifications, electrolyte optimization, and separator designs. These improvements address dendrite formation, shape change, and capacity fade issues that traditionally limit zinc battery recharge reliability. Advanced zinc electrode compositions and structures enable extended cycle life comparable to other rechargeable battery systems.- Zinc-based battery recharge cycle improvements: Technologies focused on enhancing the recharge reliability and cycle life of zinc-based batteries through improved electrode materials, electrolyte formulations, and charging protocols. These innovations address dendrite formation, electrode degradation, and capacity fade during repeated charge-discharge cycles to extend battery lifespan and maintain performance consistency.
- Polymer electrolyte battery charging stability: Developments in polymer-based battery systems that utilize solid or gel polymer electrolytes to improve recharge reliability. These technologies focus on maintaining ionic conductivity, preventing electrolyte decomposition, and ensuring stable interface properties during charging cycles. The polymer matrix provides enhanced safety and dimensional stability compared to liquid electrolytes.
- Comparative electrode degradation mechanisms: Analysis of degradation patterns in zinc versus polymer-based battery electrodes during recharge cycles. Research addresses issues such as zinc dissolution and redeposition, polymer swelling, and structural changes that affect long-term reliability. Solutions include protective coatings, modified electrode architectures, and optimized current densities to minimize degradation.
- Charge management systems for different battery chemistries: Specialized charging control methods and circuits designed to optimize recharge reliability for both zinc and polymer-based batteries. These systems monitor voltage, current, and temperature parameters to prevent overcharging, manage charge acceptance rates, and adapt charging profiles to specific battery chemistry requirements for maximum cycle life.
- Hybrid and composite battery architectures: Innovative battery designs that combine zinc and polymer technologies or incorporate composite materials to leverage advantages of both systems. These approaches aim to achieve superior recharge reliability by balancing the high energy density of zinc with the stability and safety characteristics of polymer-based systems through novel cell configurations and material combinations.
02 Polymer electrolyte systems for rechargeable batteries
Development of polymer-based electrolyte systems that improve battery recharge reliability through enhanced ionic conductivity, mechanical stability, and interfacial properties. These polymer electrolytes can be solid, gel, or composite materials that provide better safety characteristics and cycling performance. The polymer matrix helps prevent short circuits and enables stable recharge operations over extended periods.Expand Specific Solutions03 Comparative performance testing methodologies
Methods and systems for evaluating and comparing recharge reliability between different battery chemistries including zinc and polymer-based technologies. These approaches involve standardized testing protocols, accelerated aging studies, and performance metrics that assess capacity retention, charge acceptance, and cycle stability. Testing frameworks enable objective comparison of recharge characteristics across battery types.Expand Specific Solutions04 Hybrid zinc-polymer battery architectures
Battery designs that combine zinc-based active materials with polymer components to leverage advantages of both technologies for improved recharge reliability. These hybrid systems may incorporate polymer binders, separators, or electrolytes with zinc electrodes to enhance cycling stability and charge efficiency. The integration addresses limitations of each individual technology while optimizing overall rechargeable performance.Expand Specific Solutions05 Charging control systems for enhanced reliability
Advanced charging algorithms and control systems specifically designed to optimize recharge reliability for zinc and polymer-based batteries. These systems monitor battery parameters during charging and adjust current, voltage, and timing to prevent degradation mechanisms. Smart charging protocols extend battery life by managing charge rates and preventing overcharge conditions that compromise recharge reliability.Expand Specific Solutions
Key Players in Zinc and Polymer Battery Industry
The zinc versus polymer-based rechargeable battery sector represents an emerging competitive landscape characterized by early-stage market development and significant technological differentiation. The market remains relatively nascent with substantial growth potential, driven by increasing demand for safer, more sustainable energy storage solutions. Technology maturity varies considerably across players, with established companies like FDK Corp., Resonac Corp., and Energizer Brands LLC leveraging decades of battery manufacturing expertise, while specialized zinc-focused innovators such as ZPower LLC, Salient Energy Inc., and Offgrid Energy Labs demonstrate advanced zinc-ion and zinc-gel technologies. Academic institutions including Zhejiang University, Central South University, and University of Waterloo contribute fundamental research breakthroughs, particularly in zinc electrode materials and electrolyte formulations. The competitive dynamics favor companies with proven manufacturing capabilities and intellectual property portfolios, as reliability challenges in recharge cycles remain the primary technical barrier requiring sophisticated materials engineering and battery management systems.
Resonac Corp.
Technical Solution: Resonac Corporation develops advanced zinc-polymer battery systems that combine zinc anodes with solid polymer electrolytes for improved recharge reliability and safety. Their technology utilizes cross-linked polymer membranes that provide selective ion transport while blocking zinc dendrite penetration during charging cycles. The company's approach includes specialized polymer formulations with high ionic conductivity and mechanical strength to maintain separator integrity throughout battery life. Their systems achieve over 1000 charge cycles with stable capacity retention through controlled zinc electrodeposition and optimized polymer-electrolyte interfaces. The solid-state design eliminates electrolyte leakage concerns and provides enhanced thermal stability compared to liquid electrolyte systems, making them suitable for applications requiring long-term reliability.
Strengths: Solid polymer electrolyte prevents leakage, good thermal stability, over 1000 cycle capability. Weaknesses: Higher manufacturing complexity, lower ionic conductivity than liquid electrolytes at low temperatures.
ZPower LLC
Technical Solution: ZPower specializes in rechargeable silver-zinc battery technology that offers superior recharge reliability compared to traditional zinc-air systems. Their proprietary electrolyte management system prevents zinc dendrite formation during charging cycles, achieving over 500 charge-discharge cycles with minimal capacity degradation. The company's sealed cell design eliminates electrolyte leakage issues common in zinc-based batteries, while maintaining high energy density of 130-150 Wh/kg. Their advanced separator technology and optimized charging algorithms ensure consistent performance across temperature ranges from -20°C to 60°C, making them suitable for critical applications requiring reliable recharging capabilities.
Strengths: Proven cycle life exceeding 500 cycles, sealed design prevents leakage, excellent temperature performance. Weaknesses: Higher cost than conventional zinc batteries, limited scalability for large-format applications.
Core Innovations in Zinc vs Polymer Recharge Systems
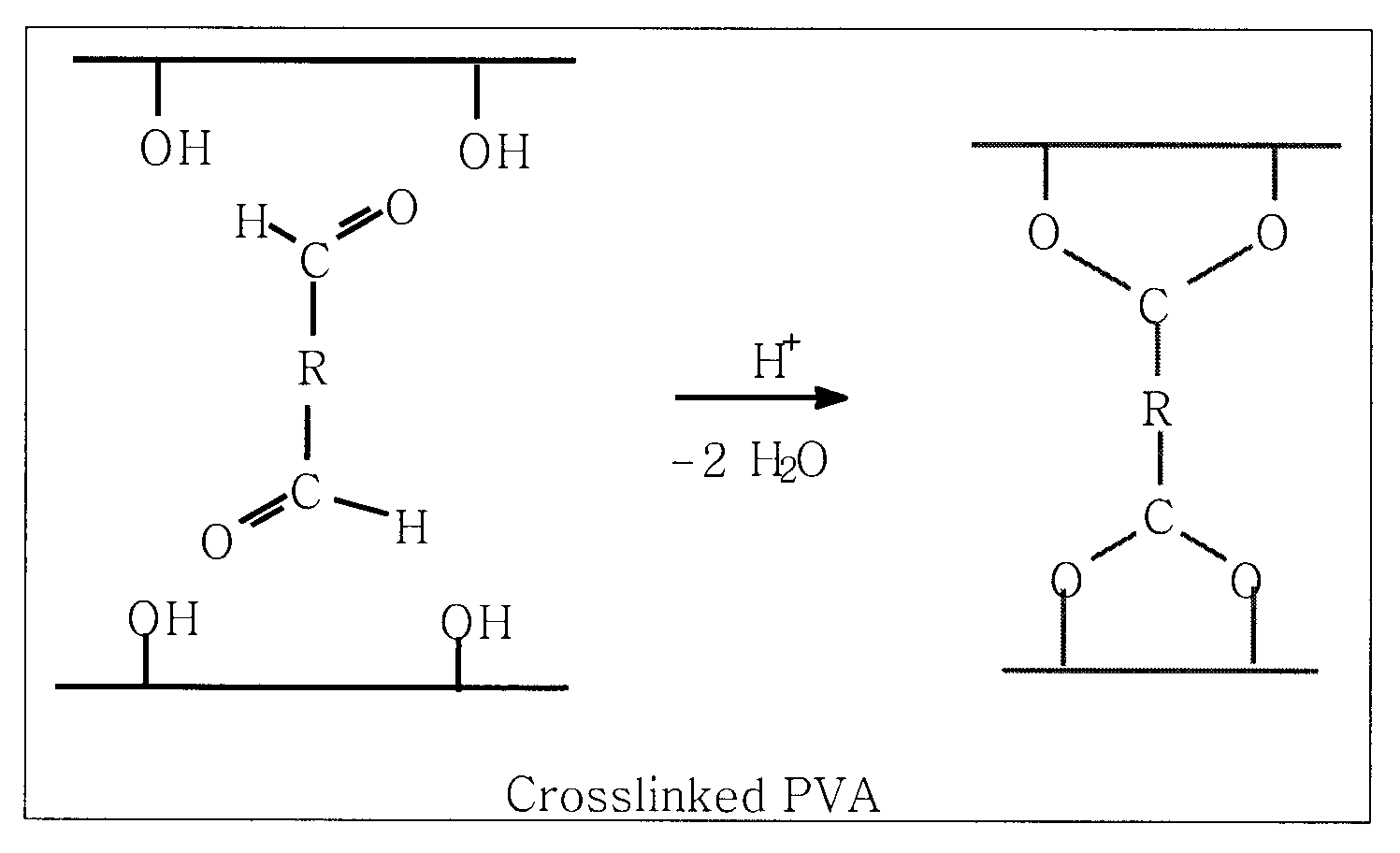
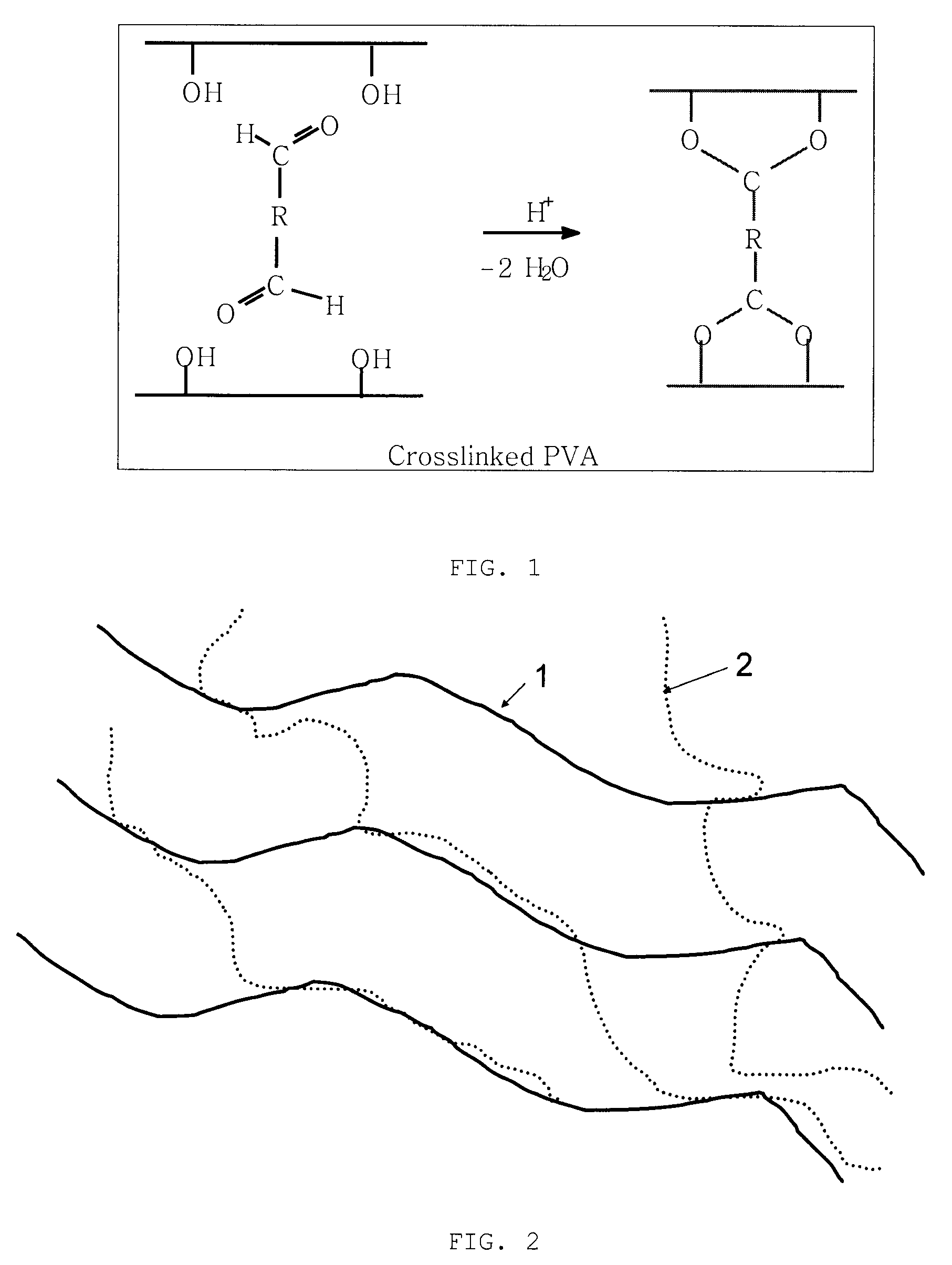
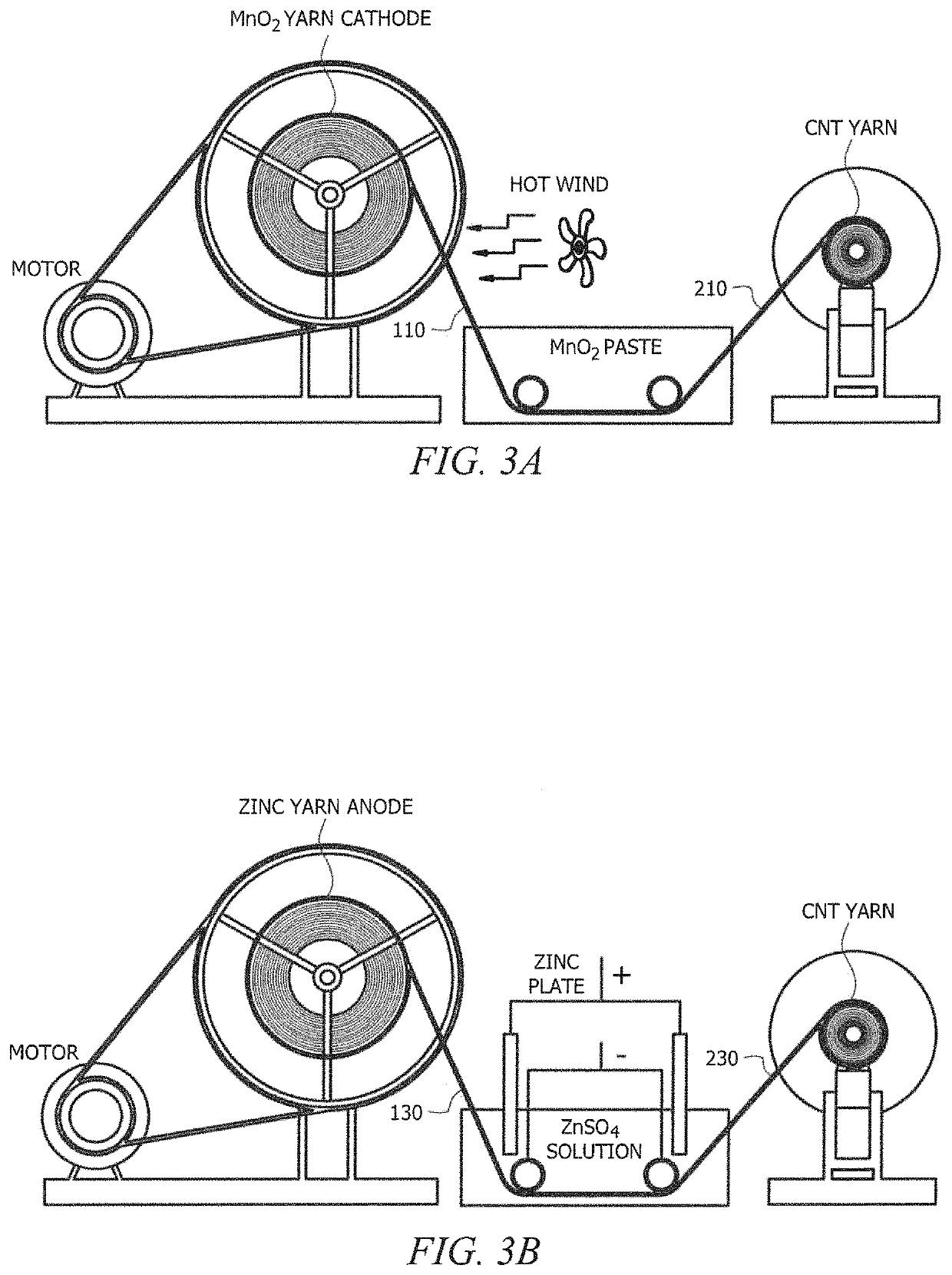
Polymer membrane utilized as a separator in rechargeable zinc cells
PatentInactiveUS20100062342A1
Innovation
- A separator membrane is developed using an interpenetrating polymer network of two different polymers, with polyvinyl alcohol or fluoro-substituted PVA and a water-soluble polymer like polyacrylic acid, combined with nanosized inorganic particles, to form a cross-linked structure that enhances mechanical strength, water absorbance, and ionic conductivity, preventing dendrite growth and electrode shape change.
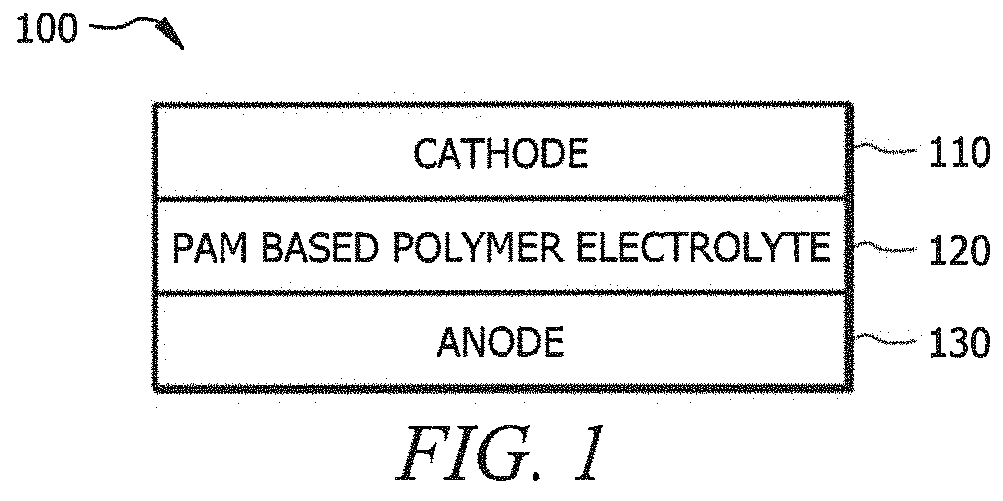
Rechargeable polyacrylamide based polymer electrolyte zinc-ion batteries
PatentActiveUS10957939B2
Innovation
- A polyacrylamide (PAM) based polymer electrolyte is used to create a flexible and rechargeable zinc-ion battery configuration with a highly porous 3D architecture, providing ultra-high ionic conductivity and mechanical strength, combining with a manganese-dioxide cathode and zinc anode to form a hydrogel electrolyte.
Environmental Impact of Battery Material Choices
The environmental implications of battery material choices between zinc-based and polymer-based systems present distinct sustainability profiles that significantly influence their long-term viability. Zinc-based batteries demonstrate superior environmental credentials due to zinc's abundant natural occurrence and established recycling infrastructure. Zinc extraction requires substantially lower energy consumption compared to lithium or cobalt mining, resulting in reduced carbon footprint during raw material procurement. The metal's non-toxic nature eliminates concerns about groundwater contamination and soil degradation typically associated with heavy metal battery chemistries.
Polymer-based battery systems present a more complex environmental landscape. While synthetic polymers offer design flexibility and potentially longer service life, their production relies heavily on petrochemical feedstocks, contributing to fossil fuel dependency. The manufacturing process of specialized battery polymers often involves energy-intensive polymerization reactions and requires sophisticated purification steps, increasing the overall carbon footprint. However, recent advances in bio-based polymer synthesis and recyclable polymer architectures are beginning to address these environmental concerns.
End-of-life management represents a critical differentiator between these technologies. Zinc-based systems benefit from well-established recycling networks where zinc can be recovered with high efficiency and reused in various applications without quality degradation. The recycling process is relatively straightforward, involving pyrometallurgical or hydrometallurgical methods that have been refined over decades. This circular economy approach significantly reduces the environmental burden associated with primary zinc production.
Polymer-based batteries face greater challenges in end-of-life processing. Traditional polymer recycling methods often result in material degradation, limiting the quality of recovered materials. Chemical recycling approaches, while promising, remain energy-intensive and economically challenging at scale. However, emerging depolymerization technologies and design-for-recycling strategies are showing potential to improve the environmental profile of polymer-based systems.
The manufacturing phase environmental impact varies significantly between these technologies. Zinc-based battery production typically involves conventional metallurgical processes with well-understood environmental controls. Polymer-based systems require specialized clean room environments and precise chemical processing, often resulting in higher energy consumption and waste generation during manufacturing.
Water usage and contamination potential differ markedly between these approaches. Zinc processing, while requiring water for purification and electrolyte preparation, generates manageable waste streams that can be treated using established methods. Polymer synthesis and processing may involve organic solvents and catalysts that require more sophisticated waste treatment systems to prevent environmental contamination.
Polymer-based battery systems present a more complex environmental landscape. While synthetic polymers offer design flexibility and potentially longer service life, their production relies heavily on petrochemical feedstocks, contributing to fossil fuel dependency. The manufacturing process of specialized battery polymers often involves energy-intensive polymerization reactions and requires sophisticated purification steps, increasing the overall carbon footprint. However, recent advances in bio-based polymer synthesis and recyclable polymer architectures are beginning to address these environmental concerns.
End-of-life management represents a critical differentiator between these technologies. Zinc-based systems benefit from well-established recycling networks where zinc can be recovered with high efficiency and reused in various applications without quality degradation. The recycling process is relatively straightforward, involving pyrometallurgical or hydrometallurgical methods that have been refined over decades. This circular economy approach significantly reduces the environmental burden associated with primary zinc production.
Polymer-based batteries face greater challenges in end-of-life processing. Traditional polymer recycling methods often result in material degradation, limiting the quality of recovered materials. Chemical recycling approaches, while promising, remain energy-intensive and economically challenging at scale. However, emerging depolymerization technologies and design-for-recycling strategies are showing potential to improve the environmental profile of polymer-based systems.
The manufacturing phase environmental impact varies significantly between these technologies. Zinc-based battery production typically involves conventional metallurgical processes with well-understood environmental controls. Polymer-based systems require specialized clean room environments and precise chemical processing, often resulting in higher energy consumption and waste generation during manufacturing.
Water usage and contamination potential differ markedly between these approaches. Zinc processing, while requiring water for purification and electrolyte preparation, generates manageable waste streams that can be treated using established methods. Polymer synthesis and processing may involve organic solvents and catalysts that require more sophisticated waste treatment systems to prevent environmental contamination.
Safety Standards for Rechargeable Battery Systems
The safety standards governing rechargeable battery systems have evolved significantly to address the distinct characteristics and risks associated with different battery chemistries, particularly zinc-based and polymer-based technologies. These standards establish comprehensive frameworks for evaluating recharge reliability while ensuring operational safety across various applications.
International standards such as IEC 62133, UL 2054, and UN 38.3 provide foundational safety requirements for rechargeable batteries, with specific provisions addressing charge cycling performance and thermal management. For zinc-based systems, standards emphasize dendrite formation prevention, electrolyte stability during repeated charging, and capacity retention metrics. The IEC 62620 standard specifically addresses secondary lithium cells and batteries, while emerging standards are being developed to accommodate zinc-ion and zinc-air technologies.
Polymer-based battery systems fall under specialized safety protocols that focus on mechanical integrity, thermal runaway prevention, and electrolyte containment. Standards like IEC 62660 for lithium-ion traction battery packs incorporate rigorous testing for charge-discharge cycling, overcharge protection, and thermal abuse scenarios. These standards mandate specific testing protocols including nail penetration, crush tests, and extended cycling evaluations to assess long-term reliability.
Safety certification processes require comprehensive evaluation of charging algorithms, battery management systems, and fail-safe mechanisms. Standards mandate minimum performance thresholds for cycle life, typically requiring batteries to maintain 80% capacity after specified charge cycles. For zinc systems, this often involves 500-1000 cycles, while polymer systems may require demonstration of 2000+ cycles depending on application requirements.
Regulatory compliance involves multiple testing phases including electrical safety, environmental stress testing, and abuse tolerance evaluation. Standards specify maximum allowable temperature rises during charging, gas emission limits, and mechanical deformation thresholds. These requirements directly impact the design of charging infrastructure and battery management systems for both zinc and polymer technologies.
Emerging safety standards are increasingly incorporating smart charging protocols and real-time monitoring requirements to enhance recharge reliability. These evolving standards recognize the need for adaptive charging strategies that optimize both safety and longevity across different battery chemistries.
International standards such as IEC 62133, UL 2054, and UN 38.3 provide foundational safety requirements for rechargeable batteries, with specific provisions addressing charge cycling performance and thermal management. For zinc-based systems, standards emphasize dendrite formation prevention, electrolyte stability during repeated charging, and capacity retention metrics. The IEC 62620 standard specifically addresses secondary lithium cells and batteries, while emerging standards are being developed to accommodate zinc-ion and zinc-air technologies.
Polymer-based battery systems fall under specialized safety protocols that focus on mechanical integrity, thermal runaway prevention, and electrolyte containment. Standards like IEC 62660 for lithium-ion traction battery packs incorporate rigorous testing for charge-discharge cycling, overcharge protection, and thermal abuse scenarios. These standards mandate specific testing protocols including nail penetration, crush tests, and extended cycling evaluations to assess long-term reliability.
Safety certification processes require comprehensive evaluation of charging algorithms, battery management systems, and fail-safe mechanisms. Standards mandate minimum performance thresholds for cycle life, typically requiring batteries to maintain 80% capacity after specified charge cycles. For zinc systems, this often involves 500-1000 cycles, while polymer systems may require demonstration of 2000+ cycles depending on application requirements.
Regulatory compliance involves multiple testing phases including electrical safety, environmental stress testing, and abuse tolerance evaluation. Standards specify maximum allowable temperature rises during charging, gas emission limits, and mechanical deformation thresholds. These requirements directly impact the design of charging infrastructure and battery management systems for both zinc and polymer technologies.
Emerging safety standards are increasingly incorporating smart charging protocols and real-time monitoring requirements to enhance recharge reliability. These evolving standards recognize the need for adaptive charging strategies that optimize both safety and longevity across different battery chemistries.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!