Metal Hydride vs Solid-State Storage: Which Is More Suitable for Portable Hydrogen Applications?
JUL 21, 2025 |
As the world shifts towards cleaner energy solutions, hydrogen has emerged as a promising alternative. Its high energy content, combined with the fact that it produces only water as a by-product when used in fuel cells, makes it an attractive option. However, one of the critical challenges in harnessing hydrogen's potential lies in its storage. Two of the primary methods for storing hydrogen, especially for portable applications, include metal hydride and solid-state storage. Each method has its own set of advantages and challenges, making the choice between them crucial depending on the specific requirements of the application.
Understanding Metal Hydride Storage
Metal hydrides store hydrogen by chemically bonding it with metals. This process occurs at relatively low pressures and ambient temperatures, making it a safer option compared to high-pressure gas storage. The metal hydride method involves a reversible reaction where hydrogen gas is absorbed by metals like titanium, magnesium, or nickel, forming a compound. When hydrogen is needed, the process is reversed, and hydrogen is released by applying heat.
One of the key benefits of metal hydride storage is its ability to store a significant amount of hydrogen within a small volume. This high density is particularly advantageous for portable applications where space is limited. However, the primary drawback is the weight; metal hydrides can be relatively heavy, which can limit their use in applications where weight is a critical factor, such as in aviation or portable fuel cells for electronic devices.
Exploring Solid-State Storage
Solid-state hydrogen storage involves storing hydrogen in a compact, solid material matrix, such as in the form of chemical compounds or nanostructured materials. These solid-state systems often utilize materials like metal-organic frameworks (MOFs) or carbon-based nanostructures to adsorb and release hydrogen.
One of the most significant advantages of solid-state storage is its potential for achieving higher gravimetric capacities compared to metal hydrides. This means that, weight-for-weight, solid-state storage can potentially store more hydrogen. Moreover, solid-state storage systems can operate at ambient conditions, which reduces the need for complex temperature and pressure controls.
However, the technology is still largely in the research and development phase, and there are challenges related to the kinetics of hydrogen absorption and desorption. The materials must also be engineered to ensure they are stable over many cycles of hydrogen storage and release, which is crucial for practical applications.
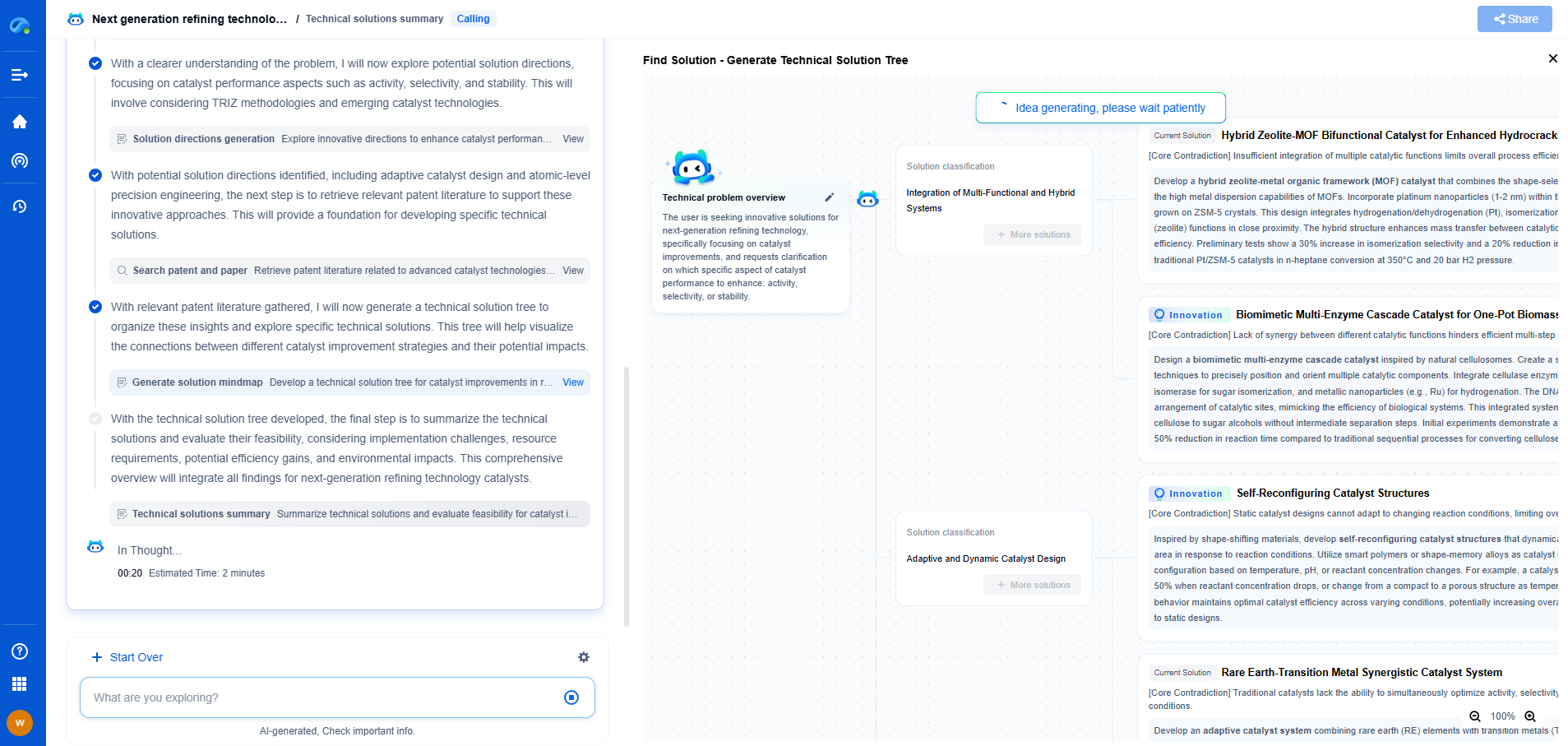
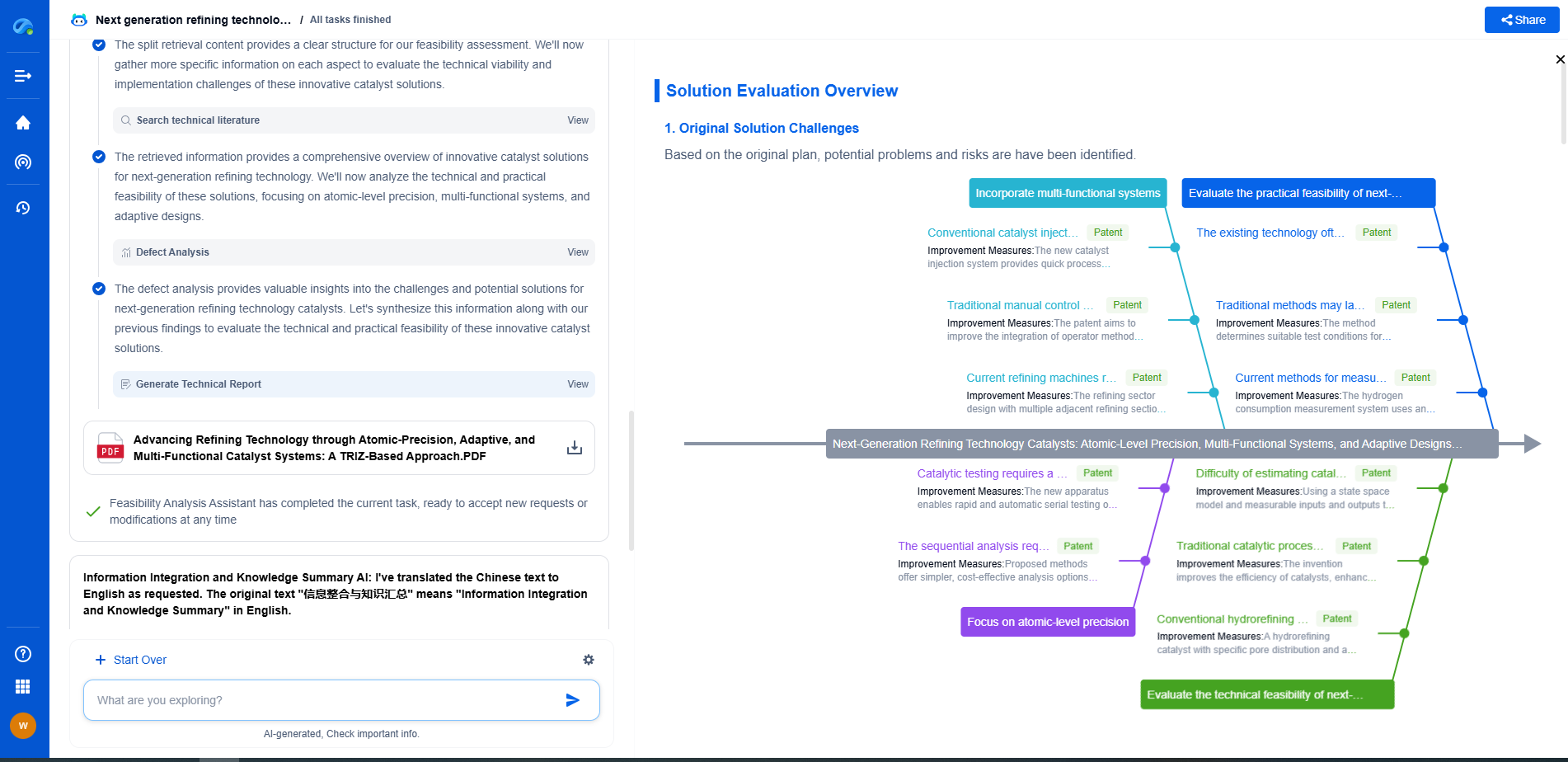
Comparative Analysis: Suitability for Portable Applications
When comparing metal hydride and solid-state storage for portable hydrogen applications, several factors must be considered: weight, volume, energy density, and system complexity.
Metal hydride systems, though heavier, offer a mature technology with established safety profiles. They are particularly well-suited for applications where safety and compactness are prioritized over weight constraints. Devices that can accommodate the additional weight, such as stationary power units or certain types of vehicles, might benefit significantly from the robust nature of metal hydride storage.
On the other hand, solid-state storage presents a promising future for applications where reducing weight is crucial. For instance, in portable electronics or lightweight vehicles, the potential for higher gravimetric capacity makes solid-state storage an attractive option. However, the nascent stage of this technology means that its implementation might require substantial advancements and cost reductions before it becomes commercially viable.
Conclusion and Outlook
The choice between metal hydride and solid-state hydrogen storage for portable applications hinges on balancing current technological capabilities with specific application needs. While metal hydrides offer a practical and safe solution for many existing applications, solid-state storage holds the promise of more efficient and lightweight systems in the future.
As research continues to advance in both areas, it is likely that new materials and technologies will emerge, further blurring the lines between these two storage methods. For now, the decision largely depends on the specific requirements of the application, including factors such as weight constraints, energy efficiency, and cost considerations. As the landscape of hydrogen storage evolves, keeping abreast of technological developments will be key to making informed decisions in this exciting field.
As clean energy and decarbonization drive new breakthroughs in hydrogen storage, CO₂ transport, and alternative gas carriers, keeping pace with technical trends and patent activity is critical to staying competitive.
Patsnap Eureka helps innovators in compressed gas storage, high-pressure tank design, gas sensor systems, and pipeline materials accelerate research by offering instant, AI-powered insights into global patents, related technologies, and emerging white spaces.
🚀 Bring speed, precision, and strategic foresight to your innovation and IP decision-making in the gas transport sector—try Eureka today and unlock a smarter path forward.
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com