
Tembotrione is a selective herbicide belonging to the triketone class and is used primarily in corn cultivation to control broadleaf and grassy weeds. Its effectiveness lies in its ability to inhibit 4-hydroxyphenylpyruvate dioxygenase (HPPD), an enzyme crucial for carotenoid biosynthesis in plants. Understanding the synthesis of tembotrione is essential for agrochemical research and manufacturing. This article outlines the chemical structure, key reagents, and synthetic steps used in the production of tembotrione, highlighting its relevance in crop protection chemistry.
Get Started with PatSnap Eureka AI Agent
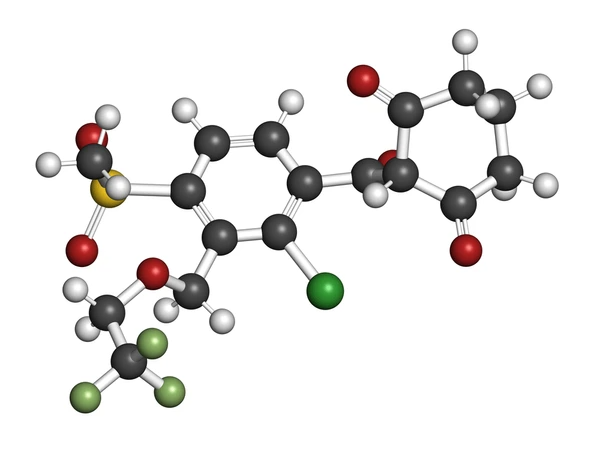
Tembotrione Chemical Structure
What is Tembotrione synthesis? Eureka Technical Q&A explains the chemical synthesis process of tembotrione, a selective herbicide, including key intermediates, functional group transformations, and its role in crop protection through targeted HPPD enzyme inhibition.
Tembotrione is a herbicidal compound with the chemical structure C17H16ClF3O6S. It is characterized by a cyclohexanedione core with a 2-chloro-4-methylsulfonyl group and a 3-(2,2,2-trifluoroethoxymethyl)benzoyl group attached to it. This unique structure contributes to its herbicidal properties, allowing it to inhibit the enzyme 4-hydroxyphenylpyruvate dioxygenase (HPPD) in plants, which is crucial for the biosynthesis of prenylquinones and carotenoids necessary for photosynthesis .
The chemical structure of tembotrione can be broken down as follows:
- The core is a 1,3-cyclohexanedione.
- Attached to this core is a 2-chloro-4-methylsulfonyl group.
- There is also a 3-(2,2,2-trifluoroethoxymethyl)benzoyl group.
This complex structure is responsible for the compound’s selective toxicity towards weeds while being safe for corn crops when used in combination with the safener isoxadifen-ethyl.
Reagents and Reactions
While the exact reagents and reactions used in the synthesis of tembotrione are not detailed in the retrieved information, the synthesis of similar triketone herbicides typically involves:
- Formation of the Core Structure: This often involves the reaction of a beta-keto ester with a chloroformate or similar reagent to form the triketone core.
- Introduction of Functional Groups: The trifluoroethoxymethyl and methylsulfonyl groups are introduced through specific substitution reactions.
- Purification and Crystallization: The final product is purified and crystallized to obtain the desired crystalline form, which is crucial for its formulation as a herbicide.
Key Synthetic Strategy of Tembotrione
Tembotrione is a herbicidal compound used primarily for controlling grass and broad-leaved weeds in corn production. Its synthesis involves several key steps and intermediates, which are crucial for achieving the desired herbicidal activity and stability.
- Synthesis Overview: The synthesis of tembotrione typically involves the preparation of key intermediates followed by cyclization and functional group transformations. The process often includes the use of specific catalysts and reagents to ensure high yield and purity of the final product.
- Key Intermediates and Reactions:
- 2-Nitro-4-methylsulfonyl toluene (NMST) Oxidation: This intermediate is oxidized using an oxidant in the presence of ruthenium (IV) oxide (RuO2) as a catalyst to produce 2-nitro-4-methylsulfonyl benzoic acid (NMSBA).
- Enol Ester Formation: The NMSBA is then reacted with 1,3-cyclohexanedione to form an enol ester intermediate.
- Cyclization and Functional Group Transformations: The enol ester is further converted to tembotrione through cyclization and other functional group modifications.
- Crystalline Forms: Tembotrione can exist in different crystalline forms, which are important for its stability and formulation in plant protection products. The crystalline forms can affect the solubility, stability, and efficacy of the herbicide.
Step-by-Step Synthesis Overview
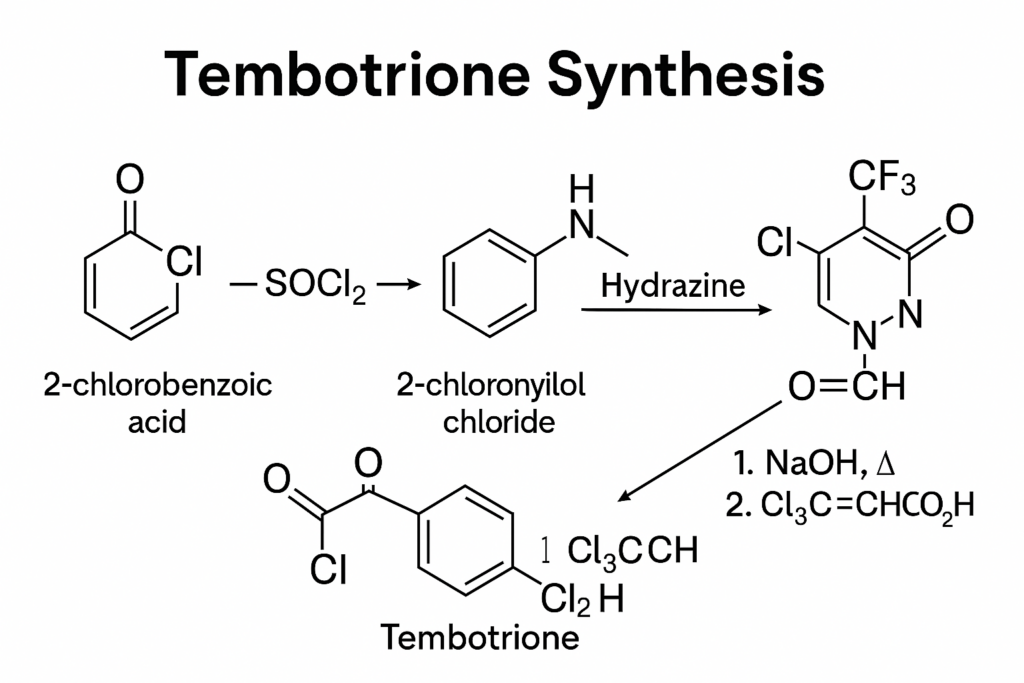
- Starting Materials: The synthesis of tembotrione typically begins with the preparation of key intermediates. These may include chlorinated benzoyl compounds and cyclohexanedione derivatives, which are essential for the core structure of tembotrione.
- Formation of the Core Structure: The synthesis involves the coupling of a chloro-substituted benzoyl group with a cyclohexanedione moiety. This step is critical as it forms the backbone of the tembotrione molecule. The reaction conditions, such as temperature, solvent, and catalyst, are optimized to ensure high yield and purity of the intermediate.
- Introduction of the Methylsulfonyl Group: The next step includes the introduction of a methylsulfonyl group at the para position of the benzoyl ring. This is typically achieved through a sulfonation reaction, where a methylsulfonyl chloride derivative is introduced under controlled conditions to avoid over-sulfonation or side reactions.
- Attachment of the Trifluoroethoxy Methyl Group: The trifluoroethoxy methyl group is then attached to the benzoyl ring. This is usually done through an etherification reaction, where the trifluoroethoxy methyl chloride is reacted with the benzoyl intermediate in the presence of a base to facilitate the formation of the ether bond.
- Final Coupling and Purification: The final step involves coupling the modified benzoyl compound with the cyclohexanedione derivative. This is followed by purification processes such as crystallization or chromatography to obtain tembotrione in its pure form.
- Quality Control and Characterization: The synthesized tembotrione undergoes rigorous quality control and characterization using techniques like NMR, Mass Spectrometry, and HPLC to confirm its structure, purity, and potency.
Applications of Tembotrione
Mode of Action
Tembotrione inhibits the enzyme 4-hydroxyphenylpyruvate dioxygenase (HPPD), disrupting the production of prenylquinones and tocopherols. These compounds are essential for photosynthesis and plant stress tolerance. As a result, weeds develop bleached, stunted tissues, which halts their growth effectively.
Application Stages
Farmers apply tembotrione between crop emergence and the V8 growth stage of corn. This timing maximizes foliar absorption and ensures effective internal distribution, stopping weed development efficiently.
Tank Mix Applications
Growers often tank-mix tembotrione with herbicides like atrazine to expand weed control and lower crop injury risks. This combination boosts overall effectiveness and broadens the spectrum of controlled weeds.
Environmental Considerations
Tembotrione performs best in light soils and under neutral to slightly alkaline pH conditions. Its high water solubility enhances weed control but also raises leaching risks. Therefore, farmers must manage its use carefully to avoid contaminating water sources.
Phytotoxicity and Safety
Tembotrione alone can harm maize. To reduce this risk, manufacturers pair it with the safener isoxadifen-ethyl. This compound speeds up herbicide breakdown in the crop, preventing injury.
Efficacy and Persistence
Tembotrione delivers fast, powerful weed control, especially in bright sunlight. It follows first-order dissipation kinetics in soil, with residues typically disappearing within 45 to 60 days, depending on the dose and soil type.
Regulatory Status
Regulators have approved tembotrione mainly for corn. Its use in other crops, such as grain sorghum, remains limited while researchers assess crop safety and selectivity.
FAQs
It belongs to the HPPD-inhibitor class, specifically the triketone herbicides.
It enables the molecule to bind effectively to the HPPD enzyme, disrupting carotenoid biosynthesis in weeds.
Yes, the synthesis is suitable for industrial scale-up with controlled reaction conditions and purification methods.
Key intermediates include the methylsulfonylbenzoyl chloride and the cyclohexanedione core compound.
Conclusion
The synthesis of tembotrione is a strategically designed process involving oxidation, acylation, and triketone formation, resulting in a highly active herbicide. Its chemical structure and mode of action make it an essential tool in modern crop protection, especially in corn agriculture. Understanding the reagents and synthetic steps behind tembotrione helps both chemists and agronomists explore new solutions in herbicide development and application.
How PatSnap Eureka Accelerates Tembotrione Synthesis Innovation
Tembotrione synthesis plays a key role in developing next-generation herbicides with improved selectivity and environmental profiles. As regulatory pressures and market demands evolve, PatSnap Eureka provides researchers and formulation scientists with a powerful platform to stay ahead of innovation.
- Patent Intelligence: Eureka scans global patent filings to uncover advancements in Tembotrione synthesis, including novel synthetic routes, intermediates, and process optimizations—helping reduce R&D redundancy and accelerate development.
- Competitive Landscape Tracking: Analyze how agrochemical leaders are innovating around Tembotrione, from synthesis scalability to formulation strategies, gaining strategic insights into competitive positioning.
- Trend Forecasting: With AI-driven analytics, Eureka highlights emerging trends in herbicide development where Tembotrione derivatives and analogs are gaining traction, supporting long-term innovation planning.
- Technical Clustering: Visual clustering reveals synthesis-related innovation hotspots, such as greener production methods, chiral catalyst applications, and integration into combination formulations.
By transforming complex IP data into actionable insight, Eureka empowers faster, smarter breakthroughs in tembotrione synthesis across the agrochemical landscape.
To get detailed scientific explanations of Tembotrione Synthesis, try Patsnap Eureka.

